Clear Sky Science · en
Sororin locks the DNA-exit gate of cohesin to preserve sister-chromatid cohesion
Keeping copied chromosomes safely together
Every time a cell divides, it must copy its DNA and then hand one complete set to each daughter cell. If those copies drift apart too soon, the result can be broken chromosomes, genetic chaos, and disease, including cancer. This study uncovers how a small protein piece called Sororin’s tail acts like a physical lock on a ring-shaped clamp that holds sister DNA copies together, explaining how cells keep chromosomes paired until the exact moment of separation.
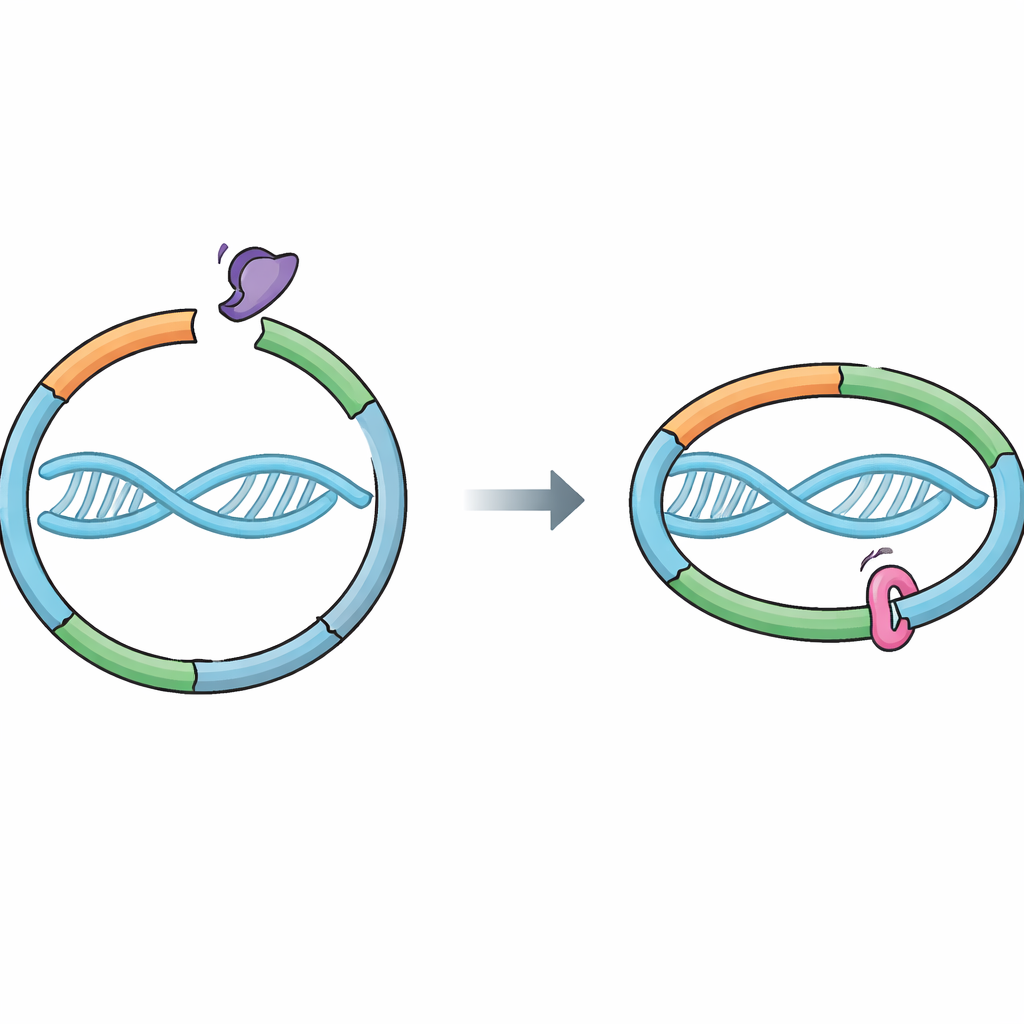
A molecular ring that guards our genes
Cells rely on a protein machine called cohesin to hold newly copied chromosomes together. Cohesin forms a ring that loops around DNA, linking the two sister strands created during DNA replication. This link must be strong enough to survive all the jostling of cell division, yet releasable at the right time so that sisters can separate cleanly. A helper protein, Wapl, promotes opening of a specific “exit gate” in the ring where two cohesin parts, RAD21 and SMC3, meet. Opening this gate allows DNA to slip out and cohesin to fall off. Until now, Sororin was thought to preserve cohesion mainly by blocking Wapl’s partner Pds5, but it was unclear whether Sororin also touches the cohesin ring directly.
A tiny tail with big holding power
The authors focused on Sororin’s extreme tail end—the last 30 amino acids, called the C‑terminal region (CTR). By trimming Sororin into pieces and testing them in human cells, they found that this tiny CTR is both necessary and sufficient to keep sister chromosomes paired. When the CTR was artificially anchored at centromeres, the central sites where sisters stay joined longest, it almost completely rescued cohesion in cells lacking full Sororin. Conversely, when the CTR was tethered all along chromatin, cohesin could no longer be efficiently removed, and chromosomes failed to compact and untangle properly. These cells showed long, fuzzy mitotic chromosomes, DNA bridges between separating sisters, and segregation errors—problems that strikingly resemble what happens when Wapl itself is removed.
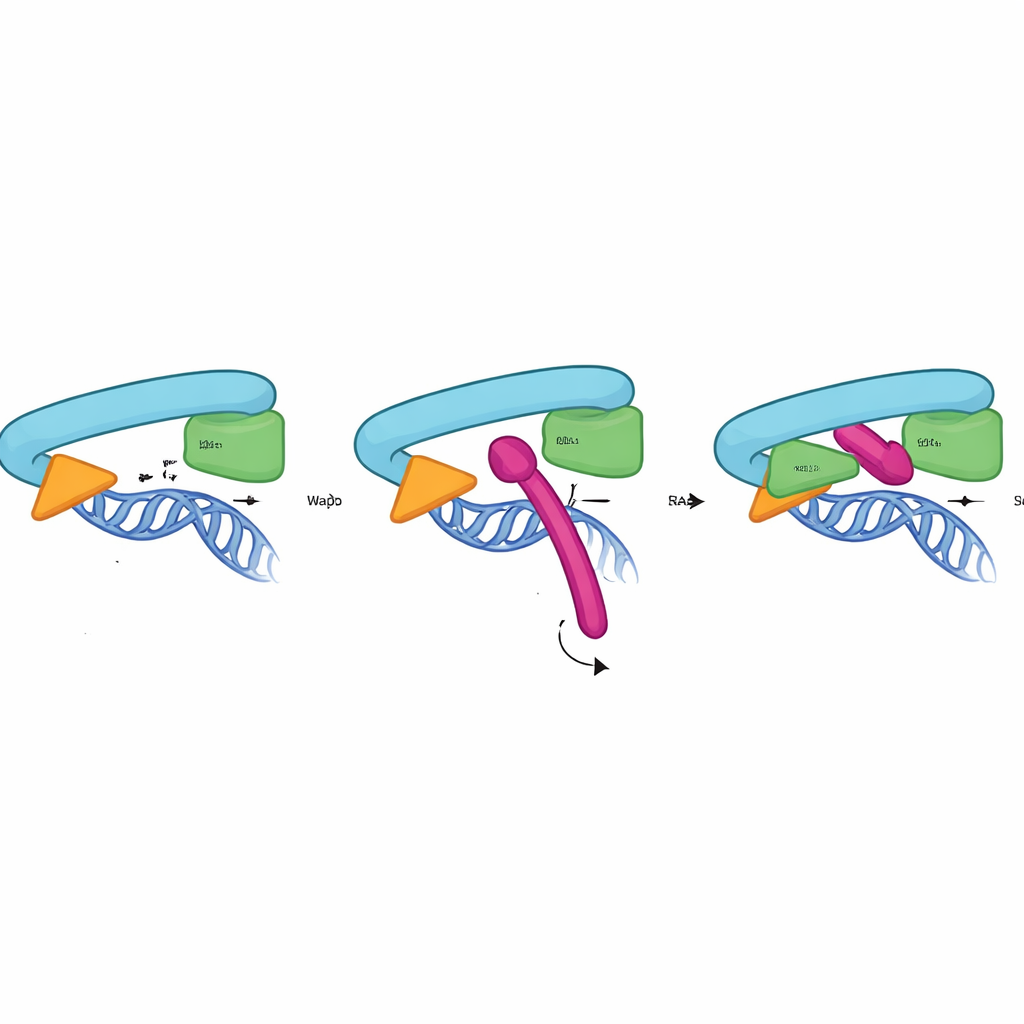
How Sororin’s lock grips the ring
To understand how the CTR works at the atomic level, the researchers combined biochemical tests, mutagenesis, and AlphaFold3 structure predictions. They showed that the CTR binds directly and specifically to the interface between RAD21 and the head region of SMC3—the very site that forms the DNA‑exit gate. This interaction does not require Pds5 or the SA2 subunit and occurs only on chromatin-bound cohesin, peaking in the phase when cohesion must be stably maintained. The structural model suggests that a short helix within the CTR nestles into a pocket formed by RAD21 and SMC3, using a cluster of conserved greasy and negatively charged residues to latch on. Mutating only a few of these contact points in Sororin, RAD21, or SMC3 was enough to abolish binding and cause premature sister separation, confirming that this interface acts as a physical gate lock.
A timed release switch for chromosome separation
Cells must also release cohesin at the right time. The team discovered that Sororin carries a nearby control site, a single amino acid (S145) that becomes phosphorylated by the mitotic kinase Aurora B. This modification specifically breaks Sororin’s grip on Pds5, allowing Wapl to re-engage and promote cohesin removal from chromosome arms, but it leaves the CTR’s contact with the RAD21–SMC3 gate largely intact. At centromeres, another factor, Sgo1, recruits a phosphatase that removes this phosphate, preserving Sororin–Pds5 binding and strong gate locking until the final cut of RAD21 at anaphase. Genetic tests showed that when Wapl is removed, the cohesion defects caused by disrupting the CTR–gate interaction vanish, underscoring that Sororin’s essential job is to shield the exit gate specifically from Wapl-driven opening.
What this means for genome stability and disease
Together, these findings recast Sororin as a dual-function guardian: it both competes with Wapl for access to Pds5 and directly clamps shut cohesin’s DNA‑exit gate via its tiny CTR. This dual control ensures that cohesion is strong where needed, especially at centromeres, yet can be peeled back from chromosome arms in early mitosis to allow proper compaction and untangling. Because mutations in cohesin and its regulators are common in developmental disorders and cancers, the precisely mapped contact surface between Sororin, RAD21, and SMC3 highlights a new vulnerability where small changes can destabilize chromosomes—and a potential target for future therapies that seek to tune how tightly our genetic material is held together.
Citation: Chen, Q., Yuan, X., Shi, M. et al. Sororin locks the DNA-exit gate of cohesin to preserve sister-chromatid cohesion. Nat Commun 17, 2284 (2026). https://doi.org/10.1038/s41467-026-70484-2
Keywords: sister chromatid cohesion, cohesin complex, Sororin, chromosome segregation, genome stability