Clear Sky Science · en
Operando insights on stable Cu2+ active sites for efficient electrochemical CO2-to-C2H4 conversion
Turning a Climate Problem into a Useful Building Block
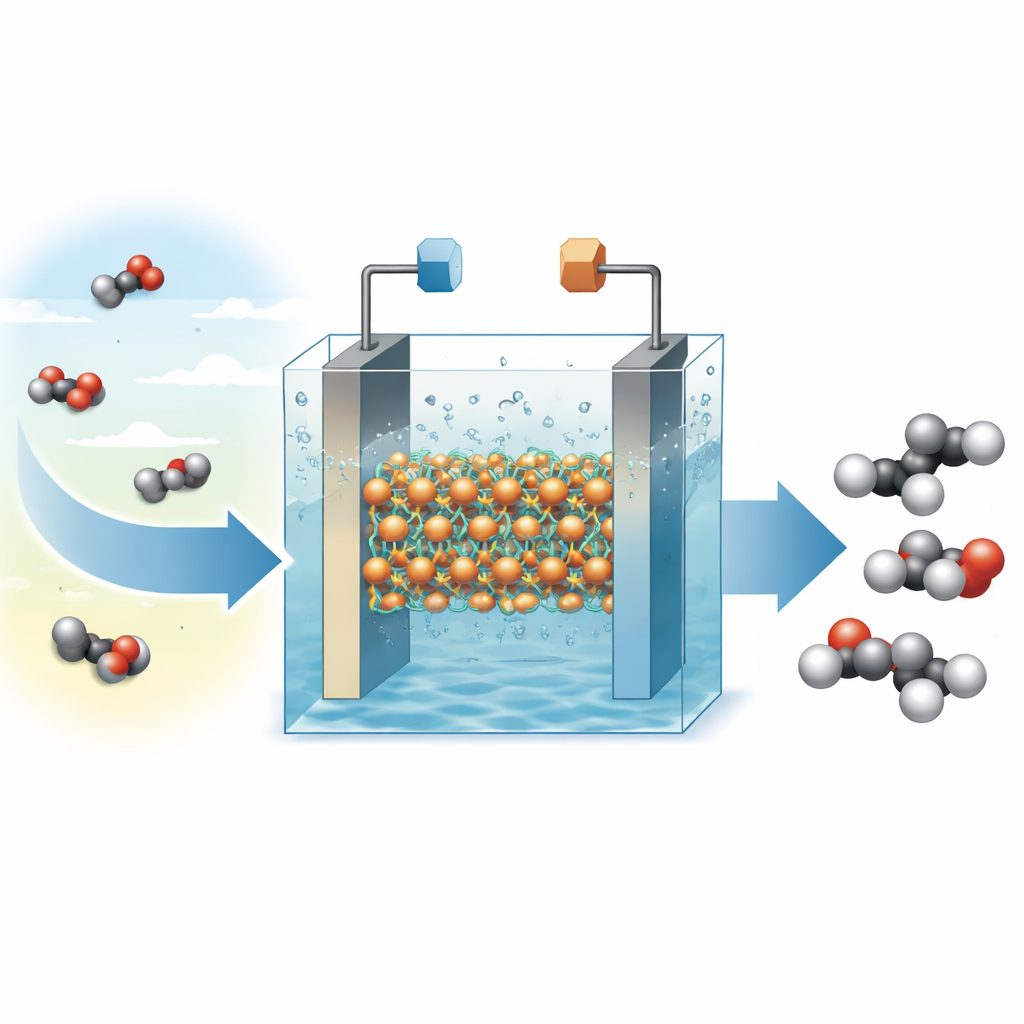
Carbon dioxide is the main greenhouse gas driving climate change, but it is also a cheap and abundant source of carbon. Chemists and engineers are racing to find ways to turn CO2 into everyday products using clean electricity instead of fossil fuels. This study reports a new copper-based material that converts CO2 into ethylene—a key ingredient for plastics and many chemicals—with high efficiency and long-term stability, bringing the idea of recycling CO2 into valuable goods a step closer to practical reality.
Why Ethylene Matters
Ethylene is one of the world’s most widely produced chemicals, used to make plastics, solvents, and countless consumer products. Today it is almost entirely made from oil and natural gas, releasing large amounts of CO2 in the process. If we could instead make ethylene directly from CO2 using renewable electricity, we could both cut emissions and create a closed carbon loop. Copper is one of the few elements that can drive CO2 toward multi‑carbon molecules like ethylene, but conventional copper surfaces tend to reshape and change their chemical state under operating conditions, which reduces their selectivity and shortens their lifetime.
Designing a Calm Home for Active Copper
The authors tackle this problem by building a metal–organic polymer—called CuBBTA—in which copper ions are locked into a repeating scaffold formed with an organic molecule named benzobistriazole. In this structure, copper atoms remain in a higher charged state (Cu2+) and sit at well-defined distances from one another, connected through nitrogen atoms and bridging hydroxyl groups. Detailed structural studies using X-ray diffraction, electron microscopy, and advanced spectroscopy confirm that copper atoms are isolated yet periodically arranged, forming a quasi two‑dimensional network with abundant, precisely spaced copper sites exposed to the reacting CO2.
Strong Performance in a Practical Device
When tested in a flowing liquid cell and in a membrane-based electrolyzer—setups closer to industrial devices—CuBBTA shows impressive performance. In alkaline solution, it converts CO2 to ethylene with a Faradaic efficiency of about 62%, meaning nearly two‑thirds of the electrical current is devoted to making ethylene rather than side products. The material also reaches a high power conversion efficiency for ethylene production and sustains currents near one ampere for over 50 hours while keeping ethylene selectivity above 55–60%. Post‑reaction imaging and spectroscopy reveal that the overall structure and distribution of copper sites remain essentially unchanged, unlike many copper catalysts that break down or clump into larger particles.
Watching Atoms Work in Real Time
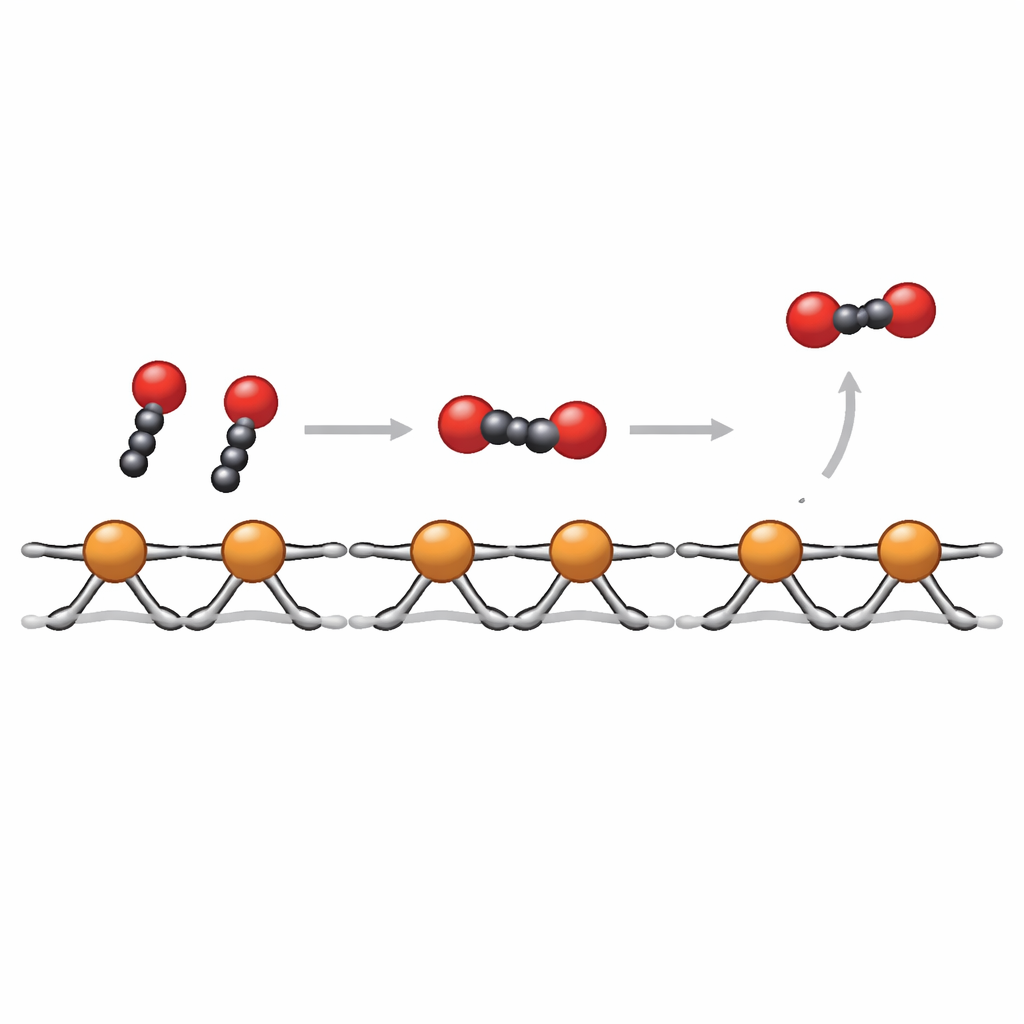
To understand why CuBBTA is so stable and selective, the team used several “operando” techniques that probe the material while it is actually converting CO2. X-ray absorption measurements show that the copper ions stay in the Cu2+ state across a wide range of applied voltages, with no sign of metallic copper clusters forming. Raman and infrared measurements confirm that the organic framework and copper–ligand bonds remain intact. Infrared spectroscopy of surface-bound molecules, together with online mass spectrometry, reveals that neighboring copper sites in the polymer favor the formation of a key paired intermediate, often written as *COCHO, created when two smaller fragments derived from CO2 couple on adjacent sites. Quantum‑mechanical calculations support this picture, indicating that the fixed spacing and strong coordination around Cu2+ lower the energy barrier for this C–C bond‑forming step compared with a conventional metallic copper surface.
How This Moves CO2 Recycling Forward
In everyday terms, CuBBTA acts like a well-organized assembly line: CO2 molecules arrive, are partially reduced on individual copper stations, and then two fragments meet at neighboring stations to form the two‑carbon backbone of ethylene. Because the copper ions are held firmly in place and shielded from overly harsh local conditions, the line keeps running smoothly without the machinery falling apart. The study shows that carefully designed copper–organic frameworks can both stabilize the most effective form of copper and arrange active sites at just the right distances to promote carbon–carbon coupling. This strategy offers a pathway to more durable, efficient devices that turn waste CO2 into valuable chemicals using renewable electricity.
Citation: Zhang, Z., Xu, Q., Han, J. et al. Operando insights on stable Cu2+ active sites for efficient electrochemical CO2-to-C2H4 conversion. Nat Commun 17, 2654 (2026). https://doi.org/10.1038/s41467-026-70442-y
Keywords: electrochemical CO2 reduction, copper catalysts, ethylene production, metal–organic polymers, carbon utilization