Clear Sky Science · en
Ribosomal modifications are associated with mesenchymal fate selection in the neural crest lineage
How tiny cell factories shape faces and tumors
Our faces and parts of our nervous system are built from a roaming group of embryonic cells called the neural crest. These cells can turn into bone, cartilage, nerves, and more. This study shows that their choices are influenced not just by genes, but by subtle chemical tweaks to the cell’s protein-making machines—ribosomes. The same molecular features that help build a normal face can, when misused, mark more dangerous forms of childhood cancer.
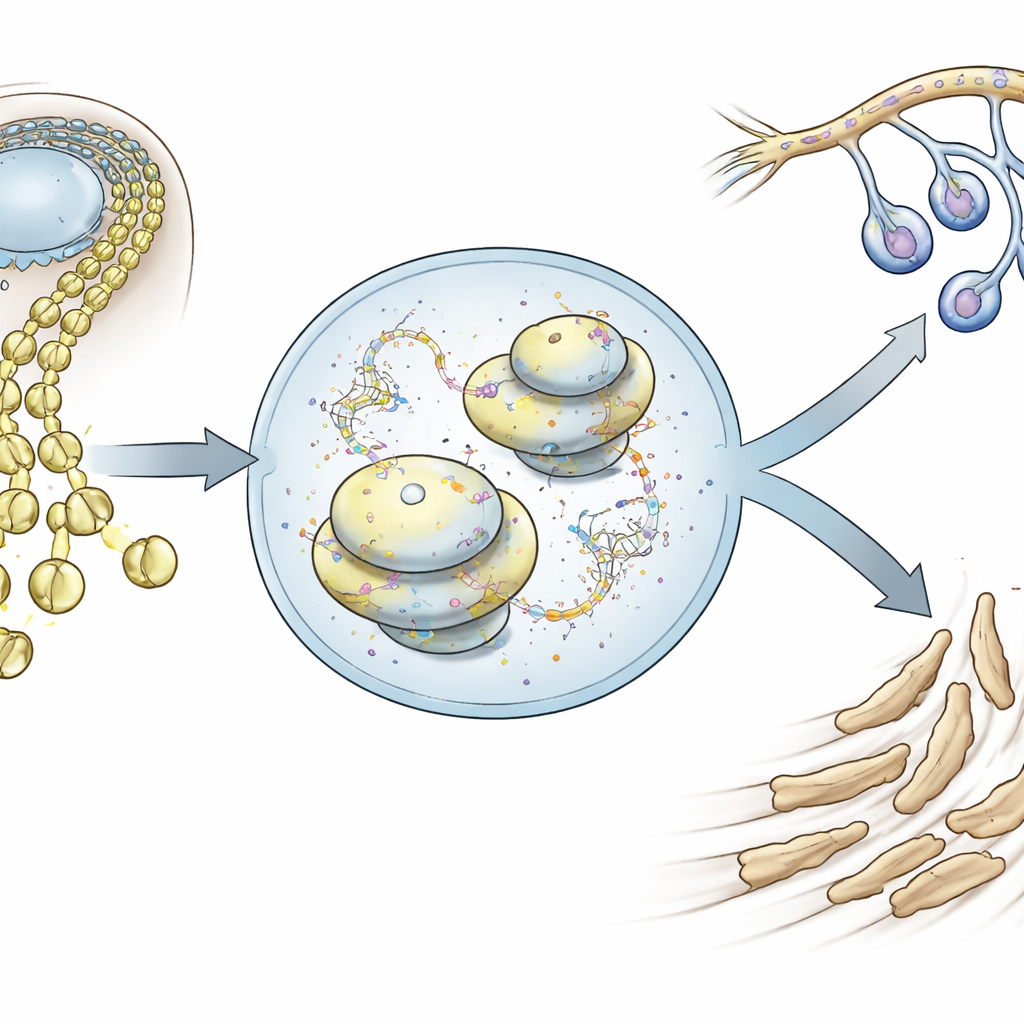
Shape-shifting cells that build the face
Neural crest cells arise along the early brain and spinal cord and then migrate outwards, eventually forming much of the skull, jaws, and peripheral nerves. Using single-cell RNA sequencing in mouse embryos, the authors tracked thousands of individual cranial neural crest cells as they left the neural tube and moved into the face. They found that very early on, these cells already show a subtle tilt toward one of two main futures: a mesenchymal path that will form facial bones and cartilage, or a neuro-glial path that will form neurons and glial support cells. These two gene activity programs are initially mixed in the same cells, but become mutually exclusive as development proceeds, forcing a fate decision.
Protein factories as decision makers
When the team examined which genes were linked to the early bias toward facial mesenchyme, they uncovered two major groups. One contained familiar players that drive cells to loosen, move, and change identity, a process known as an epithelial-to-mesenchymal transition. The other, more surprising group centered on ribosome assembly and chemical modification of ribosomal RNA, the core of the protein-making machinery. Rather than simply making more ribosomes, cranial neural crest cells switched on factors that fine-tune how ribosomes are built and chemically marked. Disrupting ribosomal RNA production in mouse embryos at the moment of fate choice largely spared developing nerves but severely stunted facial skeletal structures, showing that the mesenchymal route is especially dependent on this ribosomal control.
A special mark on ribosomal RNA
Diving deeper, the researchers focused on a single nucleotide within the 18S ribosomal RNA, at a location crucial for accurate protein synthesis. In cranial neural crest cells biased toward mesenchymal fates, sequencing repeatedly mis-read this position, a hallmark of heavy chemical modification rather than true mutation. Prior work had shown that a trio of enzymes adds a complex modification here through several steps. By combining human genomic and RNA data with mass spectrometry of mouse embryonic tissues, the study confirmed that this site is normal in the DNA but chemically altered to varying degrees in the RNA, and that the proportion of this modified form differs between brain and facial tissues. Overexpressing or knocking out the enzymes that build this modification in cell cultures and in mouse embryos disturbed neural crest development and produced striking craniofacial defects, underscoring that precise levels of this RNA mark help steer cells into the skeletogenic pathway.
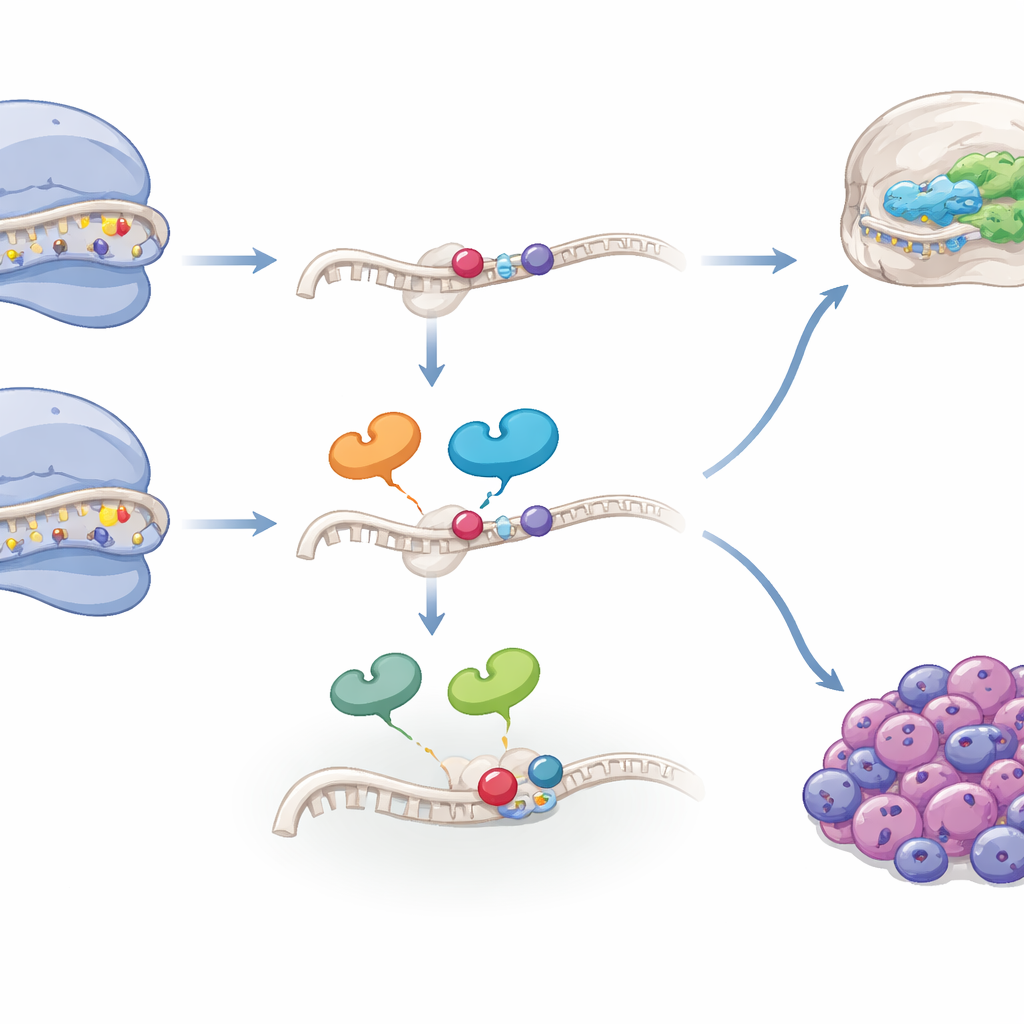
From development to cancer risk
Because neural crest cells can also give rise to tumors such as neuroblastoma, the authors asked whether the same ribosomal features appear in cancer. Analyzing single-cell and bulk RNA data from several neuroblastoma and brain tumor cohorts, they found that a ribosomal “control and RNA modification” gene signature is enriched in tumor cell subpopulations with more mesenchymal-like traits—states often linked to therapy resistance and poor prognosis. Across multiple patient cohorts, high levels of this signature strongly predicted worse survival, particularly in neuroblastoma cases lacking amplification of the well-known risk gene MYCN. Specific components, such as the assembly factor WDR74 and the modification enzyme TSR3, were elevated in aggressive cell lines and made these cells more vulnerable to drugs that stress the ribosome. Intriguingly, tumors with very low or very high levels of the key RNA modification at the highlighted site fared worse than those with intermediate levels, suggesting that both deficiency and excess of this specialized ribosome pool can be harmful.
What this means for faces and childhood cancers
Together, these findings suggest that ribosomes are not just passive protein factories but tuned devices whose chemical makeup helps decide how embryonic cells build the face and how some tumors behave. In neural crest cells, a particular chemical mark on ribosomal RNA appears to favor the production of proteins needed for forming facial bone and cartilage, while its misregulation in cancer is linked to more dangerous, shape-shifting tumor cells. Understanding and potentially targeting this ribosomal “fine print” could open new ways to prevent craniofacial malformations and to identify or treat high-risk neuroblastomas.
Citation: Poverennaya, I., Murtazina, A., Li, L. et al. Ribosomal modifications are associated with mesenchymal fate selection in the neural crest lineage. Nat Commun 17, 2326 (2026). https://doi.org/10.1038/s41467-026-70375-6
Keywords: neural crest development, ribosome modifications, craniofacial formation, neuroblastoma, specialized ribosomes