Clear Sky Science · en
Surface hydrophobicity and rigidity determines protein corona on orally delivered nanoparticles treating colitis
Turning Pills into Smarter Treatments
People living with inflammatory bowel diseases such as colitis often take powerful anti-inflammatory drugs, yet much of each dose is wasted or causes side effects elsewhere in the body. This study explores a new way to make oral medicines act more like guided missiles: by designing tiny drug-carrying particles that recruit the body’s own intestinal proteins as a natural targeting system, helping medicines home in on the immune cells that drive gut inflammation.
How the Gut Dresses Nanoparticles
When any nanoparticle enters the body, it is quickly coated by a thin layer of proteins, forming what scientists call a “protein corona.” In the diseased intestine, this corona looks very different from that in healthy tissue because the local protein mix has changed. The authors previously found that colitis causes a special intestinal corona that modestly steers particles toward immune cells called macrophages, which both fuel and resolve inflammation. Here, they set out to deliberately shape that corona by adjusting two basic features of drug-carrying particles—their water-repellency (hydrophobicity) and their stiffness (rigidity)—to turn a modest, disease-made effect into a potent targeting strategy.
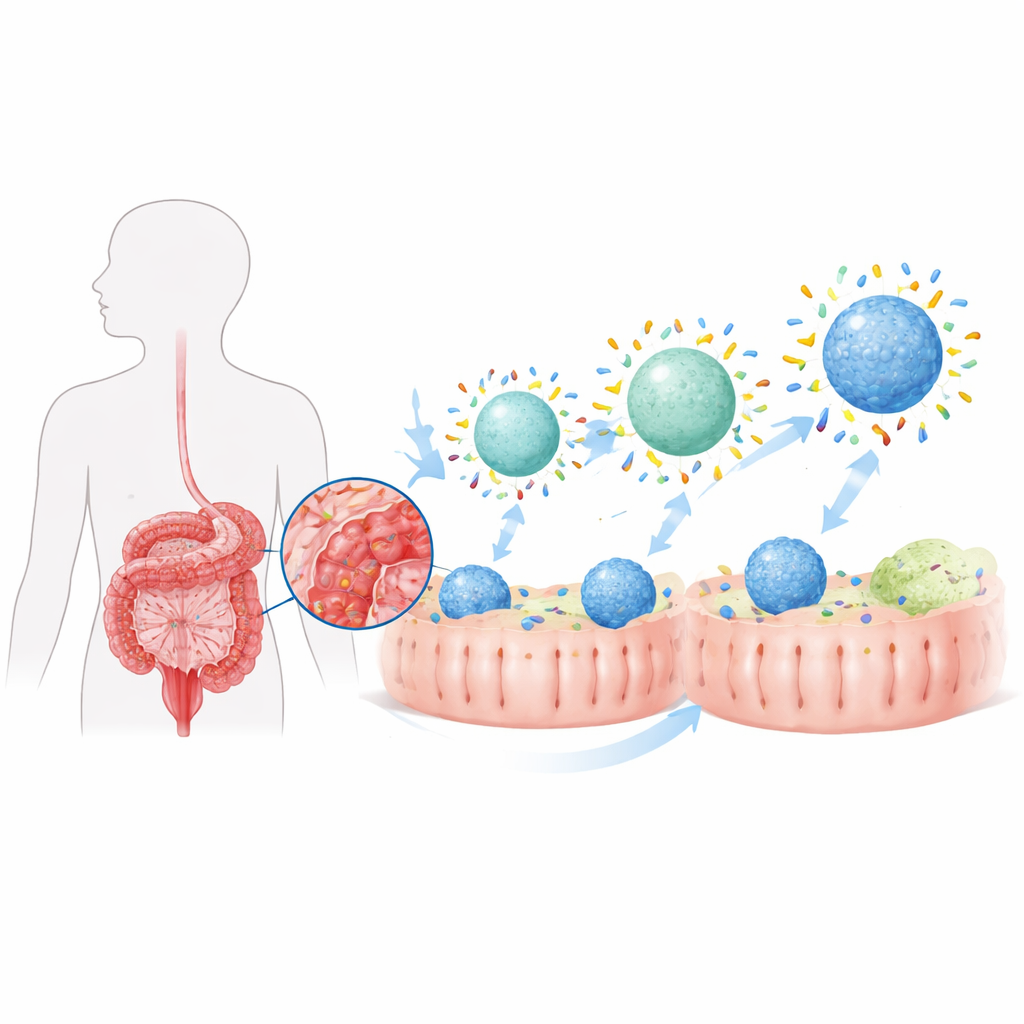
Making Particles More Attractive to Proteins
The team built lipid-based nanoparticles similar to tiny fat bubbles and tuned how water-loving or water-fearing their surfaces were by adding different amounts of a common polymer coating. Particles with more hydrophobic surfaces bound far more intestinal proteins when exposed to colitis fluids, both in animals and in test tubes. When these “phobic” particles were loaded with the steroid budesonide and given orally to rats with colitis, they delivered much more drug to colon macrophages than more water-loving versions. As a result, treated animals regained more body weight, showed less colon shortening and tissue damage, and had lower levels of inflammatory molecules in the gut. However, while inflammation improved, it did not fully return to a healthy state, suggesting that simply increasing the total amount of corona proteins was not enough.
Stiffening the Particles to Choose Better Partners
To push the concept further, the researchers kept the surfaces hydrophobic but altered the internal structure to make particles soft, intermediate, or stiff by filling their cores with plastic spheres of different sizes. All three still picked up similar total amounts of intestinal protein, but the mix of proteins changed. The stiffest particles formed coronas especially rich in proteins that can latch onto receptors on macrophages or carry small nutrients those cells take up. One such protein, S100A8, is abundant in inflamed intestines and can engage specific macrophage receptors. Blocking S100A8 on the corona sharply reduced uptake of the stiff particles by macrophages, revealing it as a key targeting component selected by the particle’s rigidity.
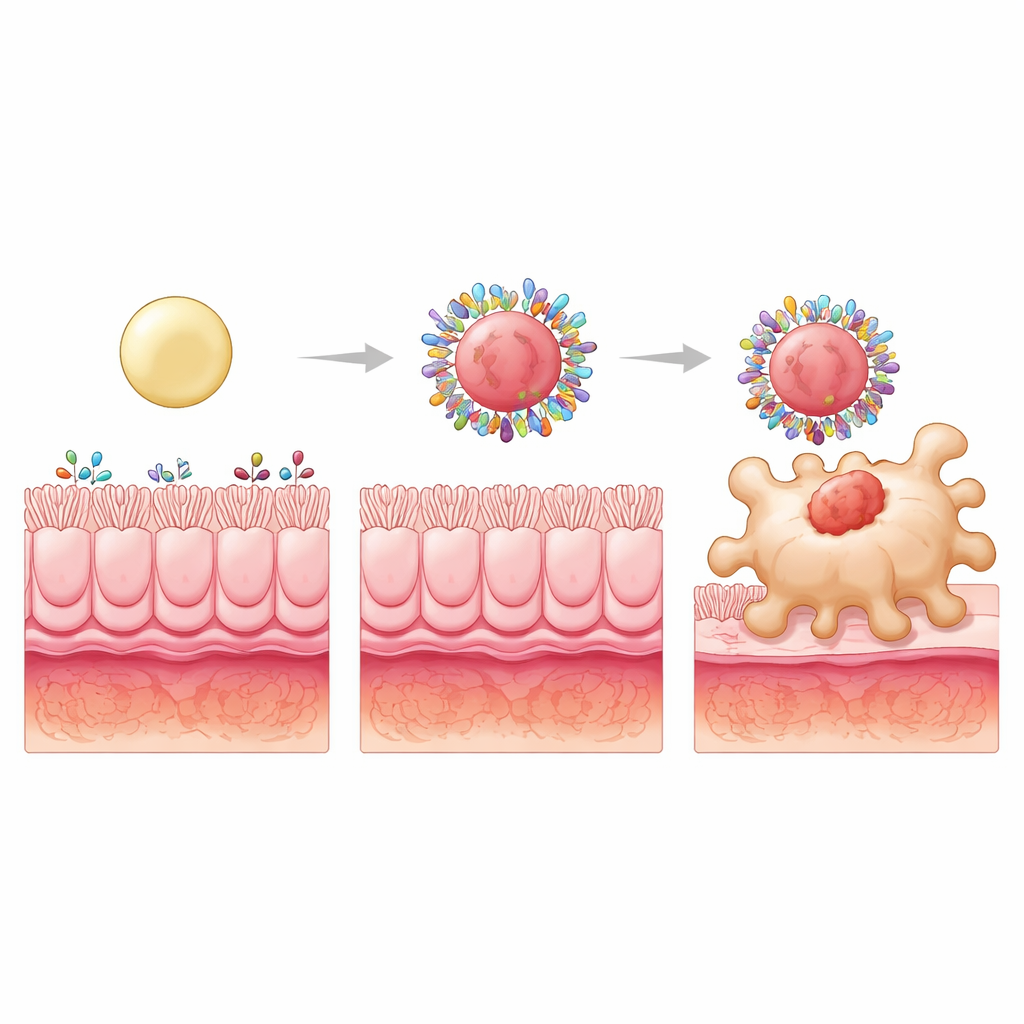
From Better Targeting to Better Healing
When budesonide was packed into these high-hydrophobic, high-rigidity particles and given orally to rats with colitis, the benefits were striking. The stiff formulation not only reduced gut damage and oxidative stress, but also normalized multiple inflammatory signals to levels seen in healthy animals. It restored a healthier balance between “attack” and “calm” macrophage types and boosted regulatory T cells, which help keep immune responses in check. Importantly, these gains depended on the disease-altered protein environment in colitis; the same design did not show special targeting in healthy rats, underscoring that the corona is a dynamic, disease-specific ally.
Why This Matters for Future Medicines
This work shows that we can guide the body’s own proteins to act as smart, self-renewing “labels” on oral drug carriers simply by tuning the carriers’ surface and mechanical properties. By first increasing protein capture through hydrophobic surfaces and then using rigidity to favor macrophage-homing proteins like S100A8, the researchers created nanoparticles that naturally seek out the immune cells driving colitis and deliver therapy where it is needed most. The same design logic—manipulating how particles feel and behave at the gut surface—could be adapted to many kinds of nanomedicines and diseases, offering a new, more subtle route to targeted treatment without bolting on artificial targeting molecules.
Citation: Wu, J., Ni, M., Xing, L. et al. Surface hydrophobicity and rigidity determines protein corona on orally delivered nanoparticles treating colitis. Nat Commun 17, 2497 (2026). https://doi.org/10.1038/s41467-026-70453-9
Keywords: nanoparticle drug delivery, inflammatory bowel disease, protein corona, oral nanomedicine, macrophage targeting