Clear Sky Science · en
SLIT3 fragments orchestrate neurovascular expansion and thermogenesis in brown adipose tissue
How Our Body’s “Good Fat” Helps Us Stay Warm
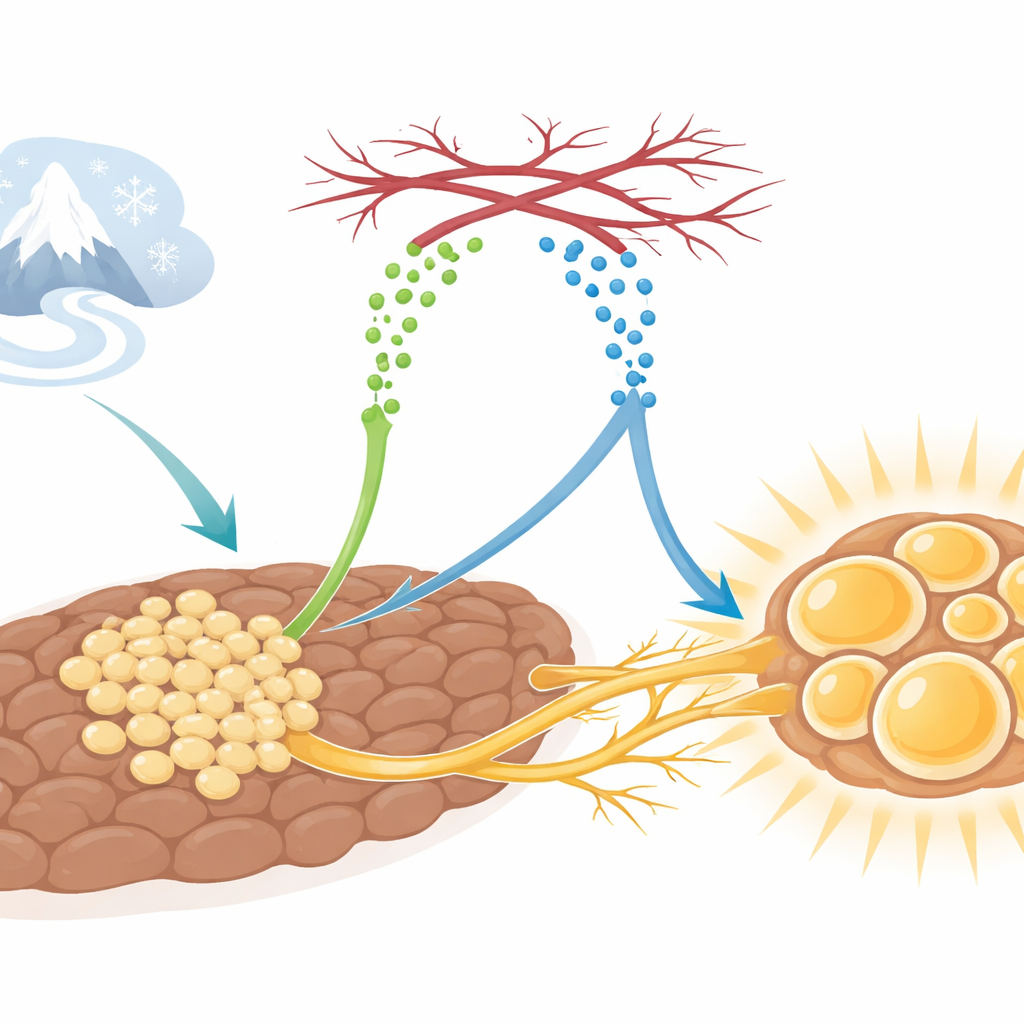
When we step into the cold, our bodies quickly work to keep us warm. A key player in this response is brown fat, a special kind of fat that burns calories to produce heat. This study uncovers how brown fat organizes its tiny blood vessels and nerve fibers to boost heat production, revealing a hidden communication system that could one day be tapped to improve metabolism and treat obesity-related disease.
A Hidden Furnace Inside the Body
Brown adipose tissue, or brown fat, acts like an internal furnace. Unlike the white fat that stores energy, brown fat consumes fuel to generate heat when we are exposed to low temperatures. To do this efficiently, brown fat cells need an abundant blood supply to deliver oxygen and nutrients, and a dense network of sympathetic nerves that signal when to turn up the heat. In the cold, these vessels and nerves expand remarkably. Yet, until now, it was unclear how these different parts of the tissue coordinate their growth so precisely.
The Messenger Molecule That Starts It All
The researchers discovered that a protein called SLIT3, produced by immature fat cells known as adipocyte progenitors, is central to this coordination. In mice exposed to cold, levels of SLIT3 in brown fat rise. When the team blocked SLIT3 specifically in brown fat or in these progenitor cells, the animals struggled to maintain body temperature in the cold. Their brown fat became “whiter,” with larger fat droplets, lower expression of heat-producing genes such as Ucp1, and diminished energy expenditure, even though food intake and activity levels were unchanged. This pointed to SLIT3 as a key organizer of brown fat’s response to cold, not by changing the basic machinery inside each cell, but by shaping the surrounding tissue environment.
Building Blood Vessels and Nerve Wires Together
Looking more closely, the scientists found that brown fat lacking SLIT3 had fewer tiny blood vessels and fewer sympathetic nerve fibers weaving through the tissue. Three-dimensional imaging confirmed that the nerve network was markedly sparser. Importantly, when the usual nerve signal was bypassed using a drug that directly activates brown fat, the cells responded normally. This showed that SLIT3 is not required for the fat cells to burn fuel, but rather for constructing the neurovascular “wiring” that allows the brain to control them. The team pinpointed adipocyte progenitors as the major local source of SLIT3 and showed that deleting SLIT3 only in these cells was enough to blunt vessel growth, nerve growth, and heat production during cold exposure.
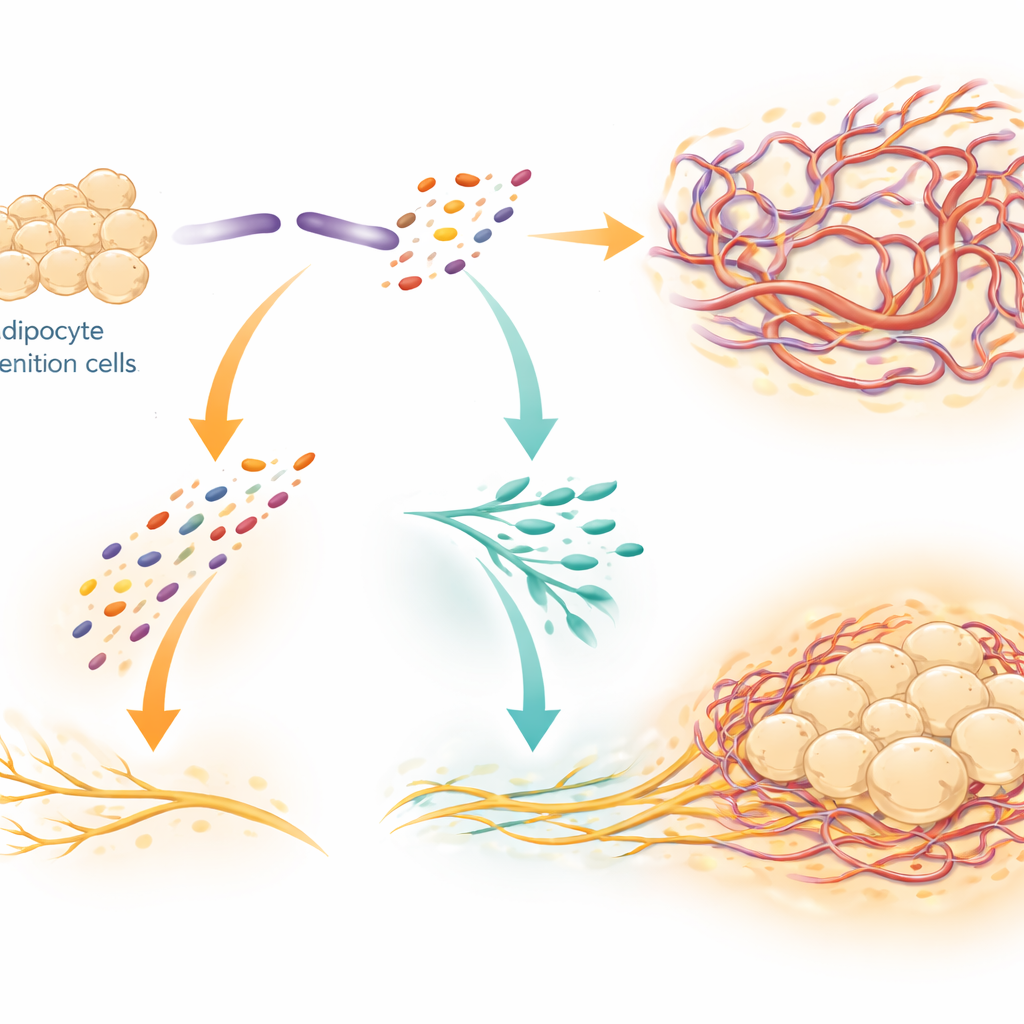
One Protein, Two Pieces, Two Jobs
SLIT3 does not act as a single, static signal. The researchers showed that an enzyme called BMP1 cuts SLIT3 into two separate fragments with distinct roles. The N-terminal fragment (SLIT3-N) mainly stimulates growth of capillaries, acting through receptors on endothelial cells that line blood vessels. The C-terminal fragment (SLIT3-C) primarily promotes sympathetic nerve expansion. They identified a receptor called PLXNA1 on nerve fibers as a direct docking partner for SLIT3-C, supported by biochemical binding tests and computer-based structural models. When PLXNA1 was reduced, the ability of SLIT3 or SLIT3-C to enhance nerve growth and raise brown fat temperature was lost, and overall nerve density and norepinephrine levels in the tissue dropped sharply.
Links to Human Metabolism and Health
To explore whether this system matters in people, the authors analyzed fat samples from large human obesity cohorts. Higher SLIT3 expression in adipose tissue was associated with healthier profiles: more of the hormone adiponectin, fewer inflammatory immune cells in visceral fat, better blood sugar control, and higher levels of the beneficial adipokine omentin. Genetic studies in other work have also linked variants near SLIT3 and its receptor PLXNA1 to obesity and insulin resistance. Together, these findings suggest that the SLIT3 pathway may help maintain healthy fat tissue and metabolic balance in humans.
What This Means for Staying Warm and Staying Healthy
Overall, the study reveals that immature fat cells in brown fat do more than just turn into mature fat cells; they actively sculpt the tissue’s support network. By releasing SLIT3 and allowing it to be cut into two fragments, they independently yet synchronously drive the growth of blood vessels and nerves needed for effective heat production. This finely tuned “two-in-one” signaling system ensures that brown fat can rapidly gear up when the environment demands more warmth. In the long run, understanding and safely harnessing this pathway could open new avenues to boost energy burning, improve fat tissue health, and combat metabolic diseases.
Citation: Serdan, T.D.A., Cervantes, H., Frank, B. et al. SLIT3 fragments orchestrate neurovascular expansion and thermogenesis in brown adipose tissue. Nat Commun 17, 2445 (2026). https://doi.org/10.1038/s41467-026-70310-9
Keywords: brown adipose tissue, thermogenesis, neurovascular remodeling, SLIT3 signaling, metabolic health