Clear Sky Science · en
Adaptations in Plasmodium tubulin determine distinct microtubule architectures, mechanics and drug susceptibility
Why malaria’s skeleton matters
Malaria parasites survive inside human blood cells, yet they must also squeeze through mosquitoes and different tissues. To do this, they rely on an internal scaffolding made of microscopic tubes called microtubules. This study asks a simple but powerful question: how can the parasite’s microtubules be so specialized and sturdy when their basic building blocks look so similar to those in our own cells? The answer reveals not only how the parasite stays in shape, but also how we might design drugs that target its skeleton without harming ours. 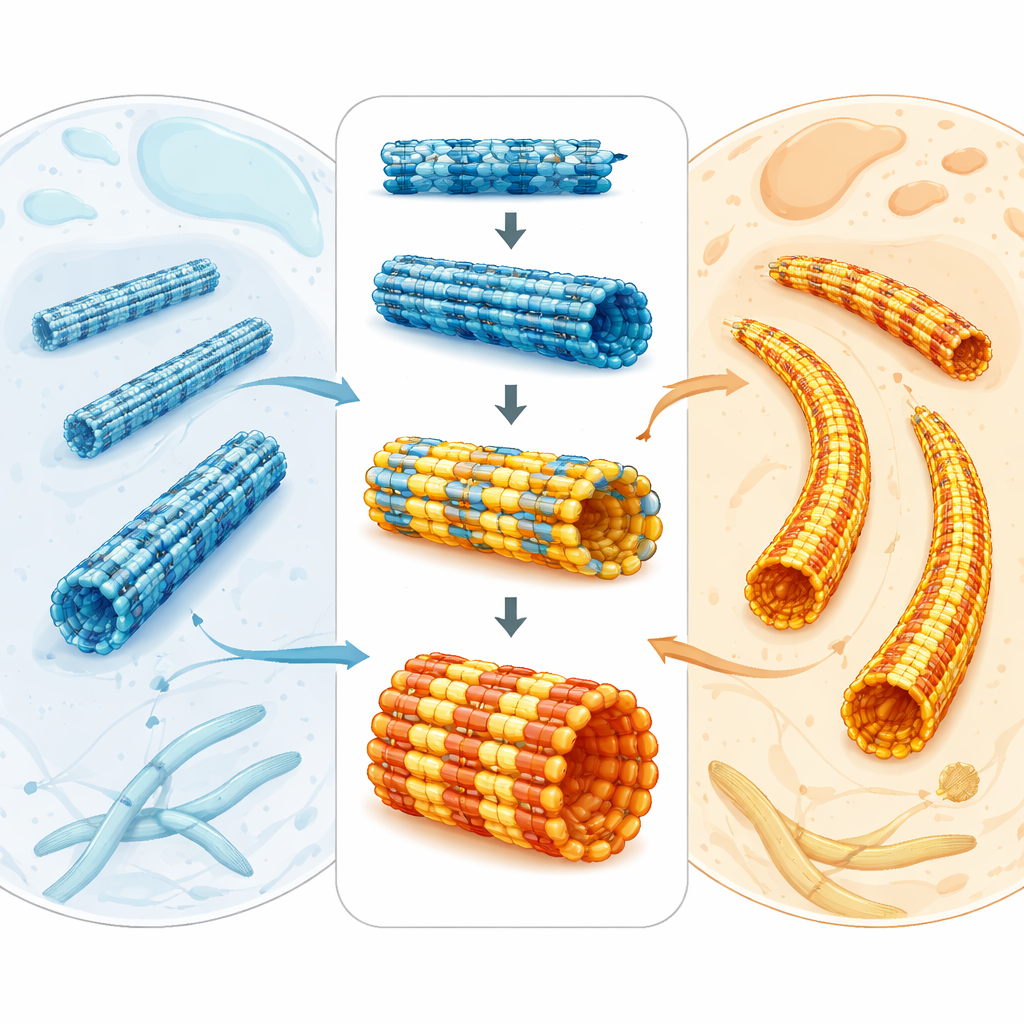
The tiny tubes that shape a deadly parasite
Microtubules are hollow protein tubes that help cells divide, move, and maintain their form. They are built from paired protein units called tubulin. Across animals, plants, and parasites, tubulin is surprisingly similar, which makes it hard to explain why microtubules can look and behave so differently in different organisms. In the malaria parasite Plasmodium falciparum, microtubules form diverse arrays: some are dynamic and help during cell division, while others are long, stiff supports under the parasite’s outer membrane. Because these structures are essential for parasite growth and transmission, they are attractive targets for antimalarial drugs—especially as resistance to current treatments continues to rise.
Seeing parasite tubulin in atomic detail
The researchers purified tubulin directly from P. falciparum parasites and used high-resolution cryo–electron microscopy to see how it assembles into microtubules. They compared these structures to the well-studied microtubules from mammalian brain. At first glance, the parasite tubulin looks almost identical to the human version and adopts the same overall fold. But careful comparison uncovered clusters of small sequence changes around key pockets where energy-carrying molecules (GTP and GDP) bind and where certain drugs interact. These subtle shifts alter how nearby helices and loops are positioned, especially on the surface of the tubulin dimer where other proteins and drugs latch on. The work suggests that, while the overall blueprint is conserved, local tweaks in the parasite subtly reshape these important sites.
Stiffer tubes from stronger side‑to‑side links
Microtubules are polymers: many tubulin dimers stack end-to-end into protofilaments, and several protofilaments line up side-by-side to form the tube wall. The team found that in parasite microtubules, the longitudinal contacts along each protofilament look very similar to those in brain microtubules. The difference shows up in the side-to-side, or lateral, contacts between neighboring protofilaments. Although the contact area is actually a bit smaller in the parasite, simulations reveal that these lateral connections are collectively stronger, making the lattice stiffer. Computer modeling of patches of the microtubule wall showed that parasite tubulin bends and twists less than brain tubulin. Electron tomography of real microtubule ends confirmed that parasite microtubules have shorter, less splayed protofilament curls, a hallmark of a sturdier tube. 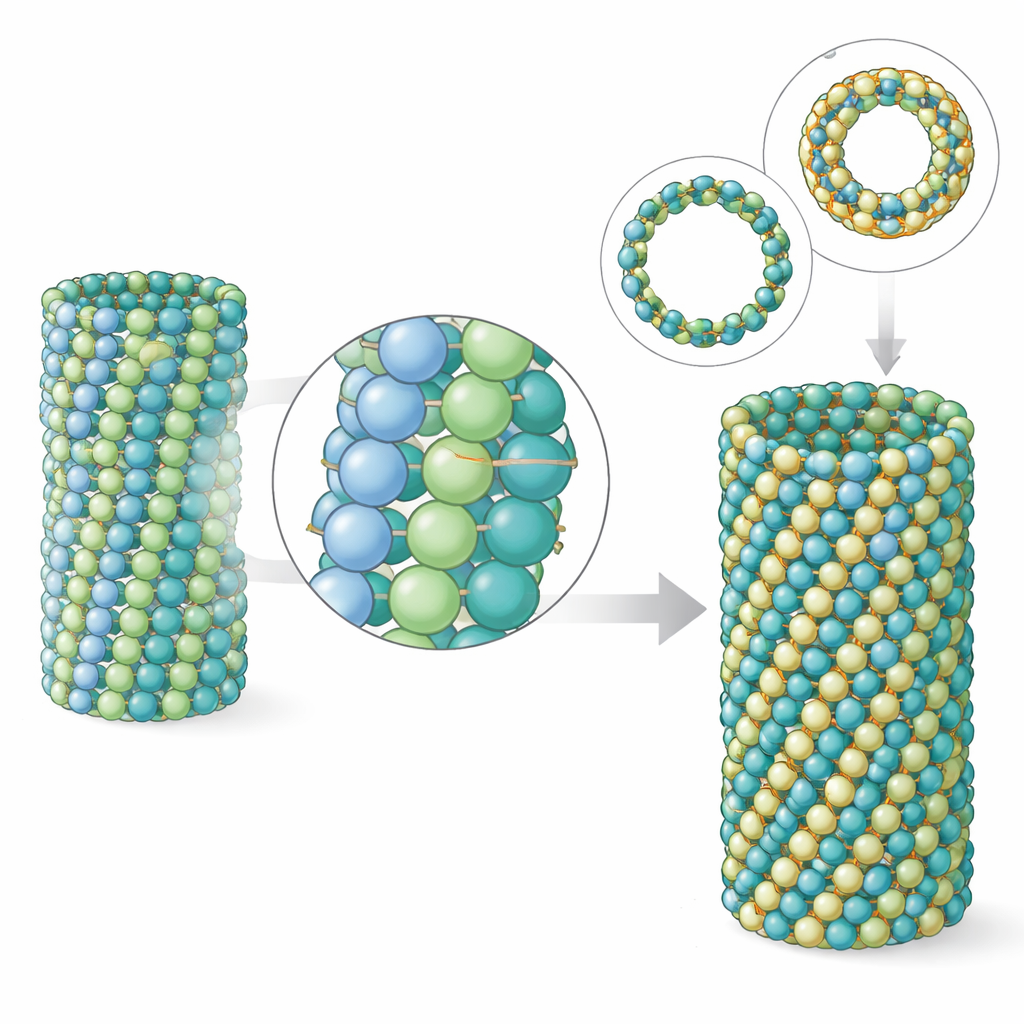
Unusual tube sizes that match the parasite in action
Another striking feature of P. falciparum is that, in some life stages, its microtubules contain more building blocks around their circumference than the usual 13 found in most animal cells—often 15 or even 17 rows of protofilaments. The team recreated this behavior in the test tube by changing the nucleotide used during assembly. With a GTP-like molecule that stabilizes the growing state, parasite tubulin naturally favored 15-protofilament microtubules. The authors solved the structure of these thicker tubes and showed how they accommodate the extra protofilaments with a slight skew of the lattice, yet remain nearly perfectly cylindrical. When they compared these lab-made 15-protofilament tubes to those seen inside parasite cells, the match in shape and dimensions was remarkably close, indicating that intrinsic properties of the tubulin itself—rather than a crowd of helper proteins—are enough to generate the parasite’s distinctive architectures.
Clues for future, parasite‑specific drugs
The study’s conclusion is that evolution has tuned the parasite’s tubulin through small, scattered sequence changes that collectively alter microtubule mechanics, architecture, and drug sensitivity. These tweaks make the parasite’s microtubules stiffer and able to form nonstandard tube sizes that suit its demanding life cycle, while still keeping the basic tubulin design recognizable. Importantly for medicine, some drug-binding pockets appear nearly identical between parasite and host (as for the cancer drug paclitaxel), suggesting they are poor targets for selective antimalarials. Others, such as the region thought to bind the herbicide-like compound oryzalin, show both sequence shifts and subtle structural displacements that could explain why parasite microtubules are more vulnerable there than human ones. Mapping these nuanced differences provides a roadmap for designing future drugs that disrupt the malaria parasite’s skeleton while leaving our own cells largely untouched.
Citation: Bangera, M., Wu, J., Beckett, D. et al. Adaptations in Plasmodium tubulin determine distinct microtubule architectures, mechanics and drug susceptibility. Nat Commun 17, 2275 (2026). https://doi.org/10.1038/s41467-026-70181-0
Keywords: malaria parasite, microtubules, tubulin structure, drug targeting, cryo-electron microscopy