Clear Sky Science · en
NAA40 and NAC cooperate in co-translational histone acetylation in humans
How cells fine-tune their genetic switches
Inside every human cell, newly made proteins are chemically tweaked as they emerge from the cell’s protein-making machines, the ribosomes. These tiny adjustments can decide where a protein goes, how long it lives, and even how our genes are turned on or off. This paper focuses on a highly specific protein called NAA40 and a helper complex called NAC, revealing how they work together on the ribosome to modify histone proteins—the spools around which DNA is wrapped—and thereby influence gene activity and potentially cancer development.
A tiny cap that changes histone behavior
Many human proteins receive a small chemical cap at their starting end, a process known as N-terminal acetylation. Histones H2A and H4, which help package DNA into chromatin, are almost always capped in this way. NAA40 is unusual among enzymes because it specializes almost exclusively in adding this cap to these histones as they are being produced. This acetyl mark competes with other chemical tags such as phosphorylation and methylation on the same region of histones, so whether or not NAA40 acts can shift how tightly DNA is packaged and how genes are regulated. Importantly, altered levels of NAA40 have been linked to several cancers, making it a promising target for new therapies.
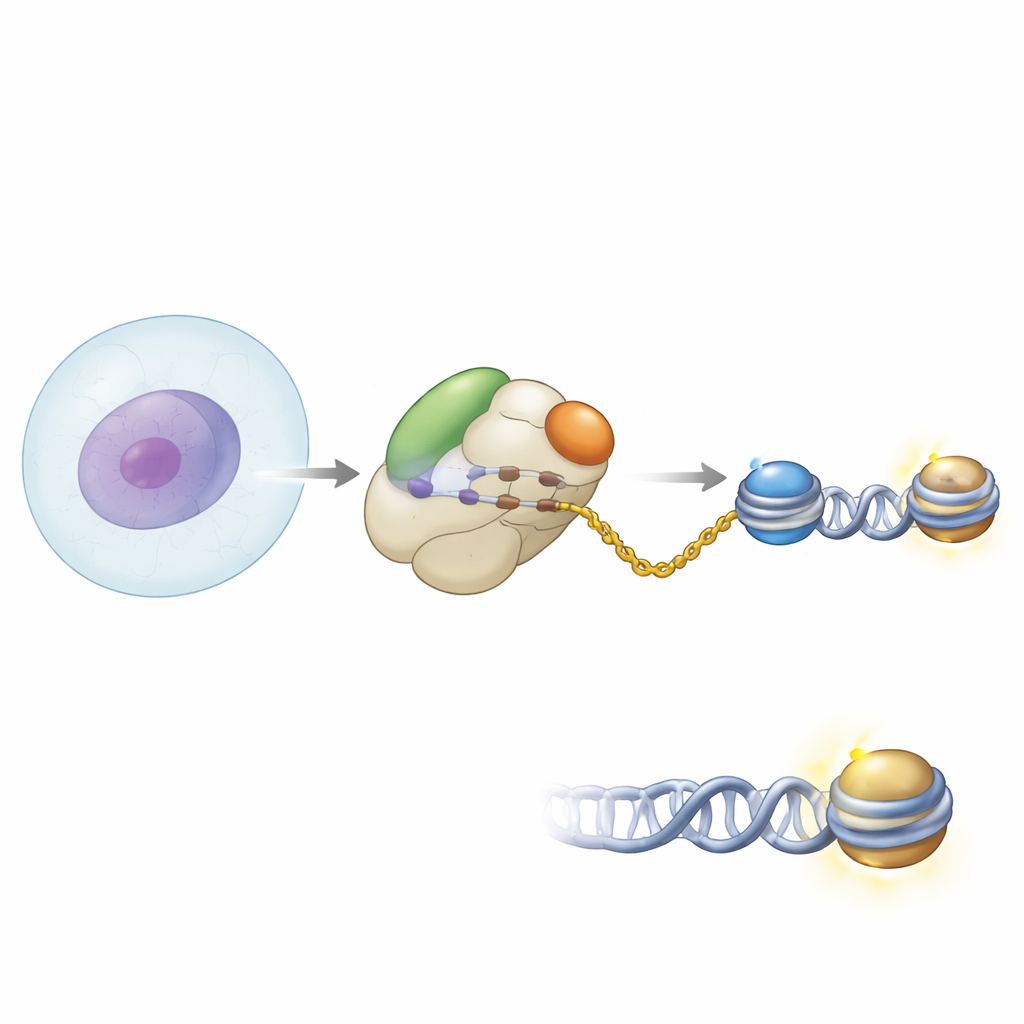
Partnering at the protein factory gate
The authors used biochemical methods and high-resolution cryo–electron microscopy to see how NAA40 is positioned on the human ribosome. They found that NAA40 sits right at the tunnel exit where newborn protein chains emerge. There, it does not work alone: it binds alongside the nascent polypeptide–associated complex, or NAC, a common ribosome-bound helper that watches over new proteins as they come out. Structural snapshots showed NAA40 anchored to the ribosome by a unique positively charged helical segment at its own beginning, while NAC is tethered via one of its subunits to the ribosomal surface. A flexible domain of NAC reaches over to contact NAA40, effectively bridging the enzyme to the ribosome.
Why NAC is needed for histone tuning
To test how important this partnership is, the researchers reconstituted these interactions with purified components in test tubes. They observed that NAA40 on its own binds only weakly to ribosomes, but when NAC is present, ribosome binding becomes much stronger. If they removed a specific “UBA” region from NAC that contacts NAA40, this enhanced binding disappeared. In human cells, depleting NAC led to reduced NAA40-dependent acetylation of histone H4 and a corresponding rise in a rival phosphorylation mark on the same histone tail. This change occurred without altering the amount or location of NAA40 itself, indicating that NAC’s main role is to recruit and position NAA40 correctly on translating ribosomes so histones can be modified efficiently as they are made.
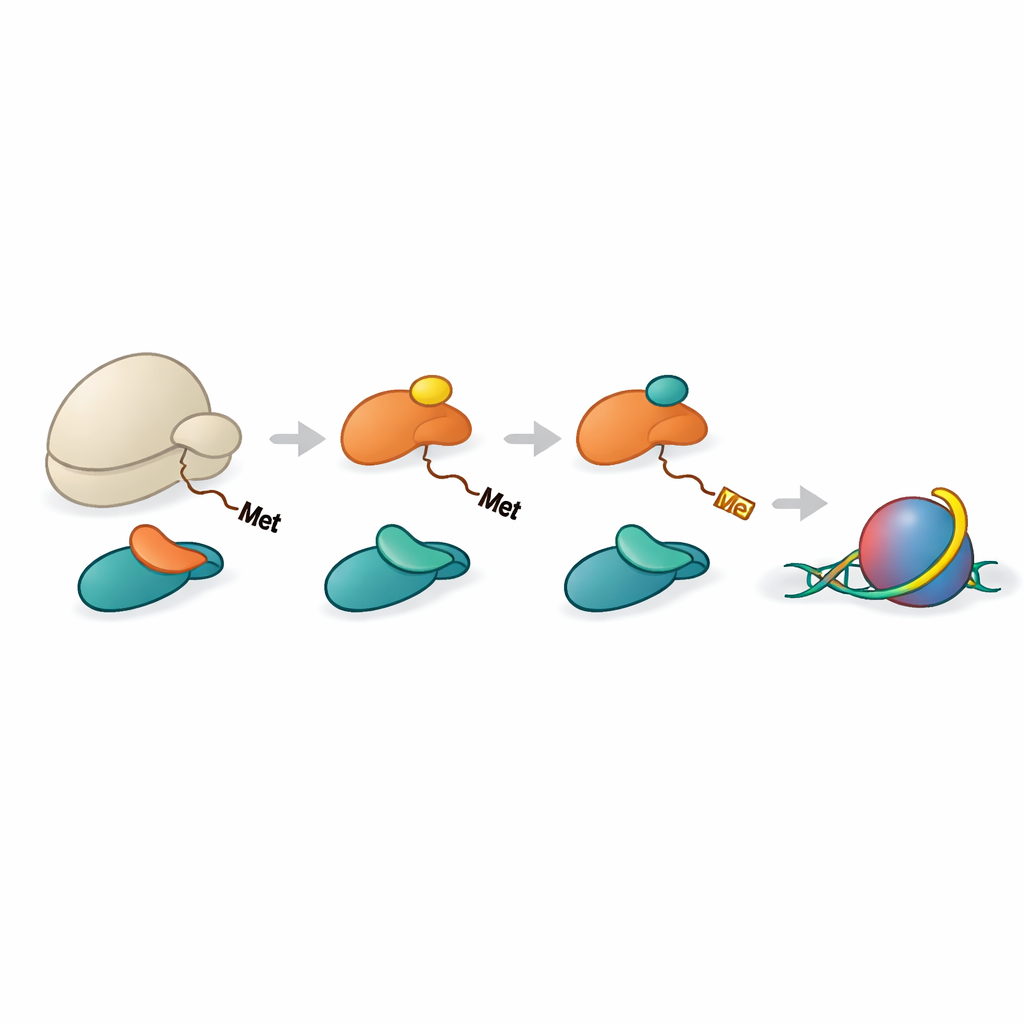
A coordinated assembly line for histone processing
Histones H2A and H4 must first lose their initial methionine amino acid before NAA40 can recognize them. Another enzyme, METAP1, performs this trimming. The team showed that NAC can help bring METAP1 and NAA40 together with the ribosome, forming a multi-enzyme assembly at the peptide exit. High-resolution structures of reconstituted complexes revealed METAP1 and NAA40 bound side-by-side on the ribosome, with their active sites positioned at almost equal distances from the exit tunnel. This geometry means that, once a histone tail extends just a dozen or so amino acids beyond the ribosome, METAP1 can remove the starter methionine and NAA40 can immediately add the acetyl cap, minimizing any delay between the two steps.
Implications for gene control and cancer
Overall, the study shows that NAA40 does not simply float through the cell looking for histones to modify. Instead, it is docked to the ribosome by NAC, forming part of an organized processing station that prepares histones the moment they are synthesized. By tightly coupling methionine removal and acetyl addition, the cell ensures that histone H2A, H4, and the DNA damage–linked variant H2A.X quickly receive an acetyl mark that can shape chromatin structure and gene expression. Because disruptions in this early histone marking step can favor cancerous growth, understanding the precise arrangement of NAA40, NAC, and METAP1 on the ribosome provides a structural blueprint that could guide the design of drugs aimed at fine-tuning this critical epigenetic control point.
Citation: Guan, D., Denk, T., Klavaris, A. et al. NAA40 and NAC cooperate in co-translational histone acetylation in humans. Nat Commun 17, 2486 (2026). https://doi.org/10.1038/s41467-026-70279-5
Keywords: histone acetylation, NAA40, ribosome, nascent polypeptide-associated complex, epigenetic regulation