Clear Sky Science · en
Carbonylolysis of waste polyesters into high-value organic acids
Turning Plastic Trash into Useful Ingredients
Plastic bottles, food trays, and synthetic fabrics are everywhere—and so is the waste they leave behind. Much of this plastic, especially the common polyester PET used in drink bottles and clothing, ends up burned or buried, wasting valuable material and adding to climate pollution. This study introduces a new way to break down these stubborn plastics and rebuild their carbon into more useful, higher-value ingredients, potentially changing how we think about plastic waste and chemical production.
Why Today’s Plastic Recycling Falls Short
Most PET recycling today is mechanical: old bottles are cleaned, melted, and reshaped. Each cycle, however, downgrades quality and demands very clean waste streams. Chemical methods can break PET back into its building blocks, but they often need high temperatures, strong bases, and large amounts of acid, generating salty wastewater and using a lot of energy. A key problem is what to do with ethylene glycol, a small alcohol released when PET is taken apart. Current approaches usually turn it into low-value, short-chain molecules and still rely on harsh conditions, making it hard to build a truly sustainable, circular system.
A One-Pot Route from Waste to High-Value Acids
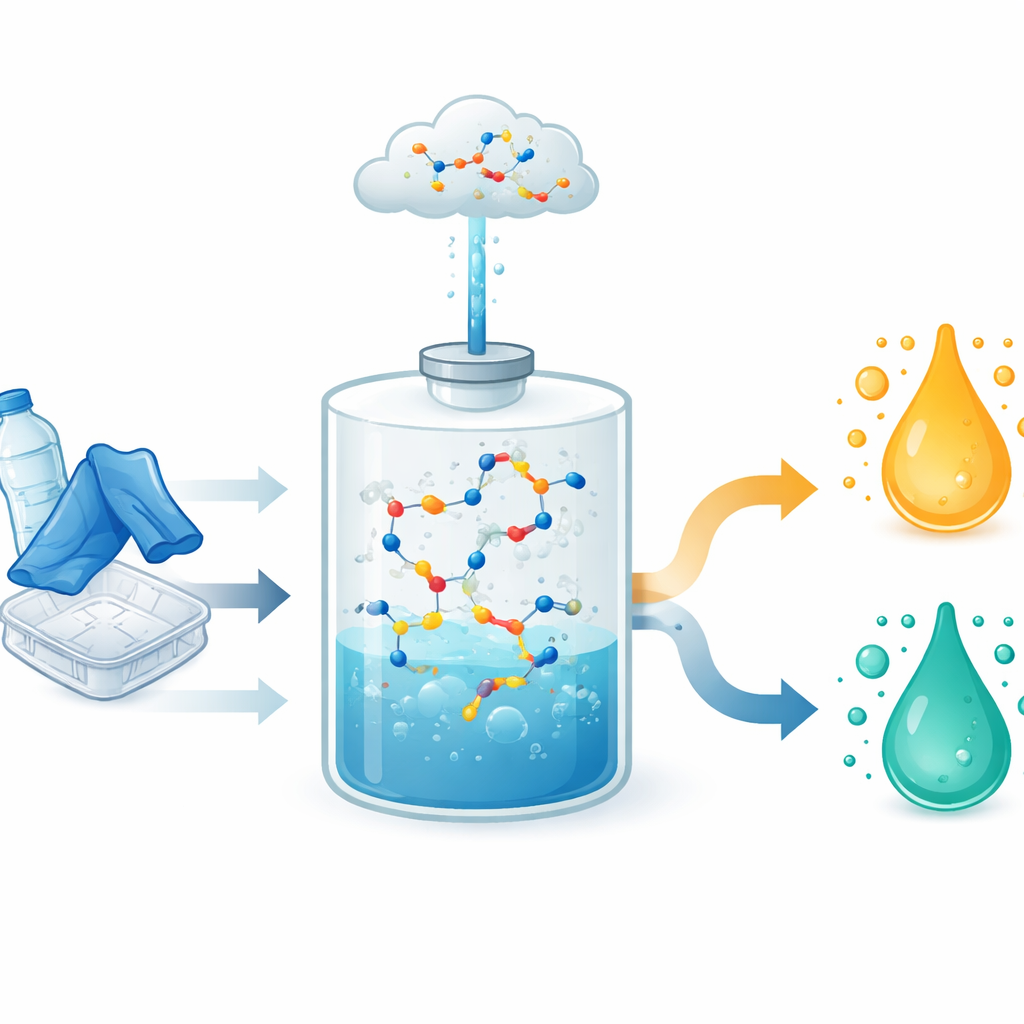
The authors present a single-step process they call “carbonylolysis” that breaks down polyesters while simultaneously rebuilding their carbon skeletons into more valuable organic acids. PET waste, together with a small amount of water, is fed into a special solvent along with a rhodium–iodide catalyst and carbon monoxide gas. Under relatively mild conditions (170 °C and moderate pressure), the plastic chains dissolve and split, releasing terephthalic acid—the main building block of PET—and ethylene glycol. Instead of letting ethylene glycol accumulate or requiring a separate step, the same mixture immediately converts it into a higher-value three-carbon acid called propionic acid.
How the Invisible Chemistry Works
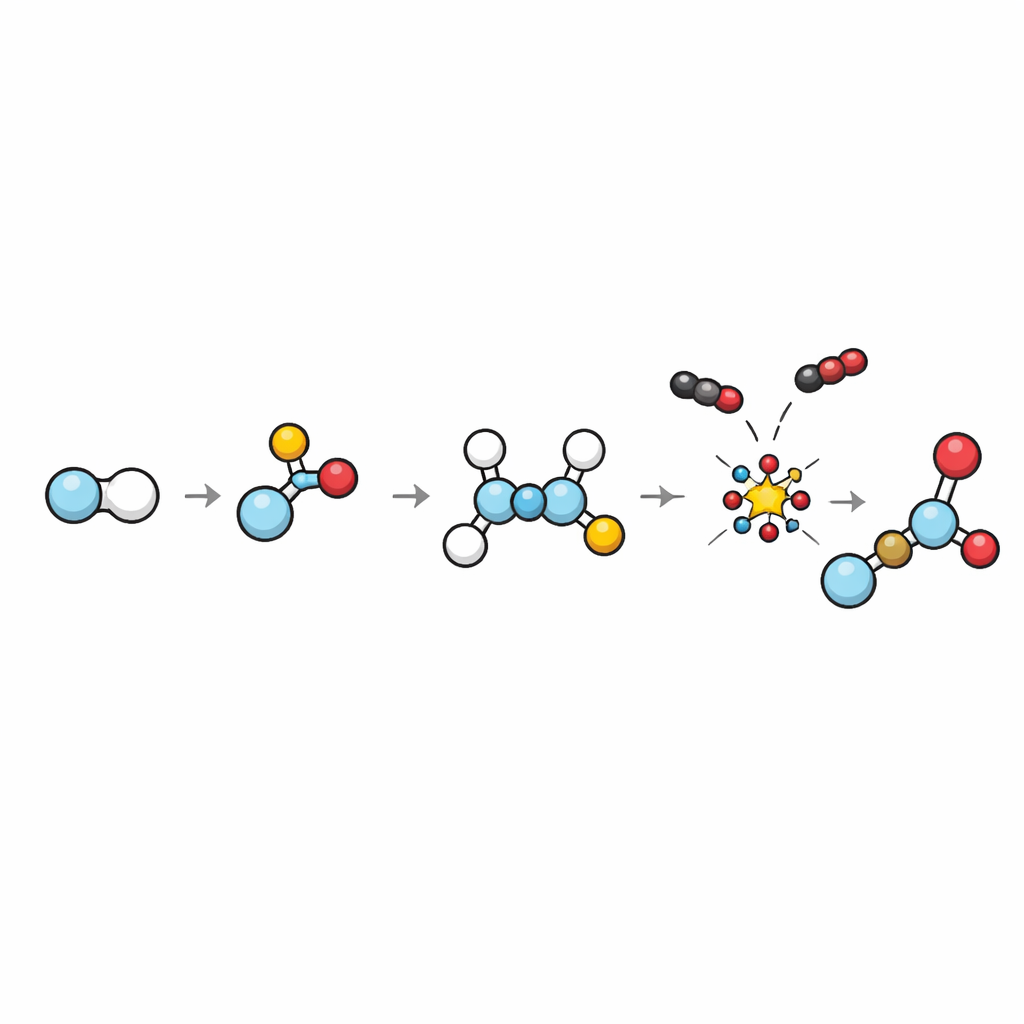
By tracking reaction rates, intermediates, and using quantum-chemical calculations, the team maps out a step-by-step picture of the hidden chemistry. First, PET is hydrolyzed: water helps snip the long chains into terephthalic acid and ethylene glycol, with the fluorinated solvent helping to dissolve the rigid polymer. Then iodide ions turn ethylene glycol into a more reactive form that sheds leaving groups to form ethylene gas. This gas then reacts with carbon monoxide on the rhodium catalyst, adding a new carbon and oxygen unit to form propionic acid. Calculations show that this “break to ethylene, then build up” route is energetically easier than alternative pathways that would give other acids, explaining why propionic acid is formed so selectively.
From Lab Plastics to Real-World Waste
The method works not only on pure PET powder but also on real waste: bottles, food trays, non-woven fabrics, ropes, and textiles that mix PET with cotton, rayon, or spandex. In most cases, both terephthalic acid and propionic acid are formed in yields around 90–99 percent, even without energy-intensive grinding. Beyond PET, the same strategy upgrades a range of other polyesters, including bio-based and longer-chain materials, into corresponding valuable acids and monomers. This shows that carbonylolysis is robust to additives and mixed materials that typically complicate recycling.
Energy, Climate, and Economic Payoff
Using detailed process simulations, life-cycle assessment, and cost modeling, the authors compare their route to traditional options such as landfilling, incineration, and standard chemical recycling. Because the key reaction releases heat, the process partly powers itself, cutting energy demand. By converting both major fragments of PET into marketable products and avoiding heavy acid–base use and brine wastewater, the new route reduces non-renewable energy use and greenhouse gas emissions to a fraction of conventional hydrolysis. An industrial-scale design treating 100,000 tons of PET chips per year is projected to be profitable, with product sales of terephthalic and propionic acids more than offsetting the cost of waste feedstock, carbon monoxide, and plant operation.
A New Vision for Circular Plastics
In simple terms, this work shows that plastic waste can be more than a nuisance—it can be a rich carbon source for valuable chemicals. By combining breakdown and rebuilding steps in one pot, the carbonylolysis strategy turns discarded polyester into two high-value organic acids under milder and cleaner conditions than many current methods. If scaled with more abundant catalysts and adapted for highly mixed waste streams, this approach could help close the loop on plastics, lowering our reliance on fossil raw materials while reducing pollution and climate impact.
Citation: Liu, D., Zhu, S. & Mei, Q. Carbonylolysis of waste polyesters into high-value organic acids. Nat Commun 17, 2279 (2026). https://doi.org/10.1038/s41467-026-70412-4
Keywords: plastic recycling, polyester upcycling, carbonylation, organic acids, circular economy