Clear Sky Science · en
Dynamically assembled photochromic cages operational in water with visible light
Shaping Tiny Light-Responsive Containers
Imagine if medicines, sensors, or catalysts could be turned on and off inside the body simply by shining safe, colored light. This study describes tiny, hollow molecules—"cages"—that assemble and rearrange themselves when illuminated, even in water and using red light that can pass through human tissue. These smart containers behave a bit like simple machines: they change shape, move between oil-like and watery environments, and interact with metal ions or other partners, all under external control.
Why Light-Controlled Cages Matter
Living cells rely on countless molecular structures that adapt rapidly to changing conditions. Chemists have long sought artificial nanostructures that can do something similar: respond to signals like pH, temperature, or chemicals, and alter what they bind or how they behave. Light is a particularly attractive signal because it can be applied with pinpoint accuracy in space and time and leaves no residue. However, most light-responsive molecular cages only work in organic solvents and often require harsh ultraviolet light, which is poorly suited to biology. This work tackles both problems by designing cages that respond to visible and red light and can operate in water, hinting at future biomedical and technological uses.
Building Cages That Reconfigure Under Light
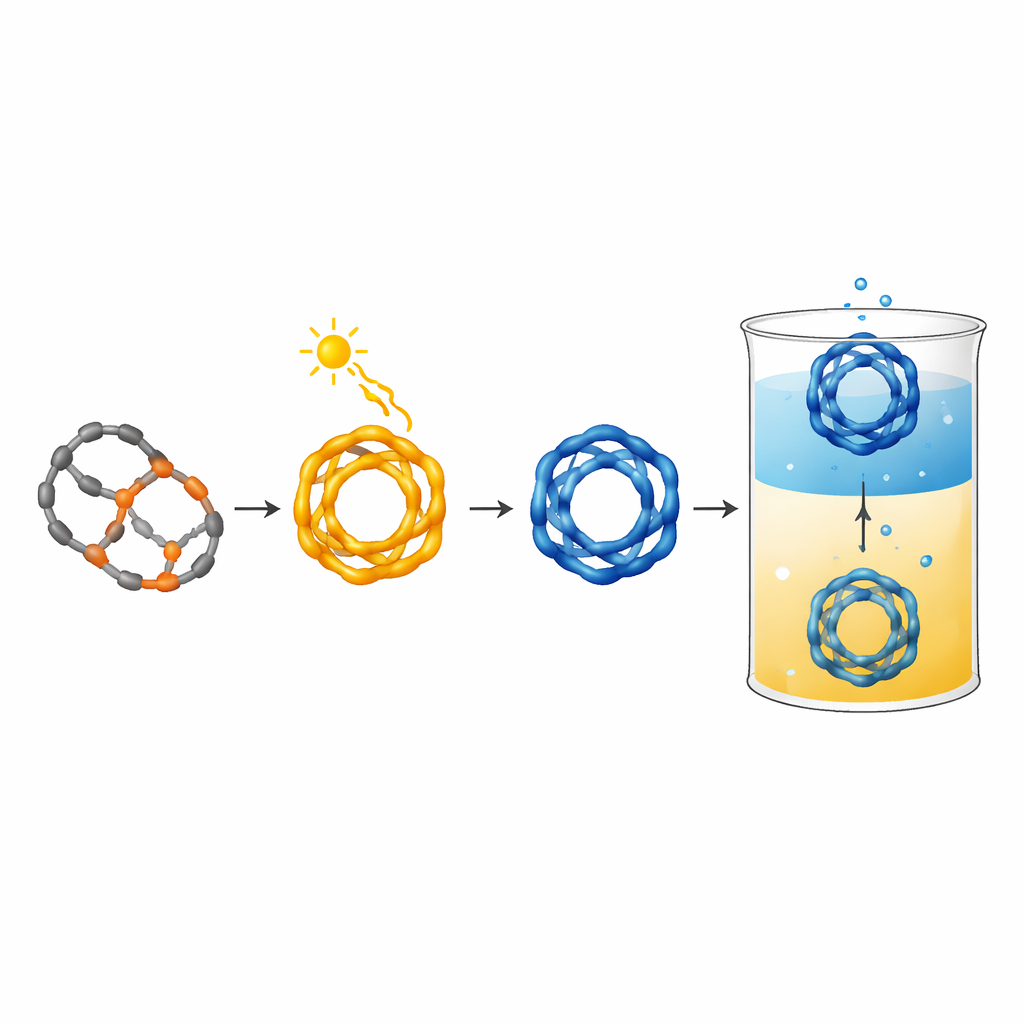
The researchers start from a special "photoswitch" based on azobenzene, a molecule that can flip between two shapes when lit with different colors. They attach aldehyde groups to create building blocks that can link with a three-armed amine through reversible chemical bonds, allowing the pieces to self-assemble into well-defined hollow cages. In their first system, three fluorinated azobenzene pillars and two amine hubs spontaneously form a dynamic cage in solution. Red light (around 660 nm) flips all three pillars into a bent form, subtly straining the cage, while violet or green light returns them toward the original, more relaxed shape. Because the cage framework holds the switches in a particular arrangement, it biases how efficiently and completely the light-driven changes occur, leading to pronounced, predictable photoresponse.
Locking the Shape and Making It Work in Water
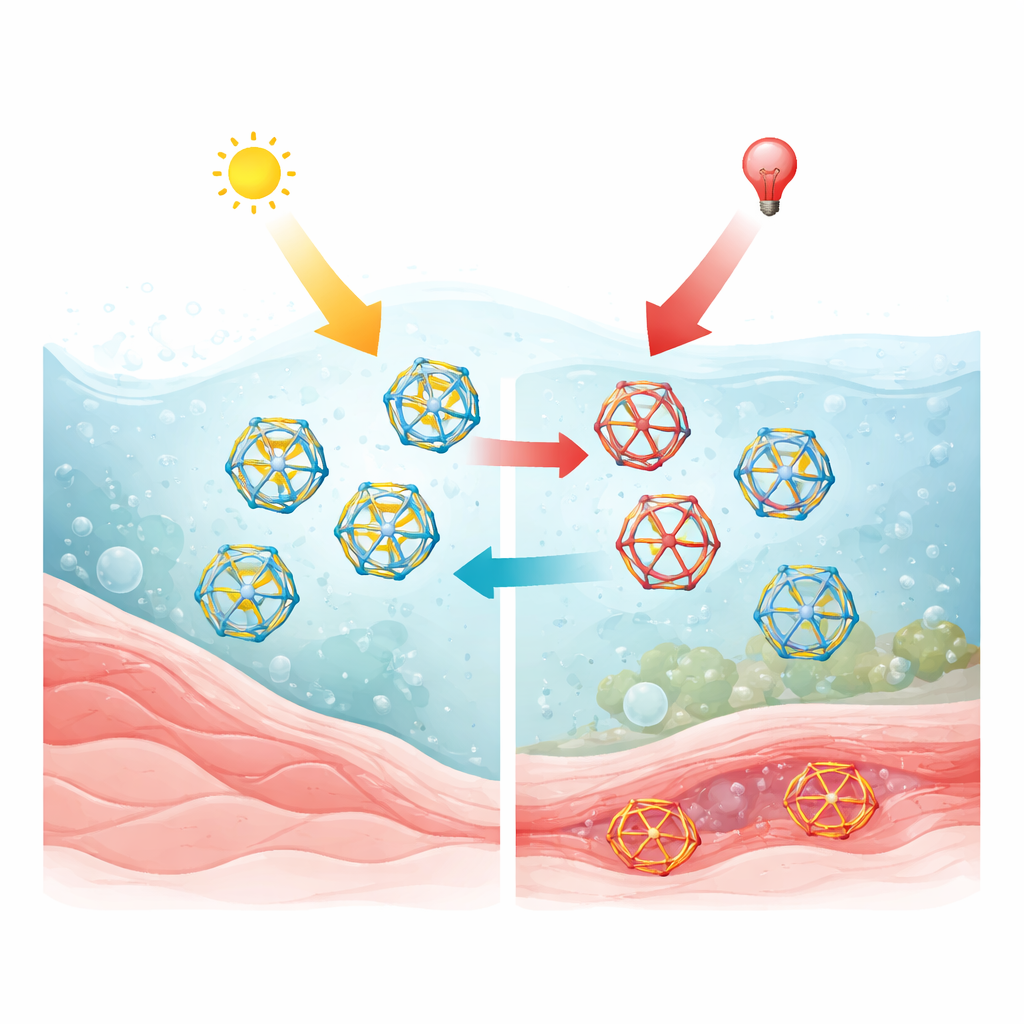
To move from a fragile, ever-rearranging network to a robust device, the authors chemically “freeze” the dynamic bonds, turning them into permanent connections and yielding a stable covalent cage. This locked cage can still switch back and forth between light-controlled shapes, but now without falling apart. A key trick is protonation: when the cage is positively charged by acid, it becomes soluble in water and can be shuttled reversibly between an organic layer and an aqueous layer by bubbling carbon dioxide in and then letting it escape. In water, the cage remains photoswitchable with visible light and can even form inclusion complexes with host molecules such as cucurbiturils, indicating that it can carry or interact with guests inside its hollow interior. Toxicity tests in human cell culture show that at low submicromolar concentrations, the protonated cage is compatible with cells, suggesting it can be used in biological experiments at carefully chosen doses.
Reaching Into the Near-Infrared and Talking to Metals
To extend control deeper into the biologically useful “therapeutic window” of light, the team designs a second azobenzene building block bearing chlorine atoms. This variant can be switched in both directions using only red and near-infrared light, without resorting to higher-energy colors. It also forms its own family of cages, though the bulkier chlorine atoms make these more crowded and prone to partial opening or rearrangement. By mixing fluorinated and chlorinated building blocks, the scientists create hybrid cages that rearrange composition in response to light and heat. They further show that similar cage frameworks built from a non-switching bipyridine unit act as multivalent ligands for metal ions such as iron, forming colorful complexes that move cleanly into water. When these metal-binding units are exchanged into a photoresponsive cage, metal ions become an additional handle to direct where the assemblies reside and how they behave.
From Tiny Cages to Life-Like Machines
Together, these experiments outline design rules for constructing molecular cages that assemble on their own, respond predictably to visible and red light, and function in water, including environments that mimic biological conditions. By combining reversible self-assembly with permanent "locking" steps, and by integrating multiple triggers such as light color, acidity, carbon dioxide, and metal ions, the authors move closer to molecular machines that can adapt in life-like ways. In the long term, such cages could serve as controllable carriers for drugs, tunable nanoreactors for chemical reactions, or responsive sensors inside living systems, all steered from the outside by carefully chosen colors of light.
Citation: Schäfer, V., Seliwjorstow, A., Fuhr, O. et al. Dynamically assembled photochromic cages operational in water with visible light. Nat Commun 17, 2488 (2026). https://doi.org/10.1038/s41467-026-70406-2
Keywords: photochromic molecular cages, light responsive nanotechnology, visible and red light switching, self-assembly in water, azobenzene photoswitches