Clear Sky Science · en
Bio-inspired asymmetric Zn-N2O2 single-atom catalysts via natural skeleton for efficient N-alkylation of nitroarenes with alcohols
Turning Waste Shells into Useful Chemistry Tools
Many important medicines, crop protectants, and specialty materials contain a simple structural feature: a nitrogen atom linked to a small carbon chain. Making these “N‑alkylated” molecules on an industrial scale usually requires harsh chemicals, high temperatures, and expensive metals. This study presents a greener alternative inspired by nature itself, using zinc atoms anchored on a support made from chitosan, a material derived from discarded seafood shells, to run these reactions more efficiently and with less waste.
Why Greener Nitrogen Chemistry Matters
Conventional ways of attaching carbon chains to nitrogen rely on reactive halogenated chemicals and strong additives that generate large amounts of by‑products. They also often need rare and costly precious metals like palladium or platinum as catalysts. In contrast, a newer approach called the “borrowing hydrogen” strategy uses common alcohols as both the source of the carbon chain and of hydrogen, with water as the main by‑product. While this concept is attractive, existing non‑noble metal catalysts generally work only under harsh conditions or with limited types of starting materials. The challenge has been to design a low‑cost, recyclable catalyst that can drive this reaction efficiently under milder conditions.
Borrowing Hydrogen with Single Zinc Atoms
The researchers tackled this problem using single‑atom catalysis, in which individual metal atoms rather than larger particles are anchored on a solid surface. They dissolved chitosan, a biodegradable polymer rich in oxygen and nitrogen groups, and turned it into porous three‑dimensional microspheres using a sol‑gel process. Zinc ions were then introduced and gently heated, locking isolated zinc atoms into the chitosan framework in an arrangement the authors describe as Zn‑N2O2: each zinc atom is surrounded by two nitrogen and two oxygen atoms from the support. This bio‑inspired local environment mimics the way metals are coordinated in many natural enzymes and maximizes the number of active sites available for the reaction.
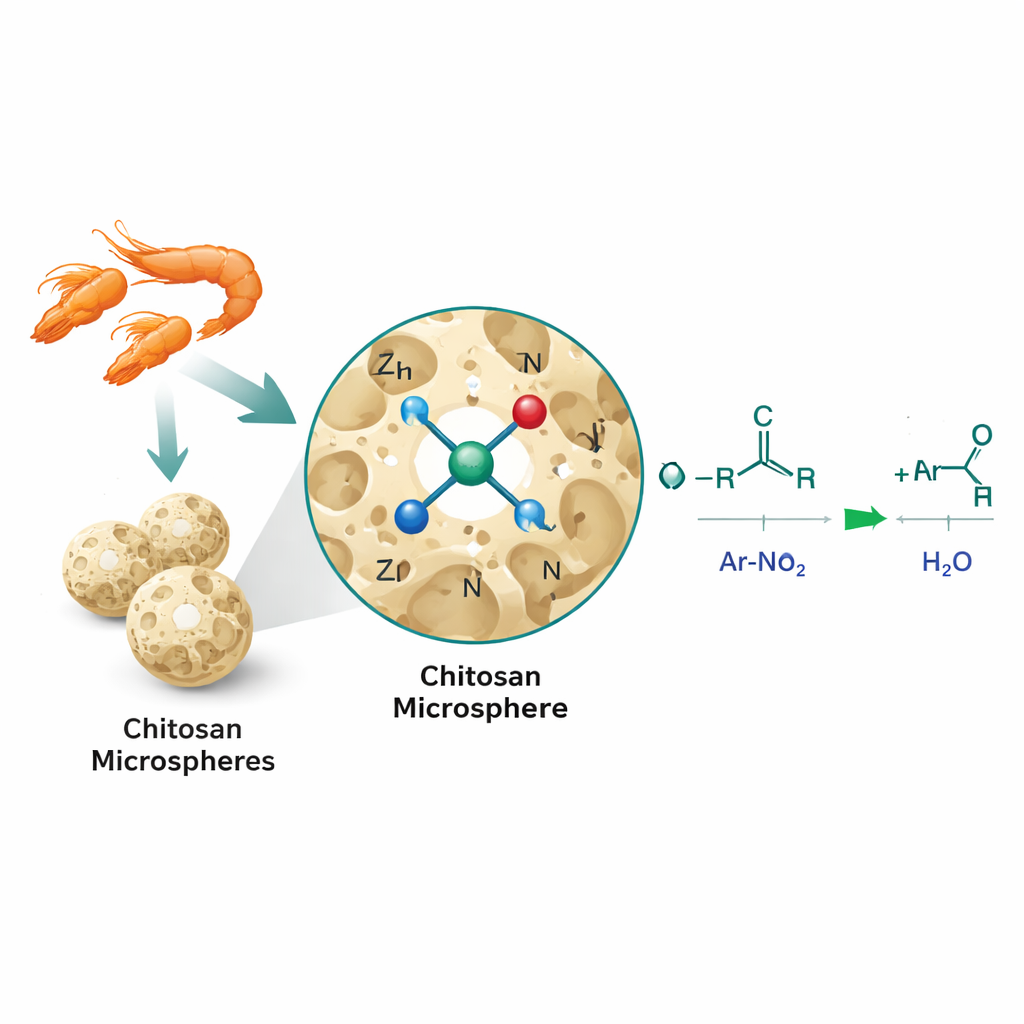
Proving the Structure and Performance
To confirm that the zinc truly existed as single atoms rather than larger particles, the team combined several advanced imaging and spectroscopy methods. Electron microscopes showed porous chitosan spheres but no visible zinc clusters, while high‑angle dark‑field images revealed tiny bright spots corresponding to individual zinc atoms scattered uniformly across the surface. X‑ray based techniques further indicated that zinc was bonded mainly to nitrogen and oxygen, with no detectable zinc–zinc bonds, supporting the single‑atom picture. These structural features translated into striking performance: in a model reaction between nitrobenzene and benzyl alcohol, the zinc‑on‑chitosan catalyst (Zn/CS) delivered very high yields with exceptionally low metal loading and outperformed commercial zinc on carbon, zinc nanoparticles, and simple dissolved zinc salts. It also worked for 56 different combinations of nitro compounds and alcohols, including complex building blocks used in drug discovery, and could be reused at least five times with minimal loss of activity.
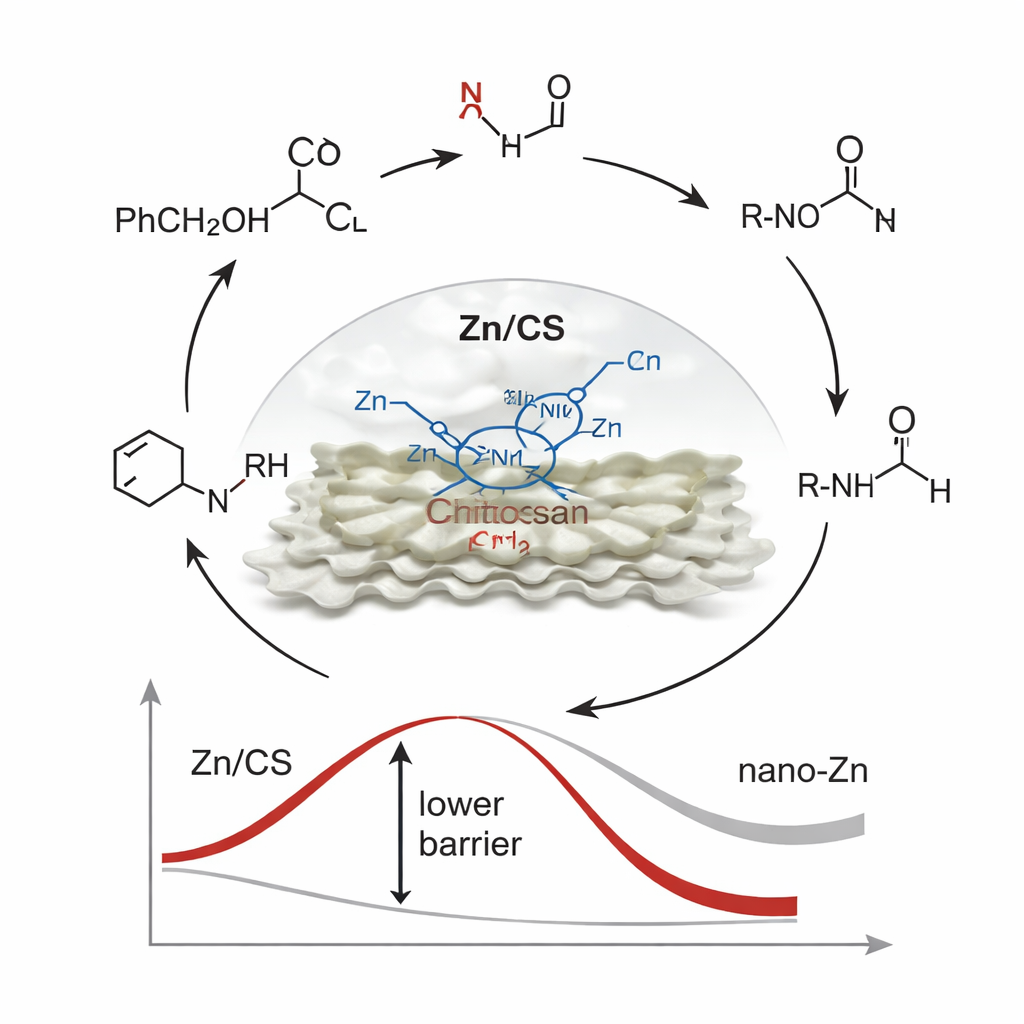
How the Catalyst Actually Works
At the molecular level, the borrowing‑hydrogen process follows a sequence of steps. First, the alcohol is temporarily oxidized to an aldehyde while transferring hydrogen to the zinc site. That hydrogen is then used to reduce the nitro group to an amine. The aldehyde and the new amine combine to form an imine, which is finally reduced to the desired N‑alkylated product, closing the cycle. Time‑resolved nuclear magnetic resonance, gas chromatography, and carefully designed control reactions confirmed the presence of key intermediates along this route. Deuterium‑labeling experiments, in which selected hydrogen atoms were replaced by a heavier isotope, showed that the zinc–hydrogen species plays the dominant role in the reduction steps. Computer simulations helped explain why this particular catalyst is so effective: the asymmetric Zn‑N2O2 environment pulls electron density away from the zinc atom, making it slightly electron‑poor. That electronic tuning makes zinc better at binding and activating the alcohol‑derived intermediate, lowering the energy barrier for the hardest step—the initial removal of hydrogen from the alcohol.
From Sea Waste to Smart Catalysts
In accessible terms, this work shows how individual zinc atoms, precisely arranged on a natural, sponge‑like support made from seafood waste, can outperform many traditional metal catalysts in an important class of chemical reactions. By using alcohols instead of toxic reagents and generating mainly water as a by‑product, the system offers a cleaner and potentially cheaper way to make a wide range of nitrogen‑containing molecules, including pharmaceutical motifs. The combination of a renewable biopolymer support with finely tuned single‑atom sites illustrates a promising path toward more sustainable chemistry, where industrially useful reactions borrow their design principles—and some of their raw materials—from nature.
Citation: Huang, Y., Li, Y., Yin, X. et al. Bio-inspired asymmetric Zn-N2O2 single-atom catalysts via natural skeleton for efficient N-alkylation of nitroarenes with alcohols. Nat Commun 17, 2242 (2026). https://doi.org/10.1038/s41467-026-70172-1
Keywords: single-atom catalysis, green chemistry, borrowing hydrogen, chitosan, zinc catalyst