Clear Sky Science · en
A human cerebral organoid model of West Nile virus encephalitis shows innate immunocompetency
Why tiny lab-grown brains matter
West Nile virus is a mosquito-borne infection that can invade the brain and cause encephalitis, a life-threatening inflammation that kills some patients and leaves many survivors with long‑term memory and movement problems. Yet doctors still have no specific antiviral drug or vaccine for people. One big reason is that it has been hard to study what the virus actually does inside the human brain. In this work, researchers use “cerebral organoids”—small, three‑dimensional clusters of human brain cells grown from stem cells—to build a realistic lab model of West Nile virus encephalitis and watch how brain cells fight back.
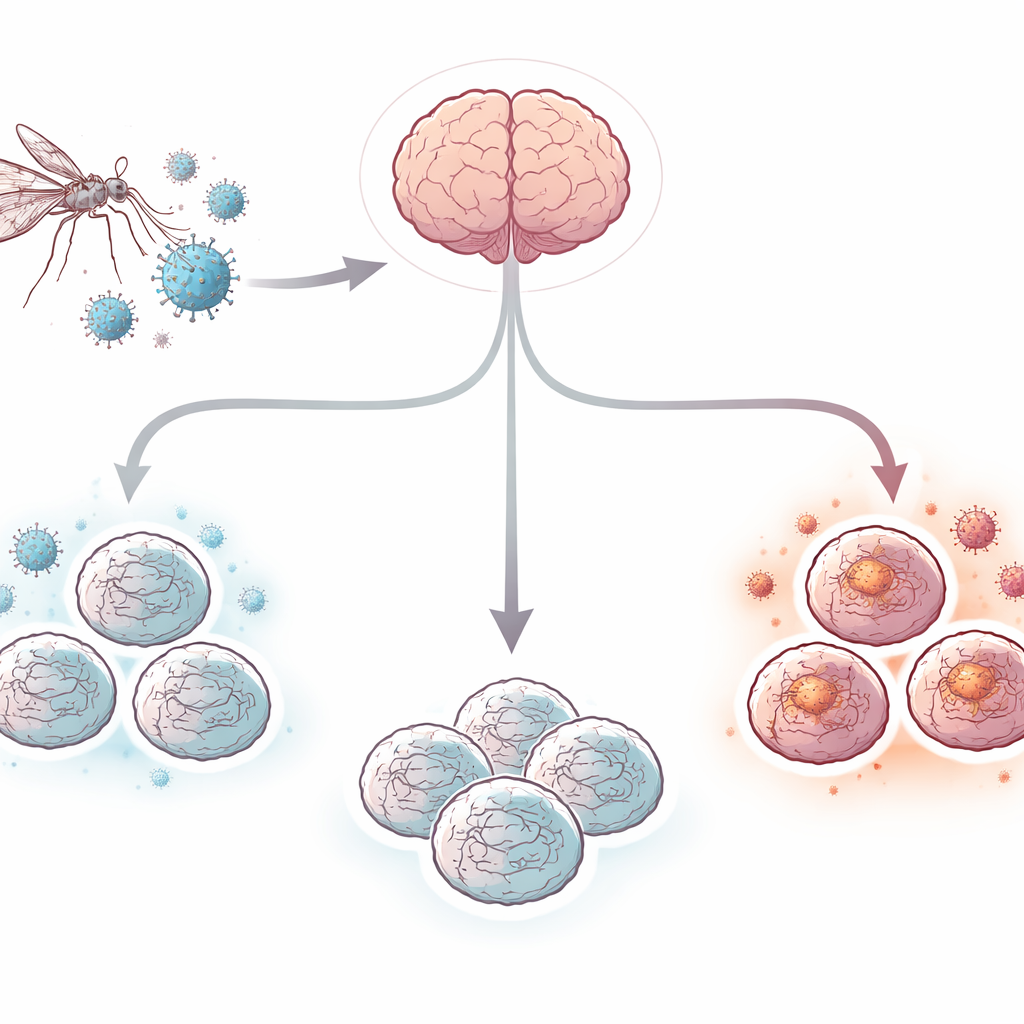
Building mini human brains in a dish
The team started with human-induced pluripotent stem cells, which can be coaxed to become many cell types. Using a stepwise recipe, they grew these cells for about 100 days into pea-sized cerebral organoids that mimic key features of the human brain’s outer layer. These mini-brains contained networks of nerve cells (neurons), support cells called astrocytes, and resident immune cells called microglia. Some organoids also formed structures resembling the choroid plexus, the tissue that produces cerebrospinal fluid and forms an important barrier between the blood and the brain. This mix of cell types created a more realistic environment than traditional flat cell cultures.
How West Nile virus behaves in mini-brains
When the organoids were exposed to a low dose of West Nile virus, the infection took hold strongly but not uniformly. Measurements of virus released into the surrounding liquid showed that some organoids quickly reached a peak within the first few days and then tended to quiet down; others ramped up more slowly and stayed infected for up to four weeks. In several of the early‑peaking organoids, virus levels later fell below the limit of detection, suggesting that the mini-brains were capable of clearing the infection on their own. Importantly, the organoids did not fall apart or show massive visible damage, indicating that the system could model both acute and longer‑term infection without simply disintegrating.
Where the virus strikes and who responds
Microscope images revealed that West Nile virus did not spread evenly through the mini-brains. Instead, viral proteins appeared in small clusters near the outer, cortex‑like regions rich in neurons and astrocytes. The viral material tended to gather around the cell nuclei, consistent with active infection inside these cells. In contrast, microglia were scattered in deeper layers and were largely absent from the virus‑positive areas, suggesting they were not the main early targets of infection and did not swarm to the infected spots in this model. This pattern mirrors findings from patient autopsies and animal studies, where neurons are primary targets but other brain cells shape the surrounding inflammation.
Chemical signals of brain inflammation
To understand how these tiny brains react, the researchers measured dozens of immune and damage‑related molecules secreted into the culture fluid over time. They saw a coordinated wave of signals associated with brain inflammation. Early on, the chemokine CXCL10 rose sharply, followed by other calling signals such as CCL2, CCL17, and CX3CL1 that would normally recruit circulating immune cells into the brain. Classic inflammatory messengers—including IL‑6, TNF‑α, and IL‑18—also increased, as did regulatory and damage‑linked markers such as IL‑1 receptor antagonist, sTREM‑1, sRAGE, and the nerve‑support factor BDNF. Many of these signals stayed elevated into the later stages of infection, hinting at how long‑lasting inflammation might contribute to lingering symptoms even after the virus is controlled.
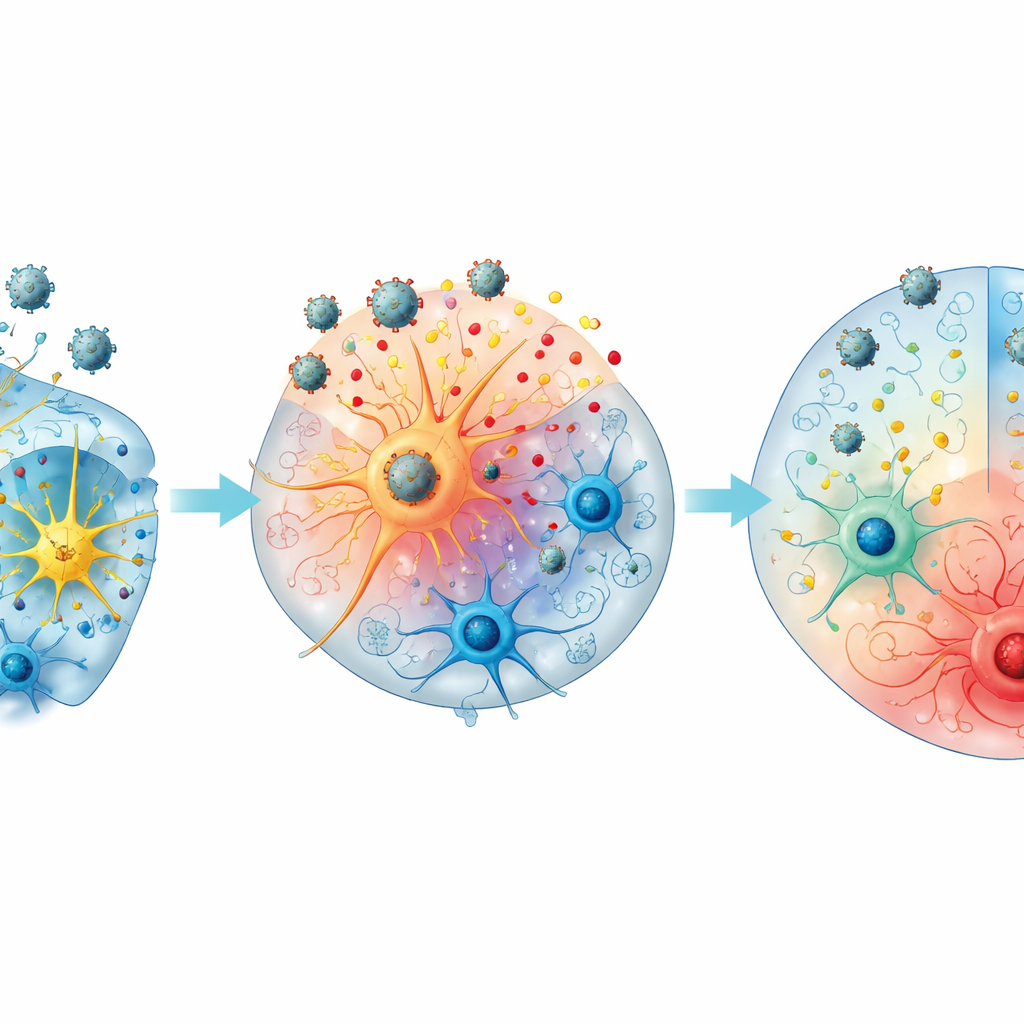
Different paths: clearing or sustaining infection
The team noticed that organoids followed two broad courses. “Type A” mini-brains, which reached their viral peak early, were more likely to later lose detectable virus and showed stronger surges in some inflammatory and regulatory signals, including CXCL10, IL‑1 receptor antagonist, and sTREM‑1. “Type B” organoids, with later peaks, more often maintained high viral levels and showed relatively higher IL‑18 and certain damage markers, suggesting a tilt toward prolonged inflammation. Organoids that developed choroid plexus–like structures tended to produce even more of some signals such as IL‑6, CXCL10, CX3CL1, and β‑NGF, pointing to a possible role of this barrier tissue in shaping the brain’s response to infection.
What this means for patients
By showing that human cerebral organoids can be infected with West Nile virus, mount complex immune responses, and even sometimes clear the virus, this study establishes a powerful new stand‑in for the human brain. For lay readers, the key takeaway is that scientists can now watch a human‑like brain tissue fight a mosquito‑borne virus in real time, without experimenting directly on patients. This model should help unravel why some infections resolve while others linger, how local brain cells contribute both to protection and to damage, and which signaling pathways might be targeted to prevent long‑term neurological problems. In the future, similar organoid systems could be used to test antiviral drugs, explore vaccines, and study other brain‑infecting viruses under controlled conditions.
Citation: Steffen, J.F., Widerspick, L., Jansen, S. et al. A human cerebral organoid model of West Nile virus encephalitis shows innate immunocompetency. Nat Commun 17, 2318 (2026). https://doi.org/10.1038/s41467-026-70281-x
Keywords: West Nile virus, cerebral organoids, viral encephalitis, brain inflammation, neurotropic viruses