Clear Sky Science · en
Pathway selection between click and acyl transfer reactions driven by aminoacyl phosphates
Why Timing Matters in Everyday Chemistry
Inside every cell, important molecules are built and modified in a very precise order, much like steps in a recipe. That timing decides whether a protein turns on, turns off, or even changes shape. Chemists would like to mimic this kind of built‑in schedule using simple, non‑living molecules in water, without relying on enzymes or outside triggers like light or pH jumps. This paper describes a man‑made reaction system that does exactly that: it uses the structure of small peptides and a popular bond‑forming reaction from "click chemistry" to decide which chemical step happens first, and which must wait its turn.
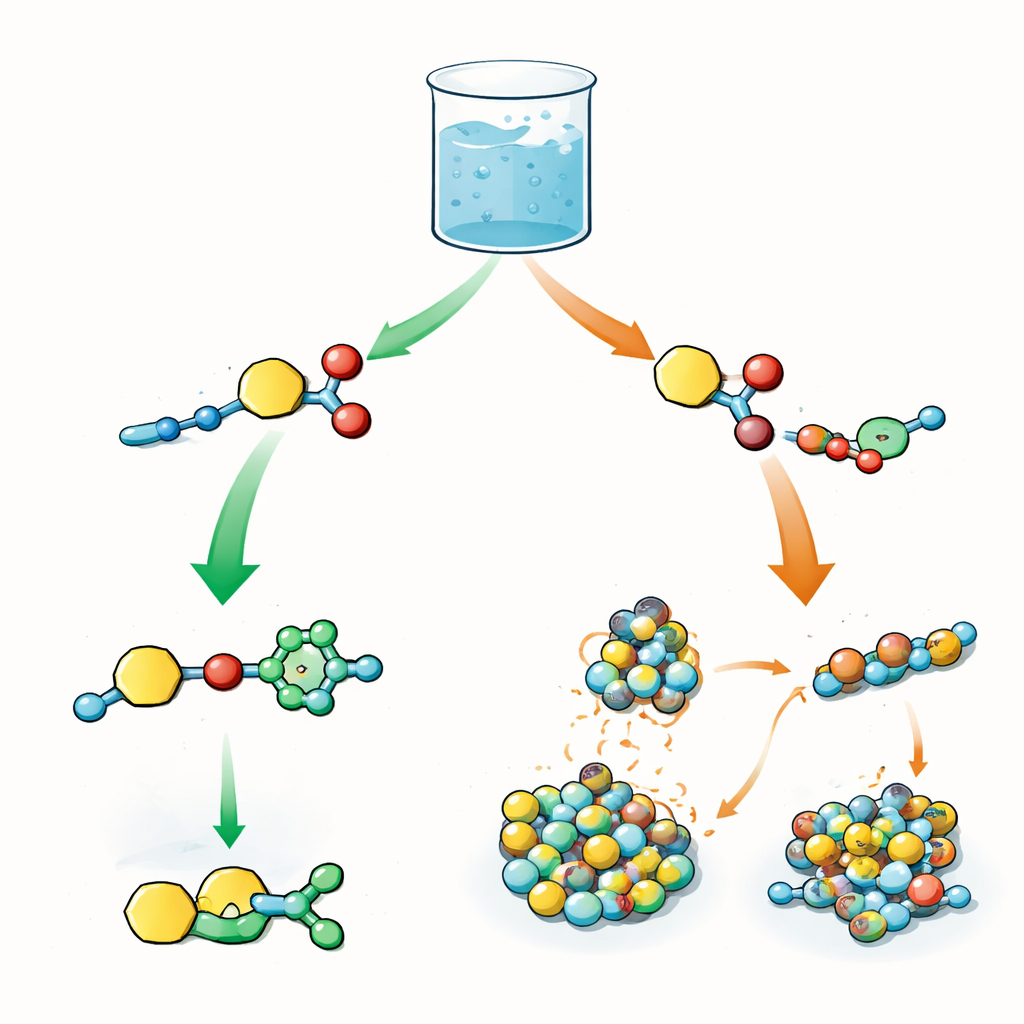
Two Rival Reactions in One Pot
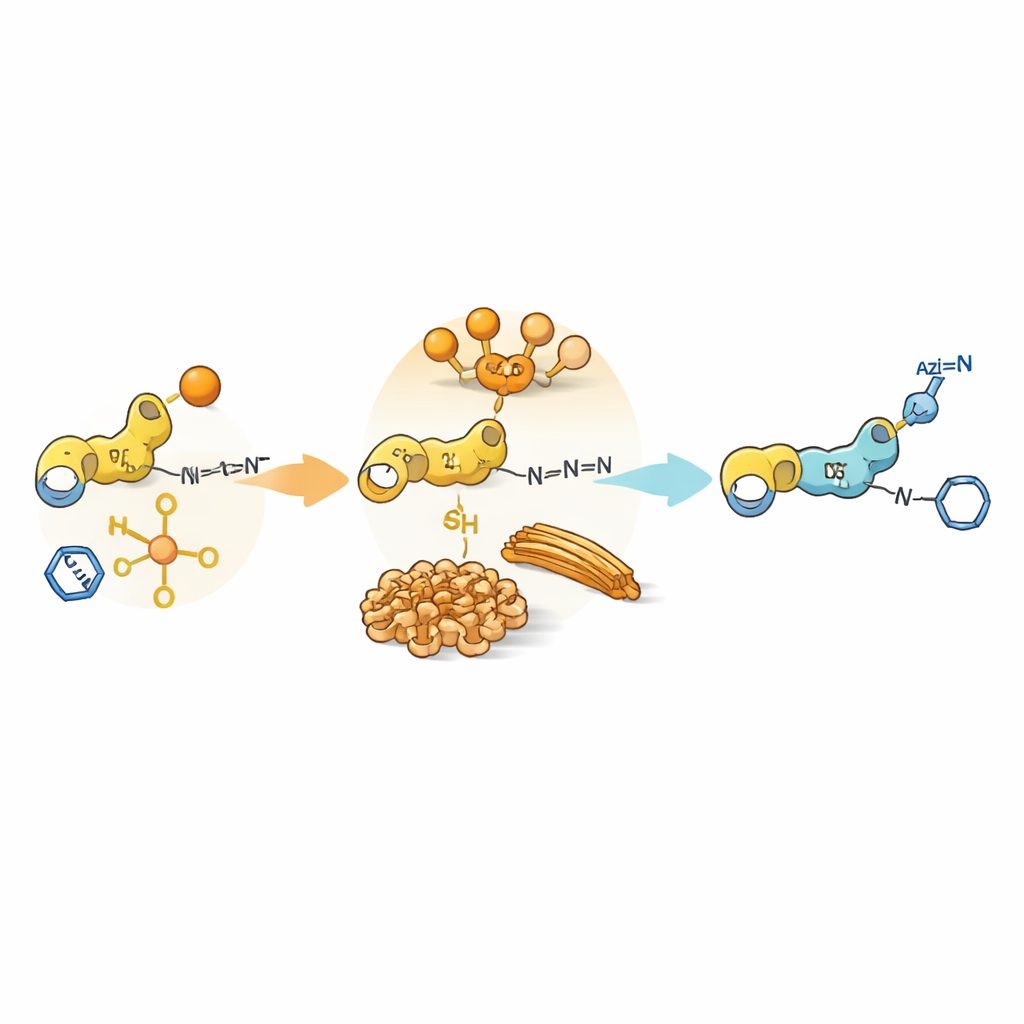
The authors design a central building block that carries two reactive features: one that can pass on an "acyl" group (a small chemical fragment often used to modify proteins) and another that can undergo a click reaction called a copper‑catalyzed azide–alkyne cycloaddition. In the same beaker, this building block is mixed with azides and with short peptides that act as nucleophiles, meaning they grab the acyl group. In biology, the order of such steps—activation, transfer, and later modifications—controls how proteins behave. Here, the question is similar: when both reactions are possible at once, does the system choose the click reaction first, the acyl transfer first, or some mixture of both?
Slow Oxygen, Fast Sulfur
The team first studies peptides containing tyrosine, an amino acid with an oxygen‑based side chain. Under mildly basic conditions, the click reaction races ahead: the alkyne and azide rapidly join, while acyl transfer to the tyrosine side chain is sluggish and barely builds up detectable intermediates. Several click‑derived products coexist, and there is little clear timing gap between steps. When the researchers switch to cysteine, an amino acid with a sulfur‑based side chain, the story flips. Cysteine quickly forms thioesters—sulfur‑linked acyl products—that appear in high yield long before any click products. Only after many hours do click products begin to emerge. This shift arises because the sulfur not only reacts more eagerly but also latches onto copper ions, tying up the catalyst and temporarily "pausing" the click path.
Self‑Assembly as a Chemical Gate
Beyond simple reactivity, the peptides themselves can clump together into larger structures, forming soft gels or fibers in water. These assemblies create tiny domains where some partners are concentrated and others are excluded. For certain tyrosine‑ or cysteine‑containing dipeptides that carry extra aromatic groups, the intermediates self‑assemble into fibrils or dense particles. Inside these packed regions, acyl transfer is favored because nucleophile and acyl donor sit side by side, while the azide and copper catalyst remain mostly in the surrounding solution. As a result, even though the click reaction is intrinsically fast in free solution, it is slowed by the need to reach the buried reactive sites. This "phase mismatch" between where the catalyst lives and where the substrates are located stretches out the lifetime of acylated intermediates and delays the onset of click chemistry.
Programming Cascades and Choices
To test how far they can push this built‑in scheduling, the authors design more complex situations. In one case, a single peptide contains both cysteine and tyrosine, offering two different acyl transfer sites. The reaction proceeds as a three‑step cascade: first a thioester forms on cysteine, then a second acyl group moves onto tyrosine, and only after these steps do click products appear. In another set of experiments, two different azides—one flexible and aliphatic, the other rigid and aromatic—compete for the same reactive center. Surprisingly, the system consistently prefers the aliphatic azide, revealing that even the shape and electronic character of the azide can bias which products dominate, all while the acyl‑transfer timing remains under peptide control.
What This Means for Future Smart Materials
In everyday language, this work shows how you can pack multiple, potentially competing reactions into the same simple water‑based mixture and still have them fire in a set order—without enzymes, pumps, or external switches. By choosing whether a peptide carries oxygen or sulfur, by tuning its tendency to self‑assemble, and by adjusting the nature of the azide partner, the authors encode a kind of internal clock into a small chemical network. Early, reversible acyl transfers create short‑lived intermediates that shape when and how the robust, nearly irreversible click step occurs. Such programmed sequences could become a foundation for smart materials and synthetic reaction networks that “decide” their own pathways over time, much like the carefully timed chemistry at work in living cells.
Citation: Bhattacharjee, D., Sharma, A., Dai, K. et al. Pathway selection between click and acyl transfer reactions driven by aminoacyl phosphates. Nat Commun 17, 2407 (2026). https://doi.org/10.1038/s41467-026-70199-4
Keywords: click chemistry, acyl transfer, supramolecular assembly, reaction networks, peptide chemistry