Clear Sky Science · en
Restoring early postnatal synaptic dysregulation rescues motor neuron degeneration in a mouse model of Spinal and Bulbar Muscular Atrophy
Why tiny early changes can matter for later muscle weakness
Spinal and bulbar muscular atrophy (SBMA) is a rare inherited condition in which adults, usually men, slowly lose strength in their limbs, trunk and throat. Symptoms appear in mid-life, but subtle problems begin much earlier. This study asks a surprising question: could brief events in the first days after birth quietly set the stage for nerve-cell loss decades later—and could correcting those early glitches protect movement?
A disease rooted in a hormone-sensitive switch
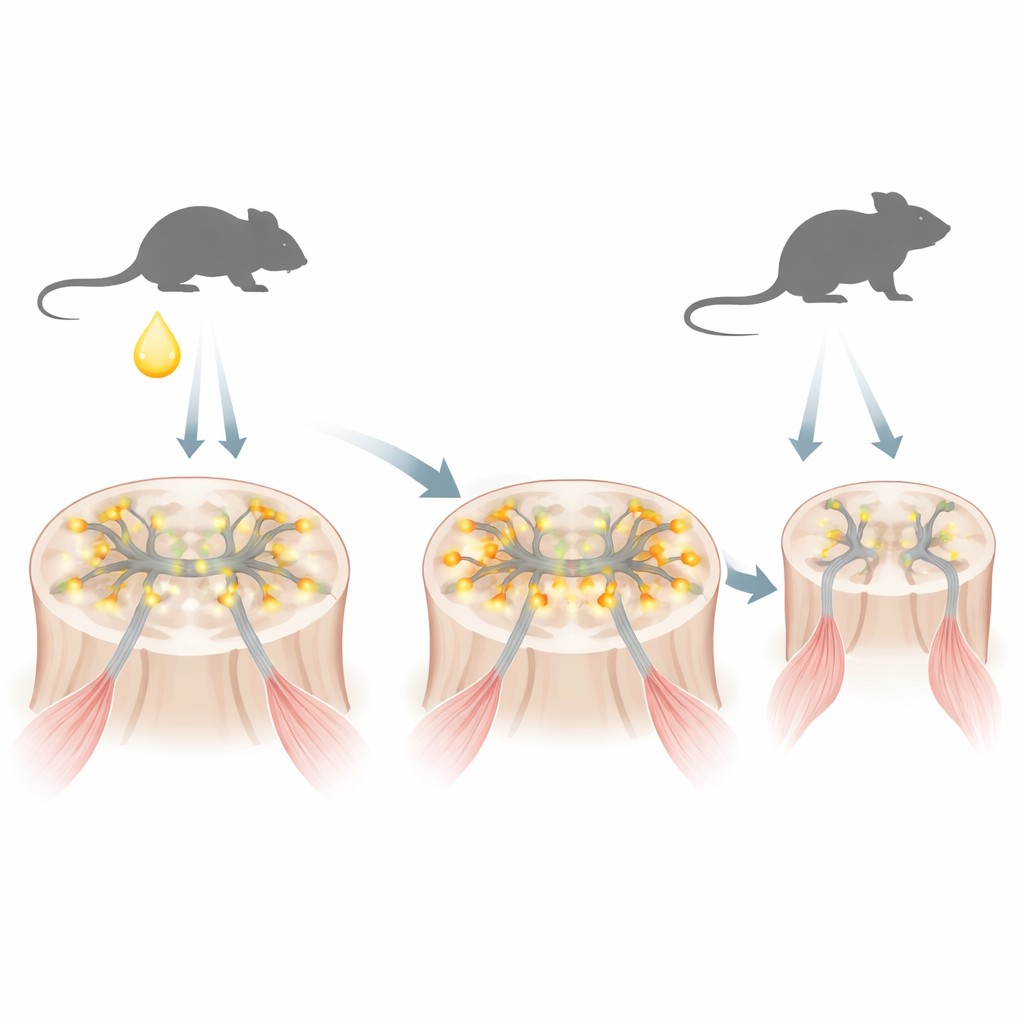
SBMA is caused by an altered version of the androgen receptor, a protein that senses male hormones such as testosterone. The altered receptor carries an overly long stretch of the amino acid glutamine. In a mouse model that mimics the human disease, the authors found that right after birth, when testosterone briefly surges in male infants, this mutant receptor rapidly accumulates inside the nuclei of motor neurons—the nerve cells that control muscles. At this early stage the protein has not yet clumped into the large aggregates typically linked to neurodegeneration, but it is already changing which genes are turned on and off.
Early synapse overdrive and restless motor neurons
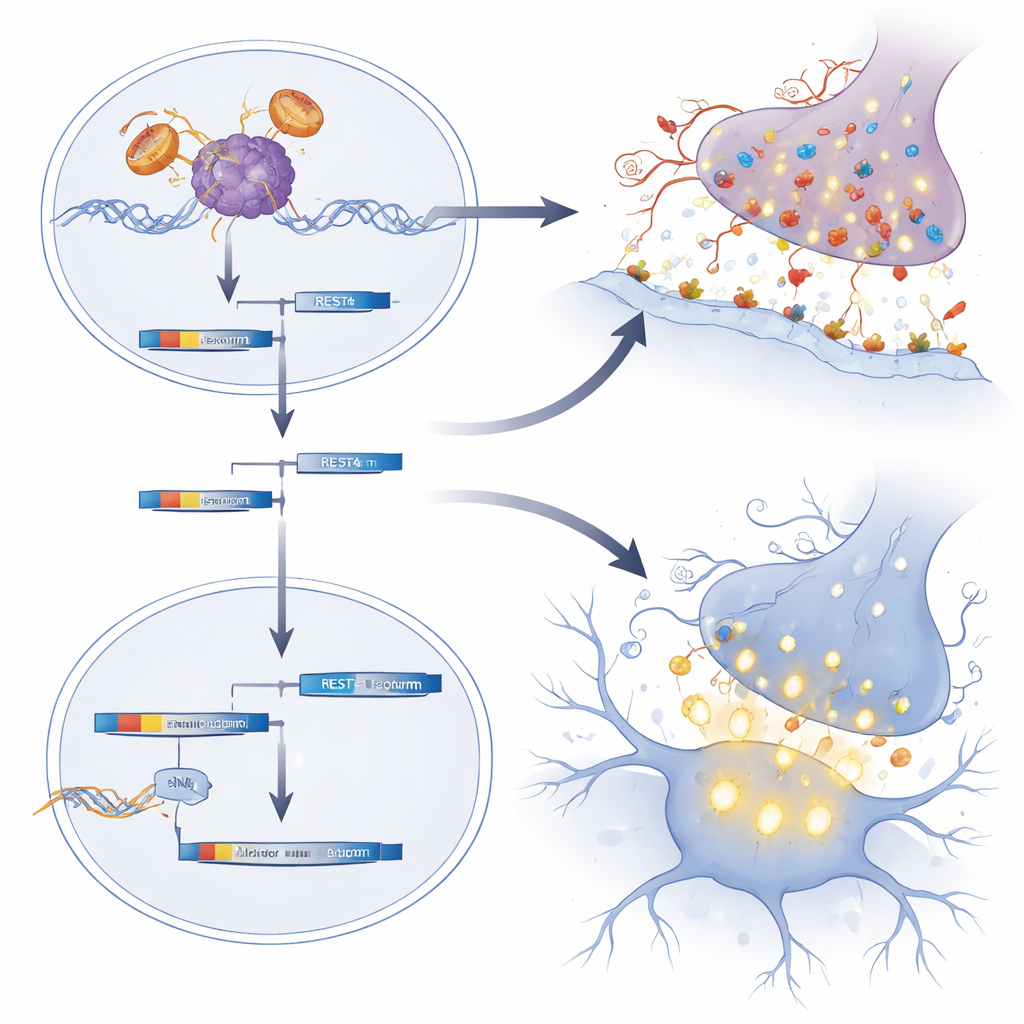
Using genome-wide RNA sequencing of newborn mouse spinal cords, the team discovered that many genes involved in excitatory synapses—the contact points where nerve cells signal to each other—were unusually active. Many of these genes encode glutamate receptors, which make neurons more likely to fire. The group traced this pattern to disruption of REST, a master “brake” protein that normally keeps such synaptic genes under tight control during development. In SBMA mice and in motor neurons grown from patients’ induced pluripotent stem cells, REST’s activity was weakened and a shortened form called REST4 was favored, lifting the brake and boosting glutamatergic synapse genes. Consistent with this, newborn SBMA motor neurons showed higher levels of c-Fos, a marker of recent firing, and human patient–derived motor neurons displayed stronger and more frequent calcium bursts, hallmarks of hyperexcitability.
Brief early treatment that changes a lifetime course
The researchers then asked whether dialing down the mutant receptor or restoring the REST brake only during this neonatal window could change the disease’s long-term course. They delivered antisense oligonucleotides—short strands of modified genetic material—into the fluid surrounding the brain and spinal cord of one-day-old SBMA mice. One type of oligonucleotide transiently lowered both the mutant and normal androgen receptor in the central nervous system. A second type skewed REST splicing away from REST4 and back toward full-length REST, thereby reining in synaptic genes. Remarkably, even though these treatments were given just once and their direct molecular effects faded within a few weeks, the mice lived longer, walked better on a rotating rod, and maintained stronger grip later in life. Their motor neurons and muscle fibers were less shrunken, and early markers of neuronal overactivity and later surges of stress-related neuropeptides were blunted.
How early hormones and gene tuning shape vulnerability
The work also highlights the special vulnerability of motor neurons to the brief burst of testosterone that occurs soon after birth. When newborn SBMA mice were given extra testosterone, their later weakness and weight loss worsened, and gene programs linked to healthy motor neuron maturation were further disturbed. Normal mice did not show this damage, emphasizing that it is the combination of the mutant receptor and the hormonal surge that is harmful. Together, the findings suggest that in SBMA, too many excitatory synapses and overly excitable motor neurons early in life slowly drive the system toward failure, even though obvious symptoms do not emerge until middle age.
What this means for people living with SBMA
To a non-specialist, the key message is that SBMA may be, in part, a disease of mistimed and miswired synapses during the first days after birth. A faulty hormone sensor nudges developing motor neurons into an overexcited state, and this early stress eventually contributes to their degeneration years later. The encouraging news is that carefully designed genetic medicines, delivered during these critical windows, can reset the balance of signals in motor neurons, calm their overactivity, and meaningfully delay or reduce later nerve-cell loss in animals. While translating such early-life interventions to humans will require great care and further study, these results point toward new strategies that target the roots of SBMA long before weakness appears.
Citation: Hirunagi, T., Sahashi, K., Iida, M. et al. Restoring early postnatal synaptic dysregulation rescues motor neuron degeneration in a mouse model of Spinal and Bulbar Muscular Atrophy. Nat Commun 17, 2412 (2026). https://doi.org/10.1038/s41467-026-70244-2
Keywords: spinal and bulbar muscular atrophy, motor neuron hyperexcitability, androgen receptor, REST synaptic regulation, antisense oligonucleotide therapy