Clear Sky Science · en
Covalent warhead assembly in fostriecin biosynthesis involves malonylation-lactonisation by a bifunctional thioesterase and enzymatic demalonylation
How nature builds a tiny chemical warhead
Cancer drugs and antibiotics often depend on a few atoms arranged just so—a tiny “warhead” that locks onto a protein and shuts it down. The natural compound fostriecin is one such molecule, with powerful activity against enzymes involved in cell growth. But its complex structure makes it hard to manufacture or modify in the lab. This study uncovers, step by step, how bacteria assemble fostriecin’s crucial warhead, revealing enzyme tricks that chemists can now borrow to design new medicines.
The special hook in potent natural drugs
Many promising drugs from soil bacteria belong to a family called polyketides. Within this family, a subgroup carries a reactive ring, known to chemists as an α,β-unsaturated δ-lactone, that acts like a molecular fishhook. It can form a permanent bond with certain amino acids in proteins, turning off key biological switches. In fostriecin and related molecules, this hook is paired with a phosphate group that helps them home in on specific enzymes controlling cell division. These features make them attractive anticancer leads, but the same reactivity and structural complexity make them tricky to synthesize and unstable during storage.
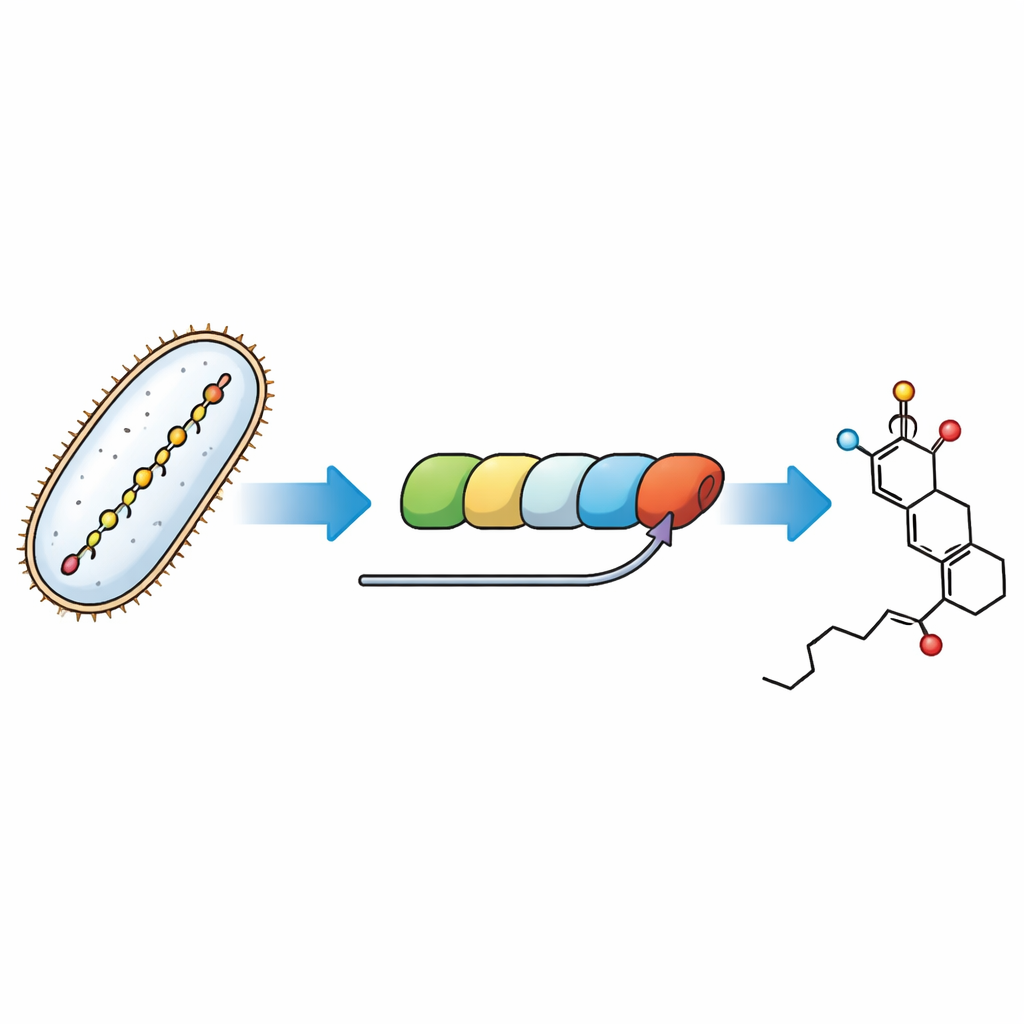
Tracing the final steps of a molecular assembly line
Bacteria build fostriecin using a massive modular protein machine called a polyketide synthase—an assembly line where each station lengthens and shapes a growing carbon chain. The puzzle addressed here is how the last module of this machine forges the warhead ring and installs a temporary “handle” called a malonyl group that later must be removed. By recreating the pathway in test tubes with purified enzymes and carefully designed small-molecule stand-ins for the natural intermediates, the researchers could watch individual steps and assign them to specific parts of the assembly line.
A multitasking enzyme with an unexpected talent
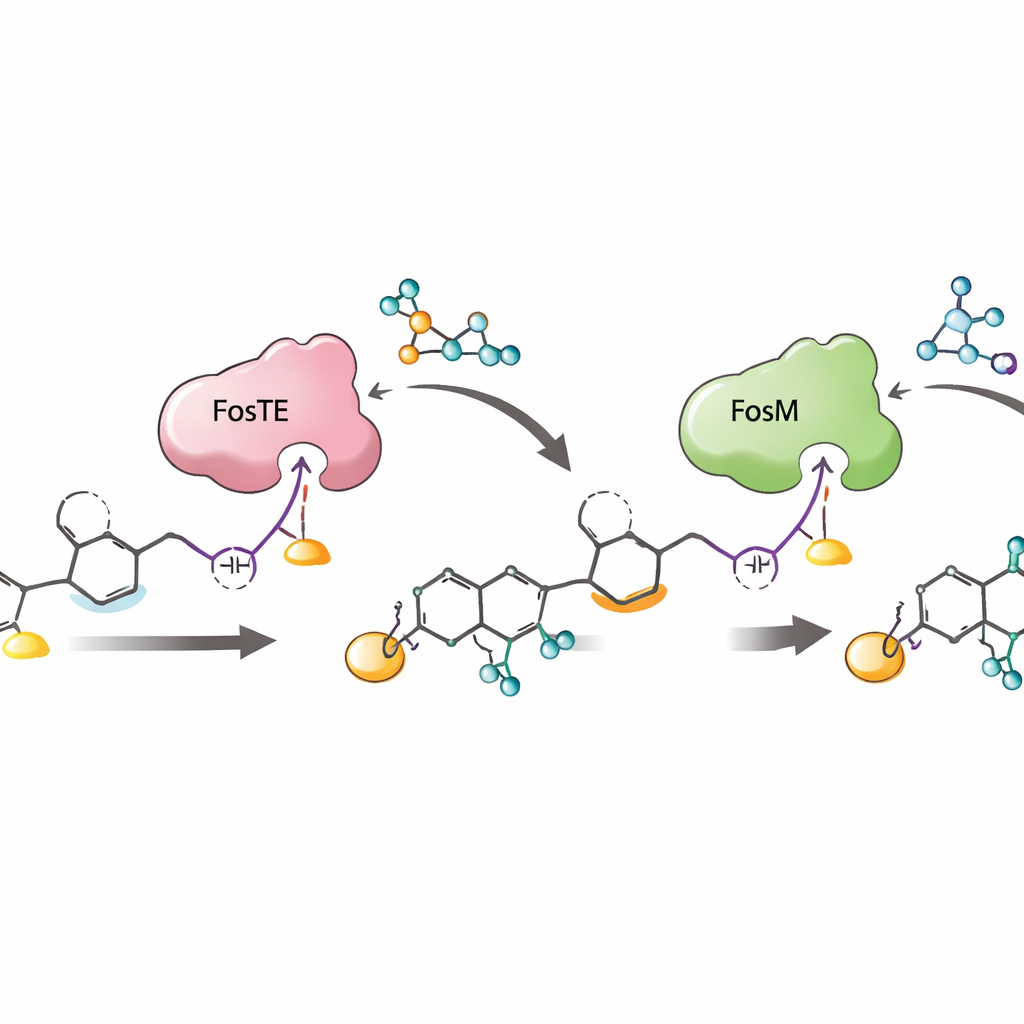
The team discovered that the final station of the assembly line, a domain named FosTE, behaves in a way not seen before. Typical versions of this domain simply cut finished products off the assembly line. FosTE, in contrast, first grabs a malonyl fragment from a common cellular building block and transfers it onto a precise oxygen site on the growing chain, and then helps the chain curl around to close into the warhead ring. Mutating a single reactive serine amino acid in FosTE shuts down both activities. Structural modeling points to two positively charged arginine residues in the active site that cradle the malonyl fragment and position it for transfer—functions normally handled by a different type of enzyme. Changing these arginines to neutral residues erases the malonyl-transfer step while leaving basic ring formation largely intact, confirming their critical role.
Locking in the warhead and preventing waste
Once the ring is formed and carries its temporary malonyl group, another enzyme, FosM, must remove this group to unmask the fully armed warhead. The study shows that FosM works efficiently only after yet another enzyme, a broad-specificity kinase called FosH, adds a phosphate to a nearby position on the molecule. When the researchers supplied the malonylated ring to FosM alone, only modest conversion occurred. Adding FosH first led to almost complete removal of the malonyl group and formation of the final reactive unit. Importantly, FosH can also add phosphate to related “shunt” intermediates that arise from side reactions, rescuing them so they can be processed correctly instead of being lost as dead-end products.
Why this enzymatic choreography matters
Taken together, the work reveals a tightly choreographed sequence: chain extension on the assembly line, FosTE-driven malonyl addition and ring closure, FosH phosphorylation, and finally FosM-triggered malonyl removal. This order not only constructs the delicate warhead efficiently, it also protects unstable intermediates from falling apart and maximizes production of the desired, phosphorylated form that is most active as a drug lead. By exposing how a single enzyme domain can both decorate and release a growing molecule—and how partner enzymes fine-tune and salvage reactive intermediates—the study offers a blueprint for engineering new warhead-bearing polyketides and for building shorter, more efficient synthetic routes to complex anticancer agents like fostriecin.
Citation: Nguyen, L.N.K.T., Schlotte, L., Hoffmann, J. et al. Covalent warhead assembly in fostriecin biosynthesis involves malonylation-lactonisation by a bifunctional thioesterase and enzymatic demalonylation. Nat Commun 17, 2365 (2026). https://doi.org/10.1038/s41467-026-70144-5
Keywords: fostriecin, polyketide biosynthesis, enzyme engineering, natural product warheads, chemoenzymatic synthesis