Clear Sky Science · en
Mechanisms of PfDNMT2 inhibition and PfATP6-mediated resistance to the antimalarial candidate SC83288 in Plasmodium falciparum
Why this matters for malaria
As malaria parasites become resistant to today’s medicines, doctors are running out of options to treat a disease that still kills hundreds of thousands of people each year, mostly young children. This study dissects how a promising experimental drug, called SC83288, attacks the deadliest malaria parasite and how the parasite sometimes manages to fight back. Understanding both the drug’s punch and the parasite’s counterpunch is essential for turning SC83288 into a future therapy that remains effective for as long as possible. 
A new weapon against a deadly parasite
SC83288 emerged from efforts to improve on an old veterinary drug and now stands out as a powerful antimalarial candidate. In laboratory tests and animal models, it rapidly clears malaria parasites from the blood at very low concentrations, including strains that already resist frontline treatments such as artemisinin-based combinations and older medicines like antifolates and quinolines. It also acts on the parasite stages that can be transmitted to mosquitoes. Extensive safety testing in animals has so far revealed no major red flags, positioning SC83288 as a realistic contender for clinical trials in humans.
How the drug shuts down parasite growth
The researchers followed the parasite’s 48‑hour cycle inside red blood cells and added SC83288 at different times. When the drug was present during the first two thirds of this cycle, parasites stopped copying their DNA and failed to divide their nuclei, a process called karyokinesis. Under the microscope, treated parasites became small and condensed, a hallmark of dying cells, while later steps such as splitting into daughter parasites and bursting out of the red blood cell were unaffected if division had already finished. This pinpointed SC83288’s main effect to the crucial window when the parasite’s genetic material is being duplicated in preparation for making many offspring.
Striking at the parasite’s “epigenetic” control center
To understand what SC83288 hits inside the parasite, the team combined gene expression profiling, metabolic measurements, and direct enzyme tests. They found strong signs that the drug disrupts how the parasite uses tiny chemical tags to control DNA and RNA. Levels of a key donor molecule for these tags, S‑adenosylmethionine, became unbalanced, and global marks on DNA and RNA dropped sharply after treatment. Focusing on one central enzyme, PfDNMT2, which normally adds methyl groups to both DNA and a specific transfer RNA, the scientists showed that SC83288 directly inhibits its activity in test tubes and in parasites. When parasites were engineered to make much more PfDNMT2, they became less sensitive to the drug, whereas extra supplies of methyl‑donor molecules partially rescued parasites from SC83288’s effects. Together, these results support a model in which SC83288 kills by undermining the parasite’s epigenetic and gene‑expression machinery, leading to stalled DNA replication and cell death. 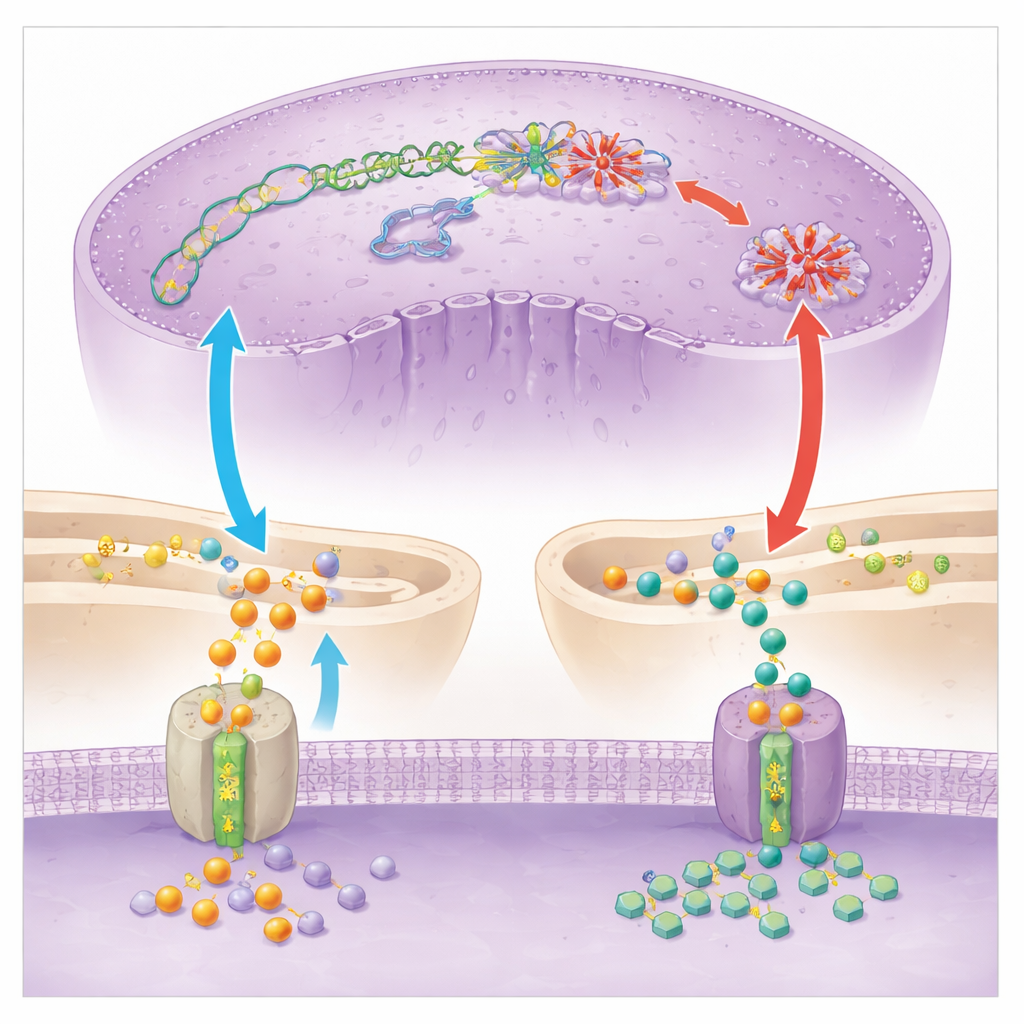
How the parasite dodges the blow
No potent drug comes without the risk of resistance, and SC83288 is no exception. After long‑term exposure in culture, some parasite lines acquired changes not in the target enzyme PfDNMT2, but in a membrane pump called PfATP6, which normally helps control calcium levels inside the parasite’s endoplasmic reticulum, a network of internal membranes. Using genetic engineering, the team introduced one of these mutations into a standard parasite strain and showed it made parasites about a thousand times less sensitive to SC83288. Detailed transport experiments in yeast and in parasites, as well as imaging with a clickable fluorescent version of the drug, revealed why: mutant PfATP6 acts like a vacuum cleaner, pulling SC83288 away from the nucleus and into the endoplasmic reticulum. The drug still gets into infected red blood cells, but is now sequestered in a compartment where it can do less harm to nuclear processes.
The hidden cost of resistance
Although the PfATP6 mutation shields parasites from SC83288, it comes at a heavy price. Resistant parasites grew much more slowly than their unmutated counterparts and showed disturbed calcium balance, indicating that the altered pump does its normal job poorly. In mixed cultures, the fitter wild‑type parasites quickly outcompeted the mutants when no drug was present. This suggests that while such resistance could arise under strong drug pressure, it is unlikely to spread widely in the field because resistant parasites would be at a disadvantage whenever SC83288 is absent. From a public‑health perspective, this high fitness cost, combined with the drug’s novel way of hitting the parasite’s epigenetic control system, makes SC83288 a particularly attractive candidate in the race to stay ahead of malaria.
Citation: Sanchez, C.P., Duffey, M., Celada, R.V. et al. Mechanisms of PfDNMT2 inhibition and PfATP6-mediated resistance to the antimalarial candidate SC83288 in Plasmodium falciparum. Nat Commun 17, 2327 (2026). https://doi.org/10.1038/s41467-026-70280-y
Keywords: malaria drug resistance, Plasmodium falciparum, epigenetic therapy, DNA methylation, antimalarial development