Clear Sky Science · en
A cell cycle-dependent transition of acetylation to phosphorylation regulates timely centrosome maturation
Keeping Our Genetic Cargo on Track
Every time a cell in our body divides, it must parcel out its DNA with exquisite precision. When this process goes wrong, cells can gain or lose chromosomes, a dangerous state linked to cancer and other diseases. This study uncovers a molecular timing device that helps cells build the tiny structures needed to separate chromosomes correctly, revealing a potential weak spot that future anti-cancer therapies could exploit.
The Cell’s Tiny Traffic Hubs
At the heart of each dividing cell sit centrosomes, small structures that act like traffic hubs for the fibers that pull chromosomes apart. Before a cell enters division, these hubs must “mature”: they bulk up with helper proteins and become powerful organizers of microtubules, the dynamic fibers that form the division spindle. If this maturation happens too early, too late, or not at all, chromosomes can be misdistributed, leading to cells with abnormal chromosome numbers. Understanding how cells switch centrosomes on at just the right moment has been a long-standing question in cell biology.
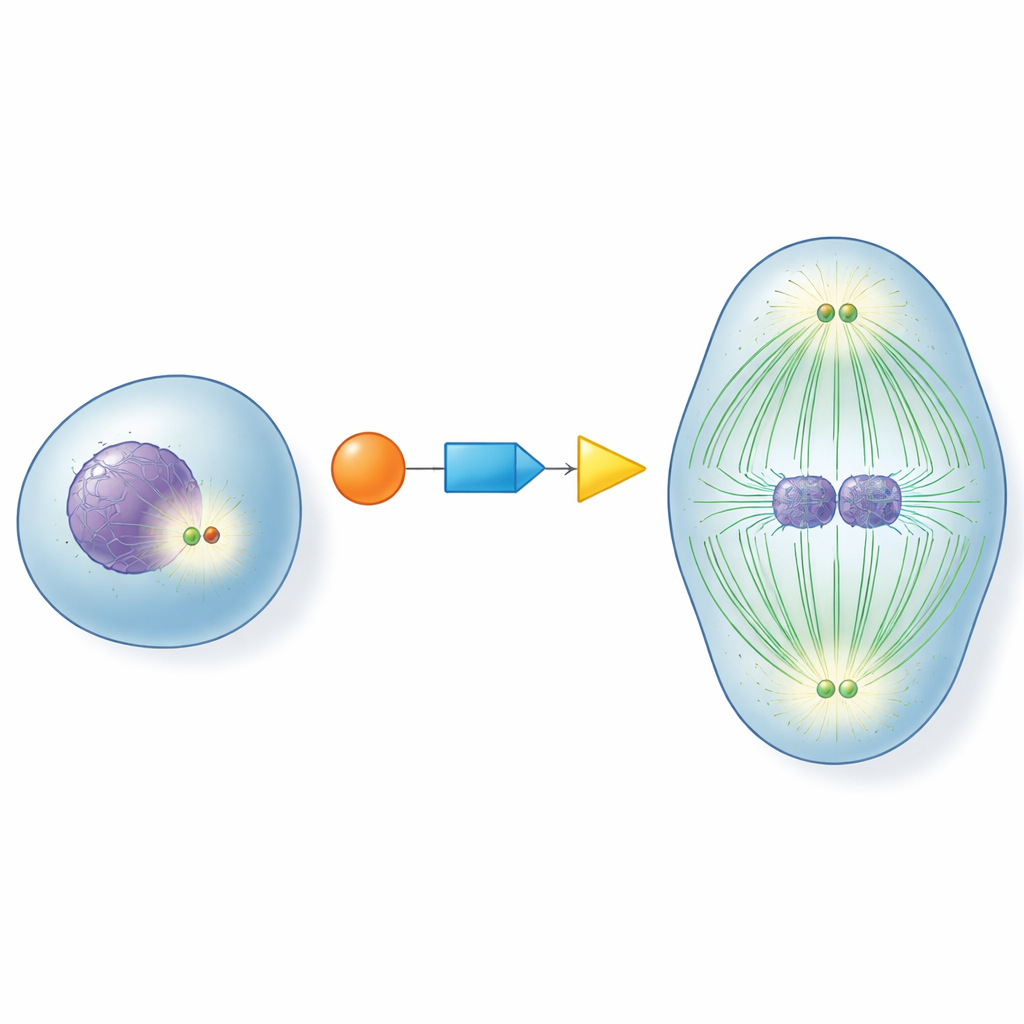
A Molecular Relay for Perfect Timing
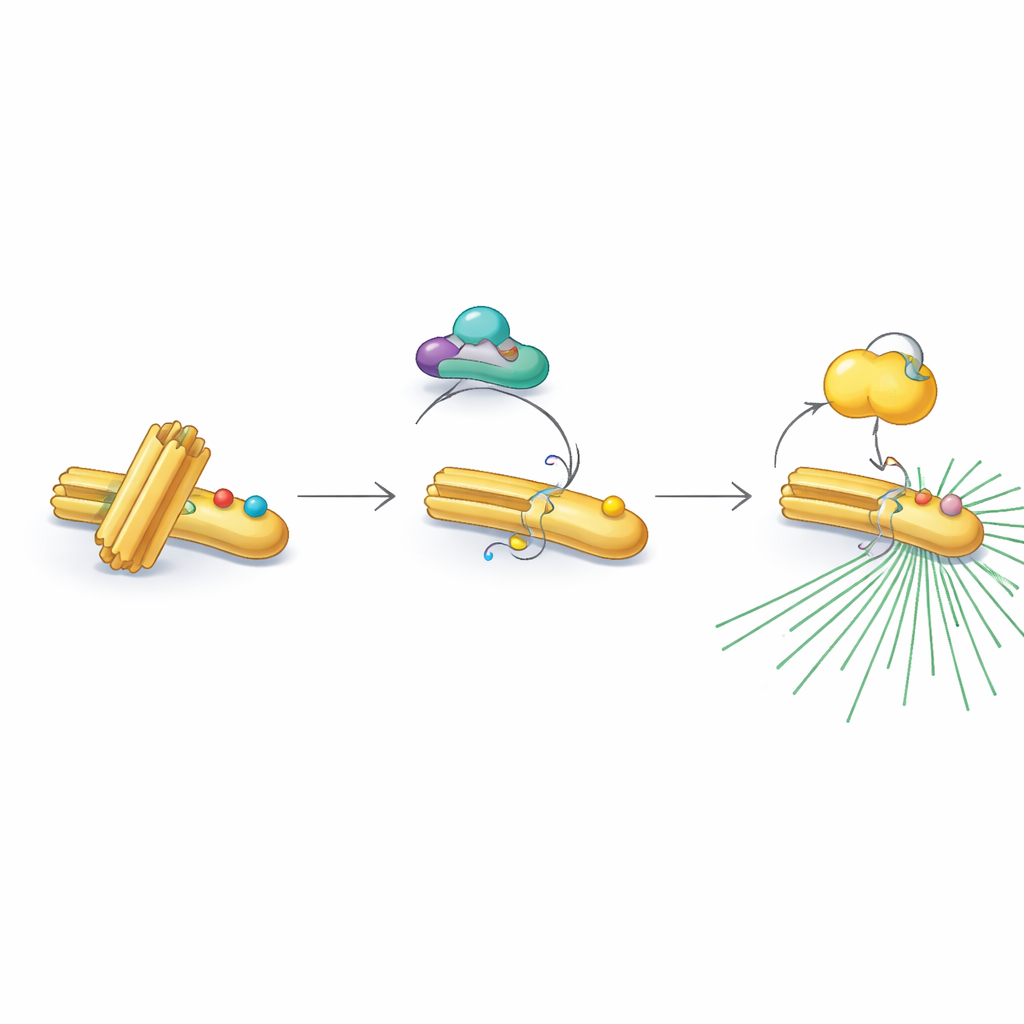
The authors focused on three key proteins that act together as a relay: CDK1, a master cell-cycle controller; RNF40, previously known mainly for modifying DNA-packaging proteins; and PLK1, a major driver of cell division. They discovered that RNF40 is physically present at centrosomes throughout the cell cycle and directly binds PLK1. As cells approach division, CDK1 chemically tags RNF40 at two specific points, which in turn makes RNF40 a better docking site for PLK1. This CDK1–RNF40–PLK1 chain ensures that PLK1 arrives at centrosomes precisely during the late preparation phase of division, triggering robust centrosome maturation, microtubule growth, and assembly of a well-formed bipolar spindle.
A Shape-Shifting Protein Switch
Intriguingly, RNF40 does not always accept these activating tags. In non-dividing and DNA-copying phases, RNF40 carries a different chemical decoration—acetyl groups—on two nearby positions. These acetyl marks are added by an enzyme called PCAF and removed later by a deacetylating partner, HDAC1. The acetylated version of RNF40 resists modification by CDK1, effectively blocking the next step in the relay. As cells move into the final pre-division stage, HDAC1 gradually erases the acetyl marks, allowing CDK1 to add phosphate groups instead. This carefully timed switch from acetylation to phosphorylation converts RNF40 from a “standby” state into an “on” state, ready to recruit PLK1 and power up the centrosome.
What Happens When the Timing Fails
To probe how crucial this switch is, the researchers engineered cells in which RNF40 could no longer be phosphorylated, or in which it was forced to stay in an acetylation-mimicking state. In both cases, PLK1 failed to accumulate properly at centrosomes. These cells showed weakened microtubule growth from centrosomes, malformed spindles, and misaligned chromosomes. Many ended up with extra or missing chromosomes—an abnormal condition known as aneuploidy—and some became binucleated, signs of catastrophic division errors. In cancer models, cells locked in the “acetylated” RNF40 state formed smaller tumors in mice and were more sensitive to common chemotherapy drugs used to treat colorectal cancer, suggesting that disrupting this switch can slow tumor growth.
Links to Human Cancer and Future Possibilities
The team also examined patient data and tumor samples. They found that RNF40 is often abnormally abundant in colorectal cancers and that certain cancer-associated mutations near its modification sites disrupt its phosphorylation, undermining proper centrosome function. These observations connect the newly described timing mechanism directly to human disease. By pinpointing a specific protein that coordinates when centrosomes mature and how chromosomes are separated, the study highlights a signaling “node” that could be targeted to push rapidly dividing cancer cells into lethal division errors, while leaving healthy cells less affected.
A New Handle on Faithful Cell Division
For non-specialists, the key message is that cells rely on a finely tuned chemical switch on a single protein, RNF40, to decide exactly when to activate the machinery that separates chromosomes. This acetylation-to-phosphorylation transition behaves like a traffic light at a busy intersection, turning green only when the cell is truly ready to divide. When the light malfunctions, cells stumble through division, accumulate genetic mistakes, and can become cancerous. Understanding and controlling this switch may open new paths to treatments that selectively destabilize cancer cells by sabotaging their ability to divide cleanly.
Citation: Li, J., Liang, J., Chen, G. et al. A cell cycle-dependent transition of acetylation to phosphorylation regulates timely centrosome maturation. Nat Commun 17, 2583 (2026). https://doi.org/10.1038/s41467-026-70271-z
Keywords: centrosome maturation, cell division, chromosome instability, PLK1 signaling, colorectal cancer