Clear Sky Science · en
3D pentaculture model unveils malignant cell-driven macrophage polarization in high-grade serous ovarian cancer
Why this research matters
Ovarian cancer is often discovered late and can be stubbornly resistant to treatment. One reason is that cancer cells live within a supportive neighborhood of immune and connective-tissue cells that help them evade drugs and the body’s defenses. This study builds a sophisticated laboratory model—using five types of human cells in 3D—to mimic that neighborhood and watch, in real time, how cancer cells push nearby immune cells called macrophages into helping the tumor instead of fighting it.
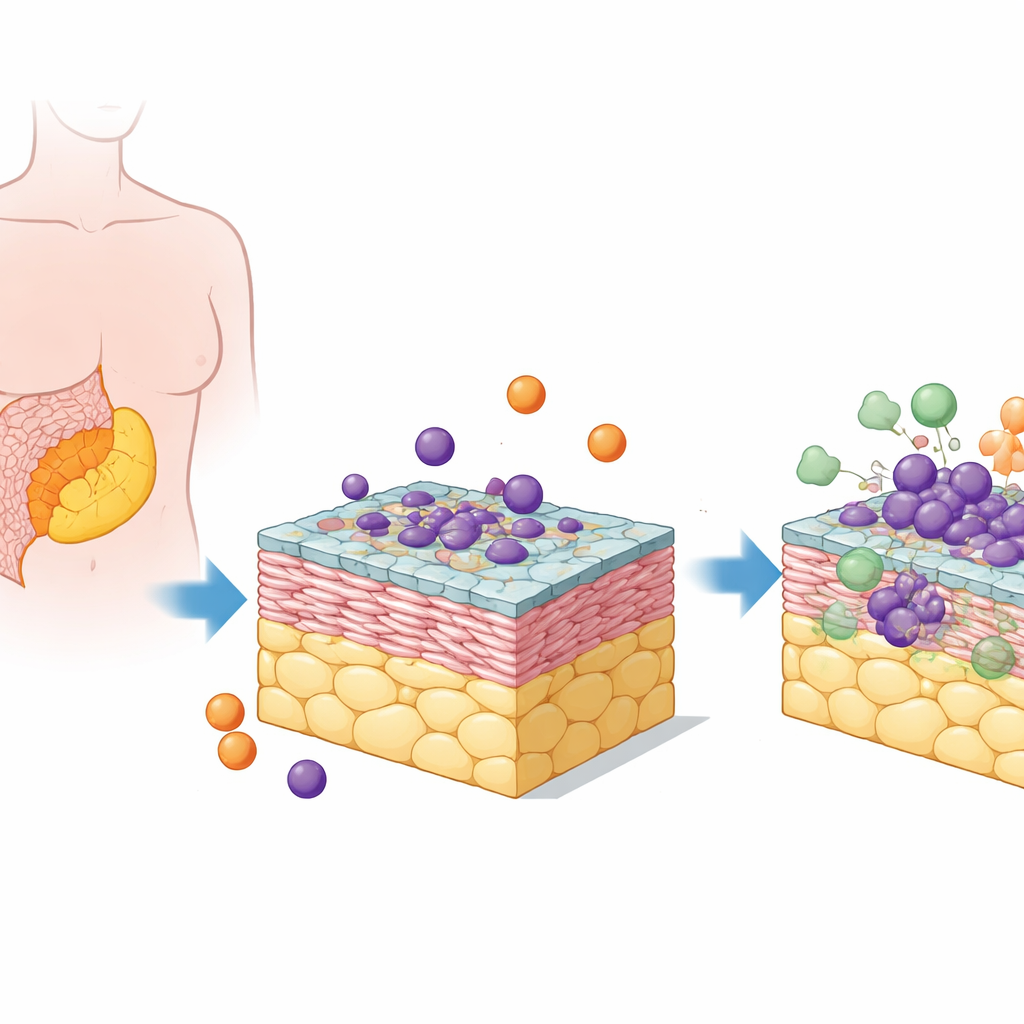
Building a tiny tumor neighborhood
The researchers focused on high-grade serous ovarian cancer, the most common and deadly form of the disease. Its metastases often grow in a fatty tissue in the abdomen called the omentum. To recreate this setting, the team combined human fat cells, connective-tissue cells (fibroblasts), a lining of mesothelial cells, ovarian cancer cells, and circulating immune cells called monocytes. These five cell types were assembled into stacked 3D “pentacultures” that more closely resemble real tumor deposits than flat cell layers in a dish.
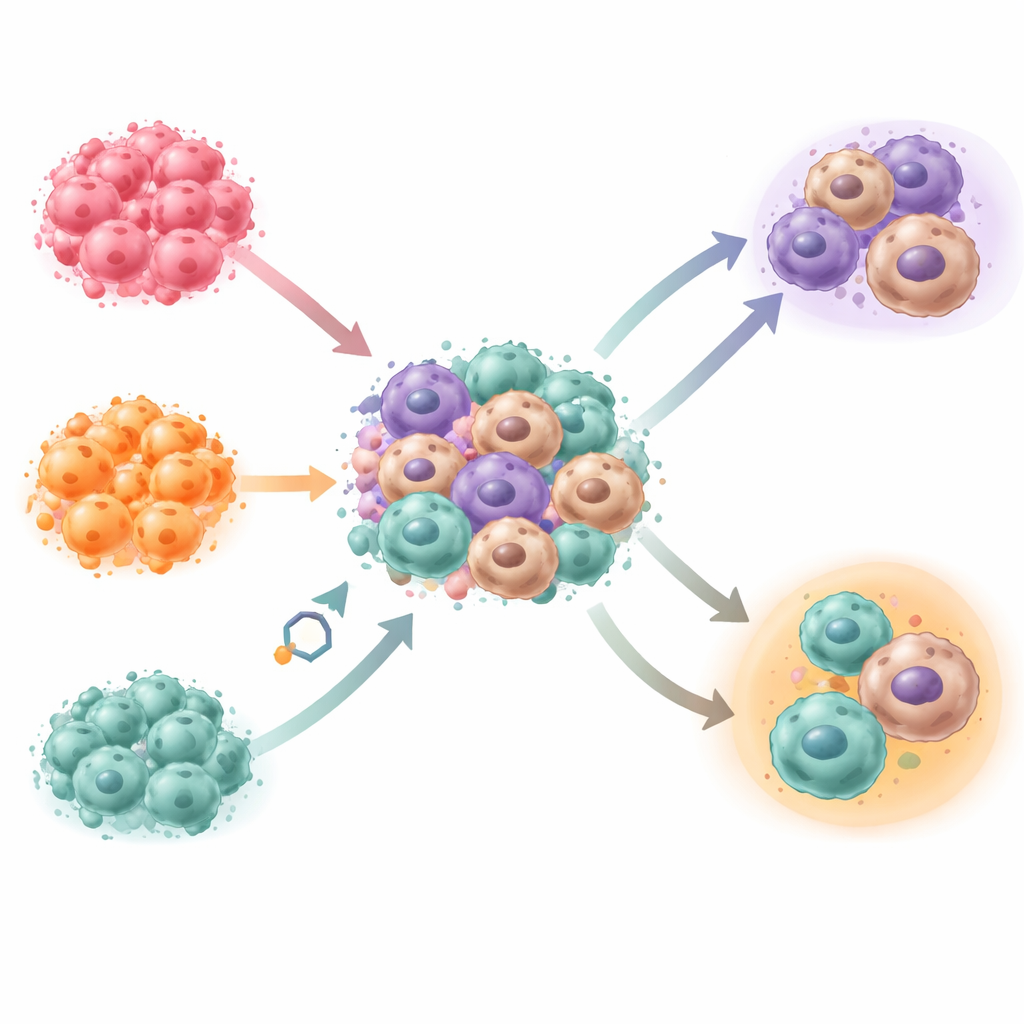
Turning immune defenders into bystanders
Within this mini–tumor environment, monocytes naturally matured into macrophages—without the artificial growth signals usually added in the lab. Using gene-activity profiling and comparisons to single-cell data from patient tumor samples, the team showed that the macrophages in the model closely match those found in real ovarian cancer metastases. Different cancer cell lines produced distinct mixtures of macrophage types and different overall architectures of the mini-tumors, indicating that genetic and molecular differences within cancer cells strongly shape the surrounding immune landscape.
How cancer cells dodge being eaten
Macrophages can swallow and destroy cancer cells, but tumors often send out “don’t-eat-me” signals on their surfaces. The team examined two such signals, CD47 and CD24, on three ovarian cancer cell lines grown in the 3D model. Cancer cells with higher levels of these signals were less likely to be engulfed by macrophages. In the pentacultures, one cell line was readily eaten, another largely escaped, and a third showed mixed behavior. Live imaging revealed that macrophages moved differently and positioned themselves closer or farther from cancer clusters depending on the cancer line, mirroring patterns seen in patient tissues.
Testing ways to reawaken immune attack
Because these surface signals can be blocked with antibody drugs, the researchers treated the models with experimental antibodies that target CD47 or CD24. In pentacultures built with one particular cancer line, blocking either signal increased macrophage contact with cancer cells, boosted engulfment, and reduced cancer cell numbers, while leaving other cells in the model largely unharmed. In another cancer line with strong resistance, blocking these signals had little effect, but inhibiting a separate communication route (the TGFβ pathway) reduced cancer cells in a way that still depended on the presence of macrophages—suggesting alternative, non‑engulfment killing mechanisms such as toxic cytokine release.
What this means for patients
To a lay reader, the key message is that not all ovarian cancers manipulate immune cells in the same way, even when they look similar under the microscope. By building a realistic, five-cell 3D model from human tissues, this work shows how tumor cells can tune macrophages to either ignore them or attack them, and how blocking specific “don’t‑eat‑me” signals can sometimes flip that switch. The model closely mirrors patient tumors, making it a promising medium-throughput test bed to sort out which immune‑targeting drugs are likely to work for which tumor profiles. In the long run, such tailored lab systems could help match individual patients to therapies that best reawaken their own immune cells against ovarian cancer.
Citation: Malacrida, B., Elorbany, S., Laforêts, F. et al. 3D pentaculture model unveils malignant cell-driven macrophage polarization in high-grade serous ovarian cancer. Nat Commun 17, 2451 (2026). https://doi.org/10.1038/s41467-026-70398-z
Keywords: ovarian cancer, tumor microenvironment, macrophages, immunotherapy, 3D cell culture