Clear Sky Science · en
Integrated electrochemical porous solid electrolyte reactor and packed bed reactor for efficient synthesis of nylon-6 precursor
Cleaner Building Blocks for Everyday Plastics
Nylon-6 is woven into countless products, from carpets and clothing to car parts. Yet the chemical steps used to make its key ingredient, cyclohexanone oxime, still depend on toxic materials and energy-hungry processes. This study introduces a cleaner, continuous method that makes the same crucial molecule using only air, water, electricity, and ammonia—offering a way to shrink the environmental footprint of a major plastics industry.
Why Today’s Nylon Pathway Is a Problem
Conventional factories make cyclohexanone oxime by reacting cyclohexanone with hydroxylamine, a compound that is both unstable and potentially explosive. To keep it under control, industry adds strong acids and then neutralizes them later, creating large amounts of salty waste and driving up costs. Alternative “greener” routes have tried to generate hydroxylamine directly from nitrogen oxides in an electrochemical cell, but these methods tend to over‑reduce the nitrogen to ordinary ammonia, wasting energy and limiting how much useful product can be made per unit of electricity.
A Two‑Step, Plug‑Together Reactor Design
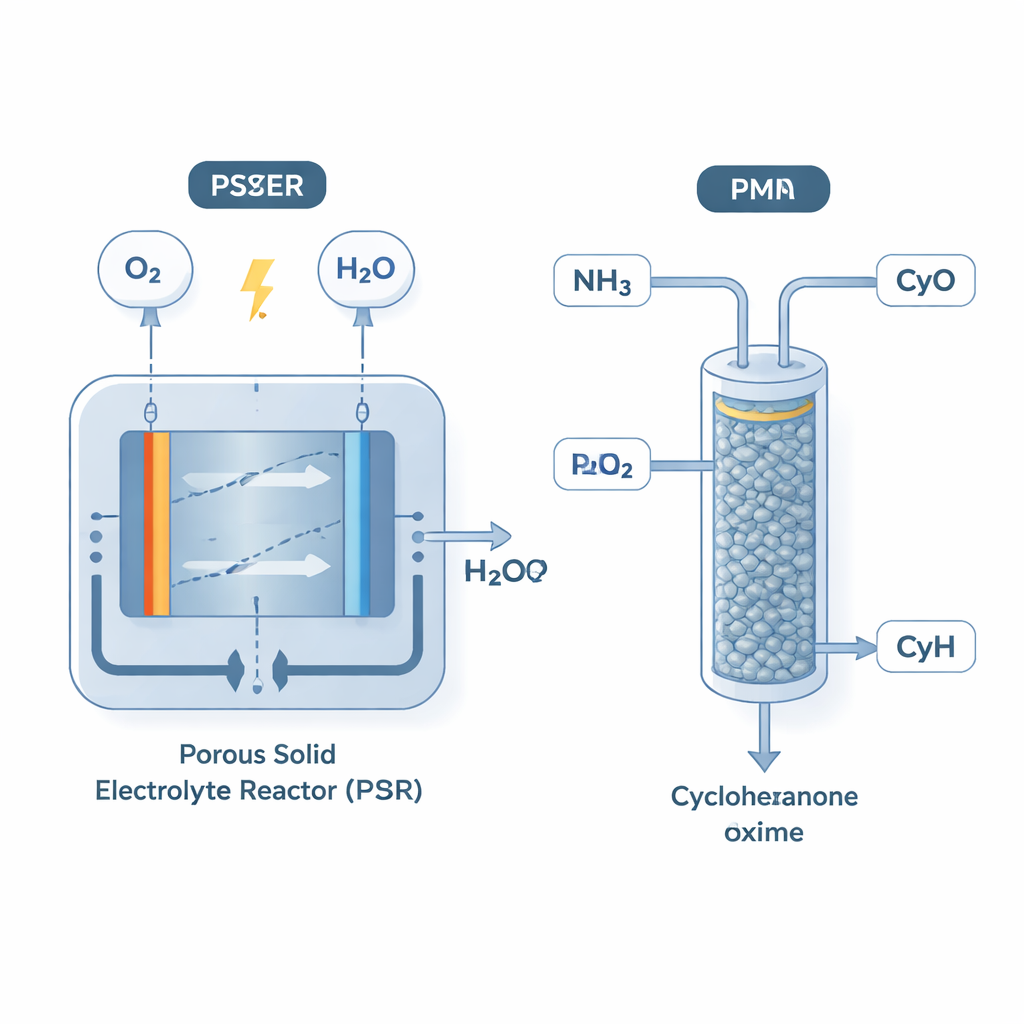
The authors tackle the problem by separating it into two tightly linked units. First, they use a porous solid electrolyte reactor (PSER) to turn oxygen from air and water into hydrogen peroxide using electricity. This device is built with three chambers and special membranes so that the peroxide solution that comes out is very pure and contains almost no added salts or stabilizing chemicals. Second, they feed this freshly made peroxide together with cyclohexanone and ammonia into a packed bed reactor (PBR) filled with a commercial catalyst called TS‑1, already used in industry. Inside the packed tube, the peroxide and ammonia form hydroxylamine in place, which then immediately reacts with cyclohexanone to give the desired oxime.
High Yields at Industrially Relevant Speeds
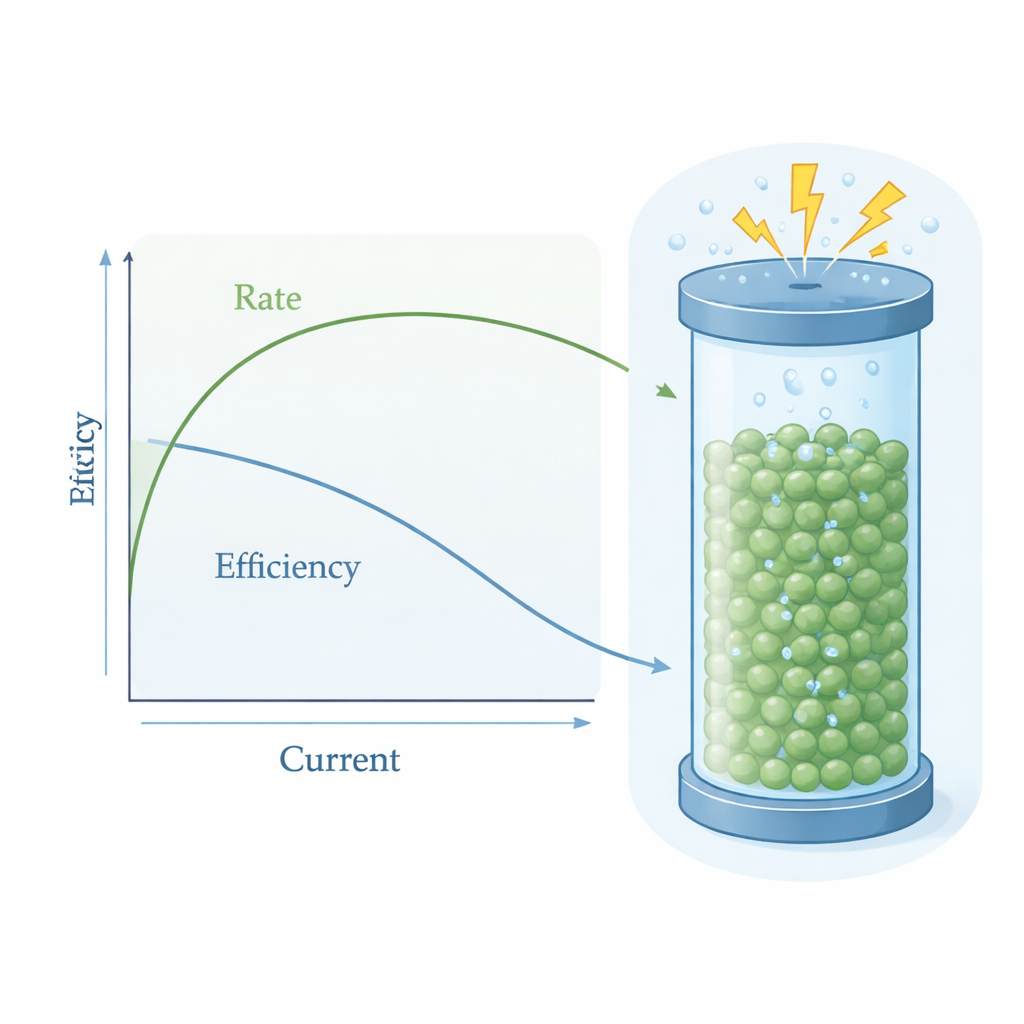
Working first on each unit separately, the team adjusted temperatures, catalyst amounts, and solvent choices in the packed bed reactor to maximize how much cyclohexanone oxime it could produce from a given feed. They found that operating around 80 °C with carefully chosen concentrations of reactants gave high conversion of cyclohexanone and very few side products. On the electrochemical side, they scaled the PSER up to 25 square centimeters—six times larger than their earlier design—and showed that it could continuously generate hydrogen peroxide at tunable concentrations simply by changing the electrical current, while keeping electrical efficiency high.
Outperforming Conventional Peroxide and Cutting Costs
When the two units were connected, the system produced cyclohexanone oxime continuously with striking performance. At moderate current, the process converted more than 96% of the starting cyclohexanone, achieved over 97% selectivity for the desired oxime, and used over 96% of the generated hydrogen peroxide—better than when using commercial peroxide stabilized with additives. At higher, industrially interesting currents, the production rate climbed to 28.3 millimoles per hour on the lab‑scale device, far above previous approaches, even though some efficiency was lost because concentrated peroxide decomposes into oxygen bubbles. A techno‑economic analysis suggests that, under reasonable electricity prices, this approach could make the nylon‑6 precursor at roughly one‑quarter of its current market price, mainly because it uses inexpensive ammonia and on‑site peroxide instead of costly reagents and complex separation steps.
Beyond One Molecule and Toward Greener Plants
To show that their setup is more than a one‑trick tool, the researchers also applied the same PSER‑PBR combination to a range of other ketones and demonstrated that it could form several different oximes with high selectivity. Long‑term trials over many hours showed stable operation and steady product quality, and the modular packed bed design would allow industrial operators to swap out and regenerate catalyst cartridges without shutting down the entire line. For a layperson, the takeaway is simple: by tightly coupling a clean, electricity‑driven oxidant source to a robust chemical reactor, this work points to a future in which key plastic ingredients are made more safely, more efficiently, and with far less waste.
Citation: Zhang, SK., Feng, Y., Hao, S. et al. Integrated electrochemical porous solid electrolyte reactor and packed bed reactor for efficient synthesis of nylon-6 precursor. Nat Commun 17, 2163 (2026). https://doi.org/10.1038/s41467-026-70236-2
Keywords: nylon-6, green chemistry, electrochemical reactor, hydrogen peroxide, cyclohexanone oxime