Clear Sky Science · en
Solubility based mechanistic profiling of combinatorial drug therapy
Why pairing medicines can matter
Modern cancer care often relies on drug combinations, but figuring out which medicines work best together is still largely trial and error. This study focuses on acute myeloid leukemia, an aggressive blood cancer that frequently comes back after treatment. The researchers present a new way to watch, in bulk, how cells’ proteins respond when two drugs are given together. Their approach helps explain why certain combinations are both more effective and less toxic, and offers a roadmap for designing smarter, more precise combination therapies for difficult cancers.
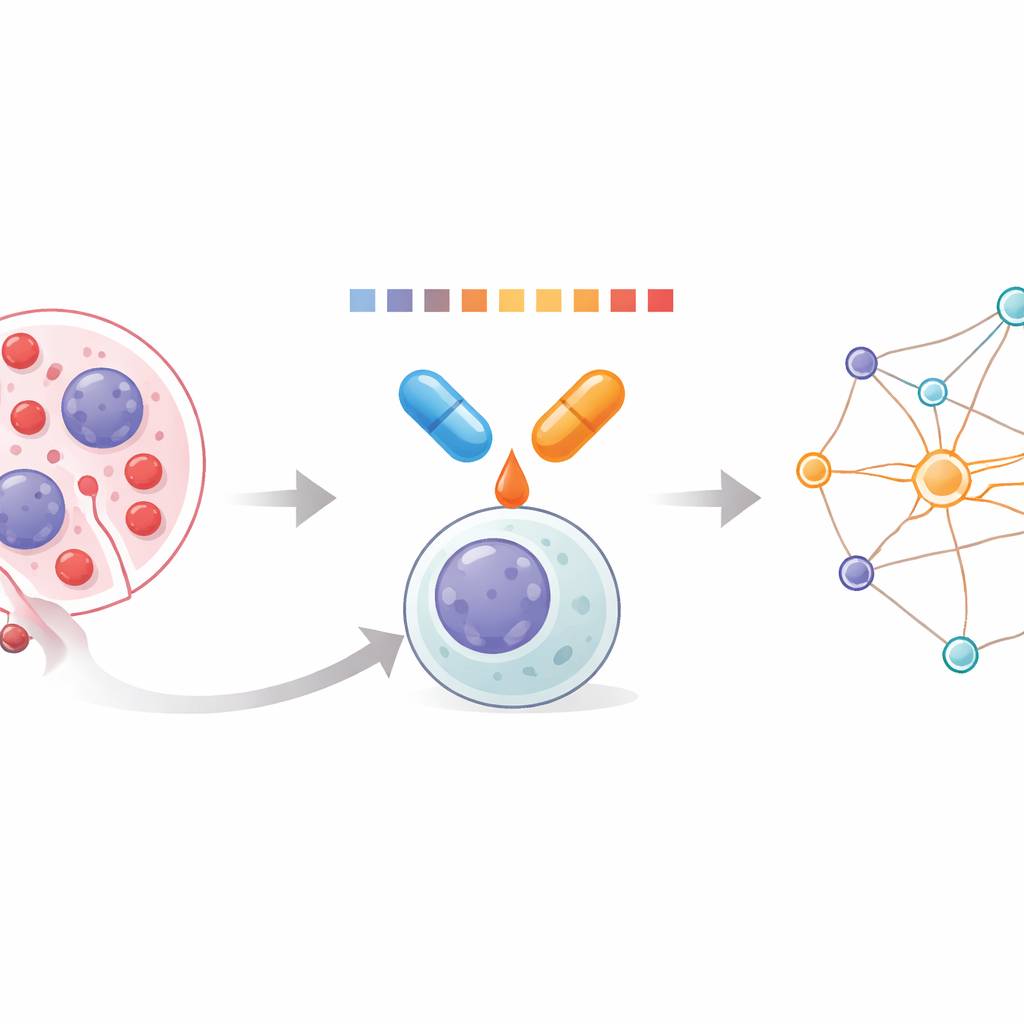
Looking inside leukemia cells
Acute myeloid leukemia (AML) arises when immature white blood cells in the bone marrow grow out of control and crowd out healthy blood formation. Because AML is driven by many different genetic changes, single drugs rarely work for long. Combinations can do better, but doctors have had limited tools to see how pairs of drugs act together at the level of thousands of proteins inside the cell. The team behind this work set out to measure those combined effects directly, using a method that reads how easily proteins dissolve or clump when heated. Changes in solubility reveal which proteins are being stabilized or destabilized by treatment, offering a window into the drugs’ true impact.
A new way to profile drug pairs
The researchers developed a workflow they call Combinatorial Proteome Integral Solubility/Stability Alteration analysis, or CoPISA. Cells, or their protein extracts, are exposed to drug A, drug B, the combination of A and B, or no drug. Each sample is then briefly heated across a series of temperatures, and the remaining dissolved proteins are captured and quantified by mass spectrometry. Instead of fitting complex curves protein by protein, the method uses the total area under each protein’s melting profile as a compact measure of its behavior. Comparing these areas across treatments shows which proteins become more or less soluble under each condition, revealing patterns unique to single drugs versus combinations.
Finding targets that only appear with both drugs
CoPISA was applied to two carefully chosen AML drug pairs: LY3009120 with sapanisertib (called LS), and ruxolitinib with ulixertinib (RU). These pairs had already shown strong activity and relatively low toxicity in patient samples, cell lines, and zebrafish models. CoPISA uncovered not only proteins affected by each drug alone, but also a distinct set of proteins whose solubility shifted only when both drugs were present. The authors describe this as “conjunctional targeting,” similar to an AND gate in simple logic: the protein responds only if both inputs (drugs) are on. For LS, these combination-only effects converged on processes such as DNA packaging, small protein tags called SUMO that control genome stability, and how leukemia cells stick to their surrounding tissue. For RU, unique targets pointed to weakened DNA-damage checkpoints, impaired energy production in mitochondria, and disturbed RNA processing.
Mapping the cancer’s weak spots
By overlaying their solubility data onto large maps of AML-related genes and pathways, the researchers could see how each treatment reshaped the cancer’s inner wiring. Many well-known AML genes—such as DNMT3A, NPM1, and TP53—were affected in ways that appeared only under combination therapy, reinforcing the idea that paired drugs can expose vulnerabilities invisible to single agents. The team also examined chemical decorations on proteins, such as acetylation, methylation and phosphorylation, which act as molecular switches. They found that certain modified forms of key proteins, including NPM1 and the DNA-repair factor BLM, were hit specifically by the combinations, hinting that altered protein localization and signaling contribute to the heightened effect.
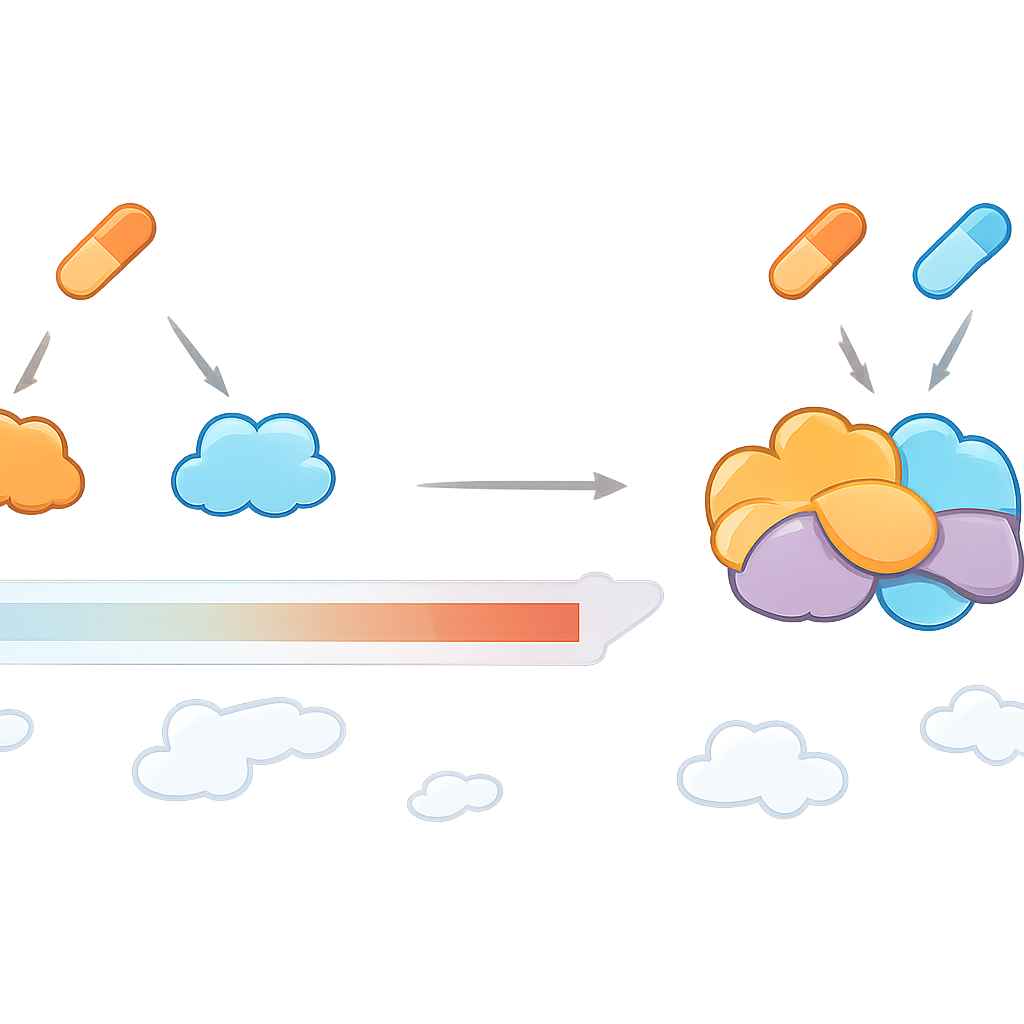
What this means for future treatments
Overall, the study shows that drug combinations can create their own unique landscape of protein targets, rather than simply adding together the effects of each drug. CoPISA provides a practical way to chart that landscape, highlighting proteins and pathways that only become vulnerable when two medicines act in concert. For patients, this could translate into combination therapies that are chosen not just because they shrink tumors in a dish, but because they engage the cancer’s deeper weak points while limiting unnecessary toxicity. Although demonstrated here in AML, the approach is broadly applicable and may help guide the rational design of combination treatments across many complex diseases.
Citation: Gholizadeh, E., Zangene, E., Vadadokhau, U. et al. Solubility based mechanistic profiling of combinatorial drug therapy. Nat Commun 17, 2744 (2026). https://doi.org/10.1038/s41467-026-70394-3
Keywords: acute myeloid leukemia, drug combinations, proteomics, protein solubility, targeted therapy