Clear Sky Science · en
Synchronous activation of striatal cholinergic interneurons induces local serotonin release
Why this brain chemistry discovery matters
Habits, motivation, and compulsive behaviors all depend on a tiny but vital brain region called the striatum. Two well-known brain messengers, dopamine and serotonin, help tune this region, and many psychiatric drugs aim to adjust their balance. This study reveals a previously hidden line of communication: a special class of nerve cells that release acetylcholine can directly tap into serotonin fibers and make them release more serotonin, but only in a specific part of the striatum. This newfound link could help explain why disorders like obsessive–compulsive disorder (OCD) and Parkinson’s disease often involve tangled changes in several brain chemicals at once.
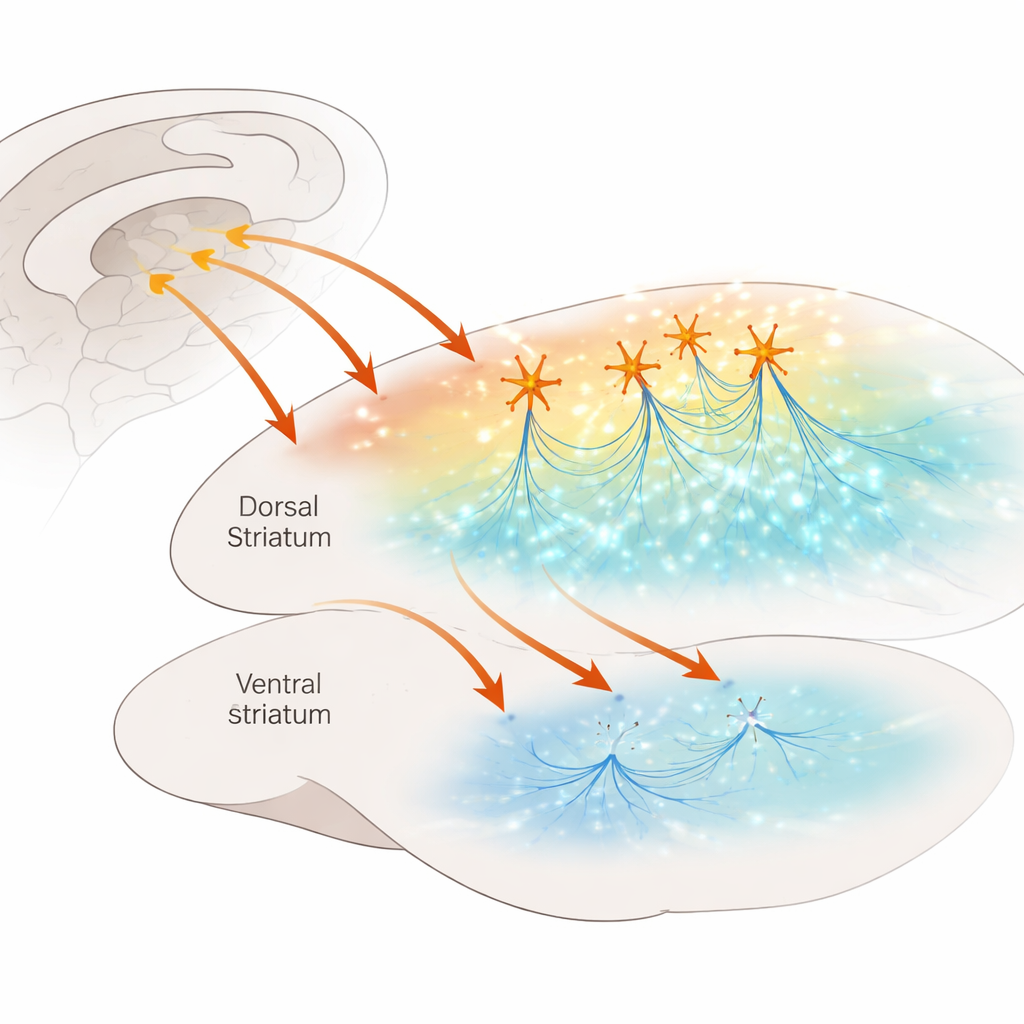
A busy crossroads in the brain
The dorsal striatum acts as a decision hub, helping us shift between goal-directed actions and automatic habits. It receives dense input from the cortex and is bathed in neuromodulators such as dopamine and serotonin, which fine-tune how signals are processed. Within this hub lives a sparse but powerful population of cholinergic interneurons—cells that release acetylcholine and fire regularly like tiny metronomes. Earlier work showed that when these cells fire together, they can strongly boost dopamine release by acting on nicotinic acetylcholine receptors sitting on dopamine fibers. Whether a similar shortcut exists for serotonin was unknown, even though serotonin is central to mood, learning, and the effects of drugs used to treat OCD and depression.
Watching serotonin light up in real time
To tackle this question, the researchers used mice in which a fluorescent sensor glows brighter when serotonin is present. They injected a virus carrying this sensor into the striatum, prepared thin brain slices, and then used two-photon microscopy to watch serotonin signals while they stimulated the tissue. Electrical stimulation inside the dorsal striatum caused clear, slowly decaying flashes of serotonin sensor activity. When the team applied a drug that blocks nicotinic acetylcholine receptors, these flashes shrank and became more confined in space, indicating that acetylcholine acting through these receptors helps both increase serotonin release and spread it over a wider area. Strikingly, doing the same experiment in the ventral striatum—a nearby region with even denser serotonin wiring—showed no such nicotinic contribution, revealing that this cross-talk is region-specific.
Triggering serotonin with targeted light
To find out if acetylcholine from cholinergic interneurons alone can drive serotonin release, the authors turned to optogenetics. They engineered these interneurons to respond to brief flashes of blue or yellow light and activated them synchronously while continuing to monitor the serotonin sensor. A single millisecond of light was enough to evoke a robust serotonin signal in the dorsal striatum, with a time course similar to the nicotinic-sensitive component of the electrically evoked response. Blocking nicotinic receptors almost completely abolished this signal, even when other chemical inputs and receptors in the slice were pharmacologically silenced. This combination of precise stimulation and receptor blockade showed that acetylcholine acts directly on nicotinic receptors along serotonin fibers to trigger local serotonin release, rather than relying on indirect routes through other cell types or dopamine axons.
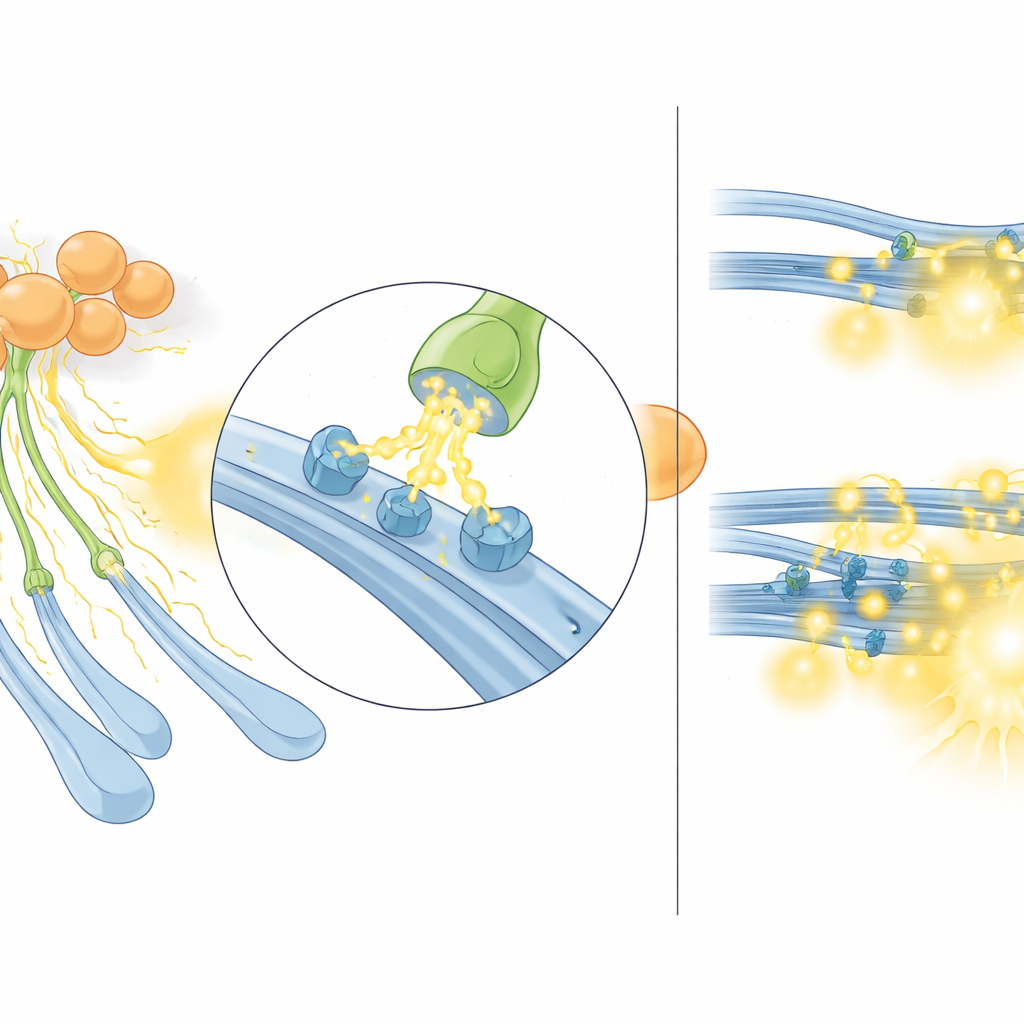
When the system runs too hot
The team next asked what happens in a disease-relevant state where acetylcholine signaling is abnormally strong. They used Sapap3 knockout mice, a well-established model that shows excessive grooming and other OCD-like behaviors, and that had previously been shown to have more active cholinergic interneurons and higher acetylcholine release. In these mice, electrical stimulation of the dorsal striatum produced larger serotonin sensor signals than in normal animals. Importantly, this difference vanished when nicotinic receptors were blocked, meaning that only the acetylcholine-dependent portion of serotonin release was boosted. A similar pattern held for dopamine. In other words, in this hypercholinergic state, cholinergic interneurons disproportionately amplify the nicotinic component of monoamine release, effectively letting acetylcholine “hijack” serotonin fibers as a main route for serotonin output.
Broader implications for brain disorders
These findings reveal a finely tuned and regionally confined conversation between acetylcholine and serotonin in the dorsal striatum. In healthy brains, synchronous bursts of cholinergic interneuron activity—such as those driven by sudden, behaviorally important events—may temporarily enlarge and strengthen serotonin signals, shaping how actions are learned or suppressed. In pathological states where acetylcholine is persistently elevated, as in Sapap3 knockout mice and possibly in conditions like OCD or Parkinson’s disease, this same mechanism may become exaggerated, contributing to abnormal patterns of serotonin and dopamine signaling. Understanding this hidden link offers a new way to think about how multiple neuromodulators interact in both normal behavior and disease, and may eventually point toward treatments that more precisely target their joint dynamics rather than treating each chemical in isolation.
Citation: Matityahu, L., Hobel, Z.B., Berkowitz, N. et al. Synchronous activation of striatal cholinergic interneurons induces local serotonin release. Nat Commun 17, 2278 (2026). https://doi.org/10.1038/s41467-026-70359-6
Keywords: striatal serotonin, cholinergic interneurons, nicotinic acetylcholine receptors, obsessive-compulsive disorder, basal ganglia