Clear Sky Science · en
NF-κB activation in astrocytes impairs wound healing after traumatic brain injury in male mice
Why Brain Bruises Don’t Always Heal Cleanly
When someone suffers a blow to the head, the visible injury on the outside is only part of the story. Deep inside the brain, a complex healing response unfolds that can determine whether a person recovers well or develops lasting problems with thinking, mood, or movement. This study in male mice uncovers how a key stress pathway inside support cells called astrocytes can turn a normal healing response into one that actually worsens damage after traumatic brain injury.
The Brain’s Hidden First Responders
Astrocytes are star-shaped cells that surround nerve cells and blood vessels, helping keep the brain’s internal environment stable. After a head injury, they rapidly change their shape and behavior, forming a barrier around the damaged area and helping to rebuild tissue. The researchers focused on a molecular switch inside these cells called NF-κB, which is well known for controlling inflammation in many tissues. They asked whether, after a closed-head injury similar to common human concussions and contusions, this switch helps or harms the brain’s ability to heal.
A Stress Switch That Lights Up After Impact
By analyzing gene activity in mouse brain tissue after injury, the team found a surge in genes linked to inflammation and NF-κB signaling, especially three to seven days after trauma, when secondary damage is ramping up. Using reporter mice, they showed that NF-κB activation was not spread throughout the brain but concentrated around the impact site. There, it was especially active in microglia (the brain’s immune sentinels) and in astrocytes forming the border of the wound. This timing and location suggested that NF-κB in astrocytes could be a critical regulator of how the brain organizes its scar and controls the immune response.
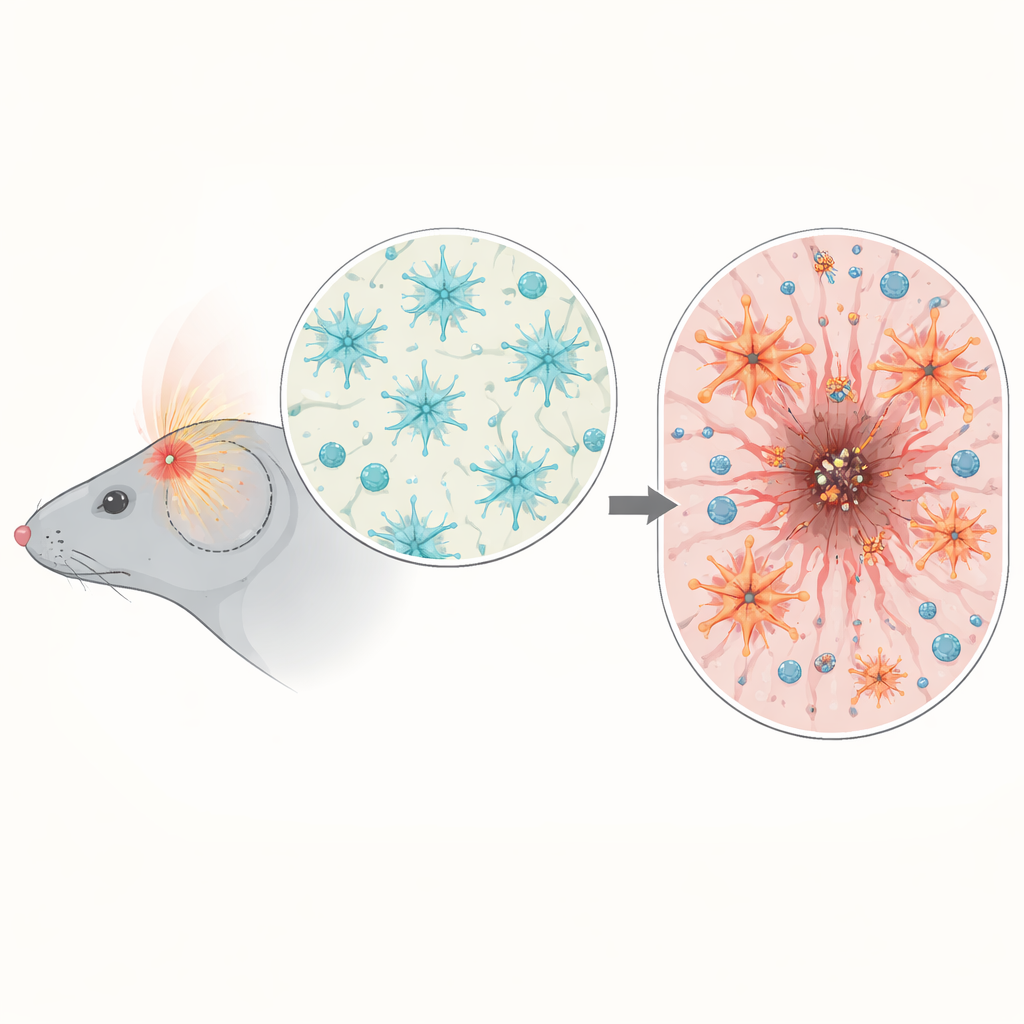
Turning Astrocytes Up Too High
To test cause and effect, the scientists engineered mice in which astrocytes either had NF-κB activity artificially boosted or dampened, but only after brain development was complete. When these mice experienced head trauma, those with overactive NF-κB in astrocytes lost more weight, showed worse early neurological scores, and had larger, more persistent brain lesions on both tissue sections and MRI scans. Instead of forming a neat, well-defined scar that walls off the damaged core, their astrocytes produced a thick, disorganized border that failed to shrink the wound over time. The normal balance of supporting matrix proteins around the injury was disturbed, and a key outer barrier structure known as the glial limitans formed poorly, leaving the surrounding brain less protected.
When Healing Programs Go Off Track
Diving into the molecular details, the team isolated astrocytes and nearby immune cells for gene-by-gene analysis. In healthy animals, injury normally suppresses some everyday housekeeping functions of astrocytes while switching on genes that support wound closure and regeneration. In contrast, astrocytes with chronically active NF-κB already looked “injured” even before trauma, and after impact they failed to fully engage beneficial repair programs such as those associated with a controlled transformation that helps cells build an effective border. Instead, they strongly favored inflammatory genes, adopted features of a toxic astrocyte state linked to aging and neurodegenerative disease, and showed signs of a “senescence-like” secretory profile associated with chronic, non-resolving wounds.
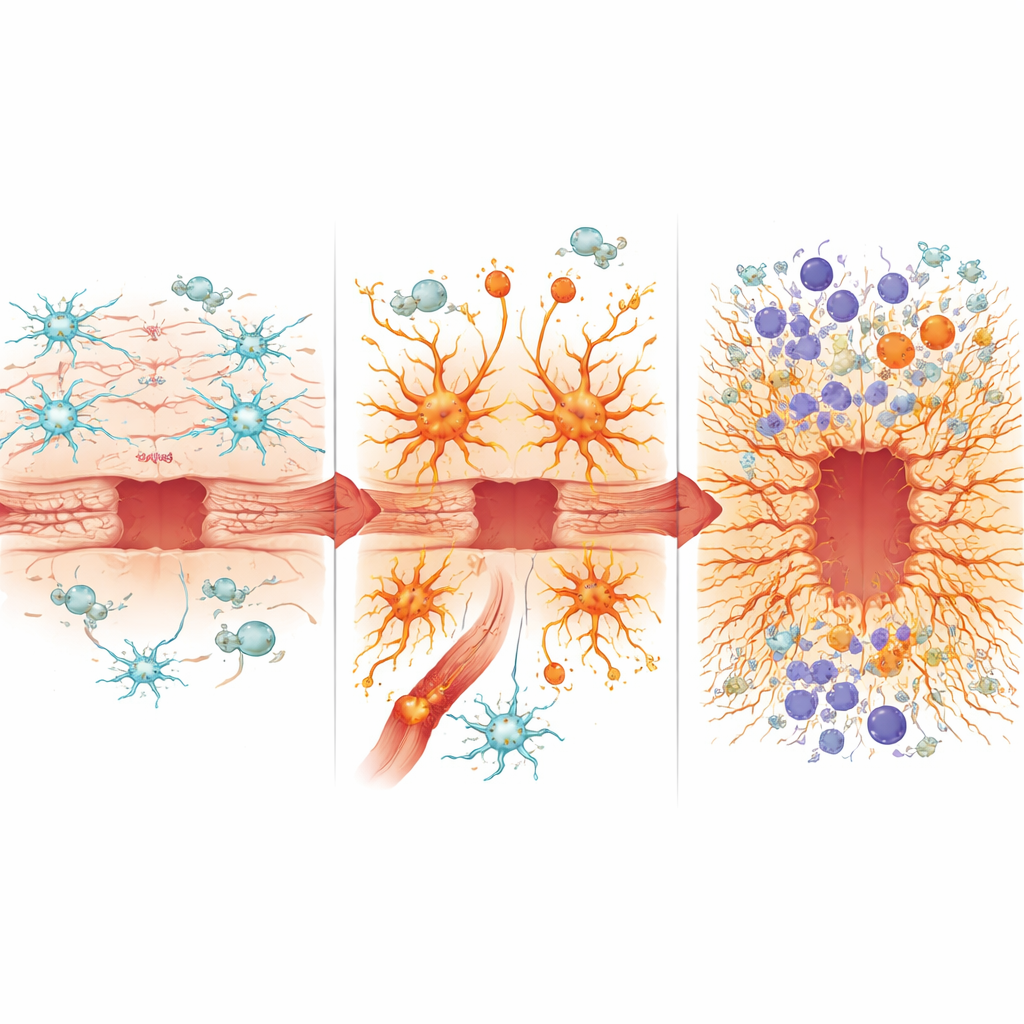
Stoking the Immune Fire
These misprogrammed astrocytes also reshaped the brain’s immune landscape. The blood–brain barrier became leakier, letting in more peripheral immune cells. Mice with overactive NF-κB in astrocytes showed heavier infiltration of inflammatory myeloid cells and altered mixtures of neutrophils, monocytes, and dendritic cells, as well as increased cytotoxic T cells that can further damage tissue. Immune cells in this environment expressed stronger signals driven by interferon and inflammasome pathways, both associated with aggressive, potentially neurotoxic inflammation. At the same time, levels of several protective or pro-repair proteins, such as osteopontin, were reduced, while others linked to oxidative stress, iron overload, and scarring, including heme oxygenase-1 and lipocalin-2, were elevated. The ratio between osteopontin and lipocalin-2 emerged as a simple marker that tracked with better or worse healing in the different mouse strains.
What This Means for Head-Injury Recovery
Together, the findings show that when the NF-κB stress pathway in astrocytes is chronically turned up, these cells shift from supporting organized repair to driving chaotic scarring and prolonged inflammation. Rather than building a clean border that contains the damage and allows tissue remodeling, they help create a leaky, inflammatory niche that enlarges the lesion and compromises long-term outcome. While the work was done in male mice, it points to astrocyte NF-κB as a promising target for therapies aimed at improving recovery after traumatic brain injury and suggests that blood levels of molecules such as osteopontin and lipocalin-2 might one day help clinicians monitor how well a patient’s brain is healing.
Citation: Hein, T.M., Nespoli, E., Hakani, M. et al. NF-κB activation in astrocytes impairs wound healing after traumatic brain injury in male mice. Nat Commun 17, 2323 (2026). https://doi.org/10.1038/s41467-026-70304-7
Keywords: traumatic brain injury, astrocytes, neuroinflammation, brain scar formation, NF-kappa B