Clear Sky Science · en
Superacid-resistant macrocyclic BODIPYs
Glowing Colors That Survive in Harsh Acids
Fluorescent dyes are the invisible highlighters of modern science, helping researchers track cells, map materials, and follow chemical reactions. But most of these dyes quickly fall apart in strong acids, losing their brilliant glow just where they could be most useful, such as inside powerful catalysts or highly acidic industrial materials. This paper reports a new family of dyes that keep shining even in some of the strongest known acids, opening the door to imaging and sensing in chemical environments that were previously off‑limits.
Why Ordinary Dyes Go Dark
One of the most popular dye families, known as BODIPYs, normally offers sharp colors and bright emission, making them favorites for biological imaging and chemical sensing. Their Achilles’ heel is acid: in strong acidic solutions, the central boron atom that anchors the dye’s light‑absorbing framework is easily kicked out, a process called deboronation. Once this happens, the orderly electronic structure that produces clean color and strong glow collapses, and the dye becomes dull or completely dark. That limitation has largely confined BODIPY use to mildly acidic or neutral surroundings and has blocked their use in very acidic materials like fuel‑cell membranes or superacidic catalysts.
Building a Protective Ring Around the Light
The researchers tackled this weakness by redesigning the microscopic “scaffolding” that holds the boron atom in place. They drew inspiration from related ring‑shaped molecules that grip boron so tightly they resist even harsh chemical treatment. By embedding a BODIPY‑like light‑absorbing unit into a three‑part ring, called a tripyrrolic macrocycle, they created what they term superacid‑resistant macrocyclic BODIPYs. In this architecture, three nitrogen‑bearing rings form a snug cradle around boron, while a separate pyrrole unit can accept extra protons from acid. Careful spectroscopic measurements and computer calculations show that, instead of ejecting boron, strong acids merely protonate this outer ring, leaving the core light‑emitting unit almost untouched.
Acid Turns the Light On, Not Off
Surprisingly, strong acid does more than just spare these new dyes—it actively switches their glow on. In neutral liquids, the dyes absorb visible light but hardly fluoresce. An internal fragment donates electrons in a way that drains the excited state before it can emit light. When acid is added, that fragment is protonated and its electron‑donating power is weakened, shutting down the quenching route. The result is a bright "turn‑on" signal: in superacids such as methanesulfonic, sulfuric, chlorosulfonic, and even fluorosulfonic acid, the dyes reach fluorescence efficiencies up to 90%, and they maintain their brightness for over a day. Tests against standard BODIPYs and specially strengthened variants show that the new macrocyclic versions far outlast them in both acidity and high temperatures, and they also resist damage under prolonged illumination.
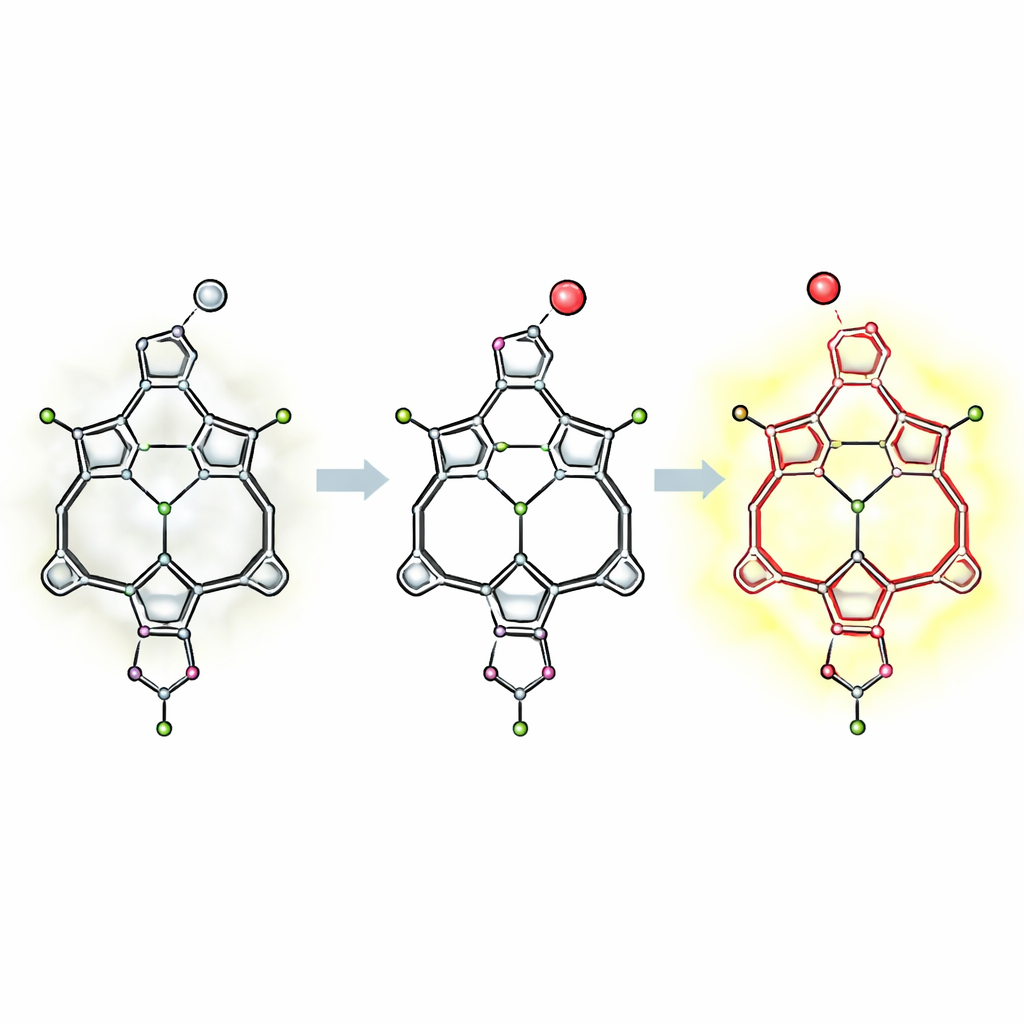
Tuning Color and Behavior Without Breaking the Core
Because the central framework is so robust, the team could decorate the dye’s outer positions with additional chemical groups to tune color, lifetime, and solubility without sacrificing acid resistance. Adding bromine atoms introduces heavy‑atom effects that encourage a long‑lived triplet state, making one derivative a modest generator of reactive oxygen species when lit—useful for applications like photodynamic therapy or photocatalysis. Attaching various aryl groups shifts the emission from yellow toward red and alters how efficiently the dyes glow, especially in thick, viscous acids where molecular motion is restricted. The boron atom can also trade its axial ligand for a fluorinated tail, yielding a version that dissolves in fluorous solvents and lights up in response to a perfluorinated acid pollutant, hinting at sensors for specialty chemical phases.
Lighting Up Strongly Acidic Materials
To showcase real‑world potential, the authors stained several notoriously harsh materials. Nafion beads, used as solid electrolytes and acid catalysts, are so acidic that conventional BODIPYs quickly lose their fluorescence. In contrast, the new macrocyclic dye soaked into Nafion and produced a stable, bright reddish‑orange glow for at least a week, fading only when exposed to base vapor and recovering when re‑acidified. Similar behavior was seen with sulfonylated ion‑exchange resins and strongly acidic double‑network gels: the dye could be immobilized, switched between bright and dark forms by acid–base treatment, and used to visually track how a neutralizing solution penetrated through a gel block over time. These demonstrations show that the dyes can act as built‑in acid indicators inside solid or gel‑like materials.
What This Means for Future Fluorescent Tools
By cleverly pairing a boron center with a protective three‑ring macrocycle, this work delivers fluorescent dyes that defy some of the strongest acids chemists can handle, all while keeping the sharp colors and high brightness that make BODIPYs attractive. Strong acid, once the enemy of these dyes, becomes a convenient switch that turns their fluorescence on and off without breaking the molecule. This design principle offers a roadmap for building other rugged dyes that operate in extreme environments, from industrial catalysts and fuel‑cell membranes to geological minerals and acid‑loving microorganisms. In short, the authors extend the reach of fluorescence imaging into chemical territory where ordinary dyes simply cannot survive.
Citation: Watanabe, K., Honda, G., Terauchi, Y. et al. Superacid-resistant macrocyclic BODIPYs. Nat Commun 17, 2332 (2026). https://doi.org/10.1038/s41467-026-70499-9
Keywords: fluorescent dyes, superacid chemistry, BODIPY, materials imaging, chemical sensors