Clear Sky Science · en
Endothelial C-type natriuretic peptide/guanylyl cyclase-B signaling prevents pulmonary arterial hypertension
Why this matters for lungs and hearts
Pulmonary arterial hypertension is a rare but deadly condition in which the blood vessels that carry blood from the heart to the lungs become dangerously tight and thick. This strain can lead to right‑sided heart failure and, ultimately, death. Current drugs mainly relax these vessels but do little to stop the underlying damage to the vessel wall. The study behind this article uncovers a natural protective signal made by cells lining lung blood vessels and shows how boosting this signal could both prevent and treat the disease.
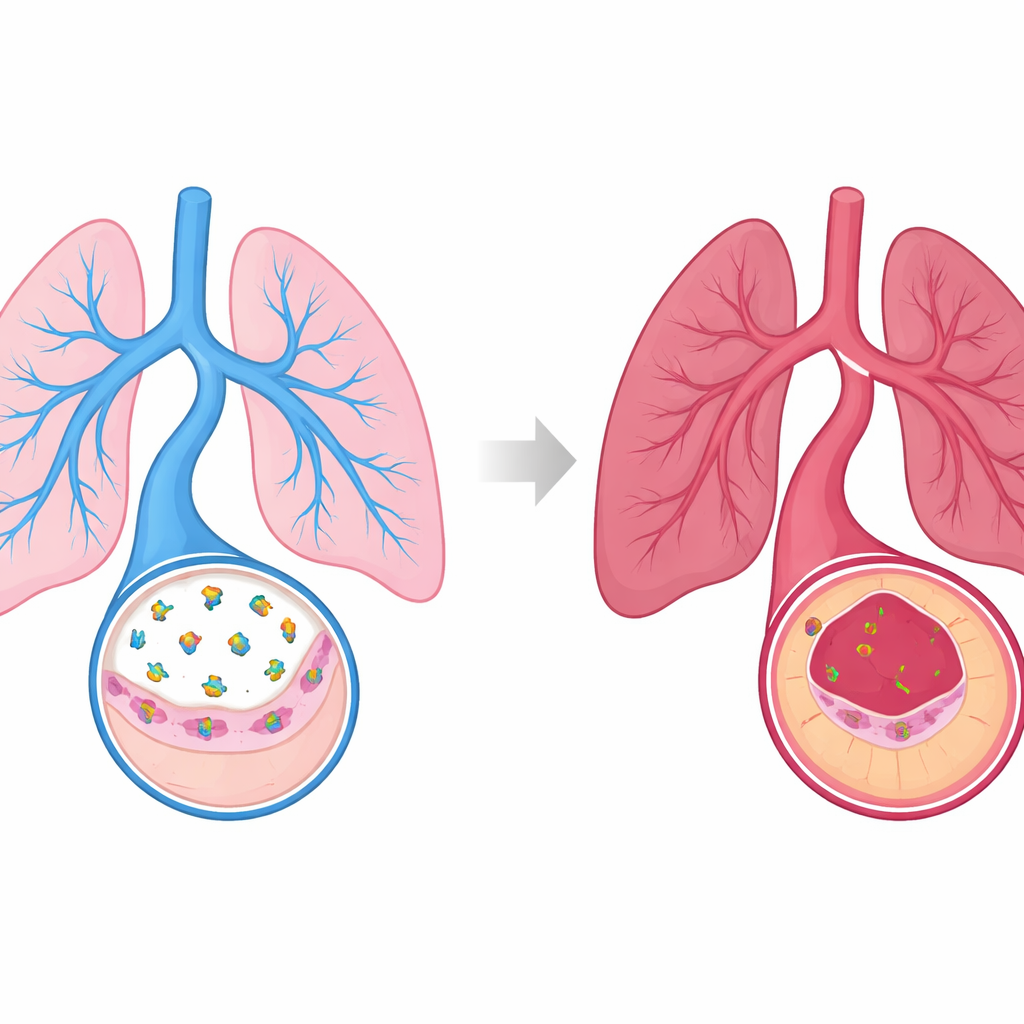
A protective message from the vessel lining
The inner lining of blood vessels is made of endothelial cells, which constantly talk to the surrounding muscle layer to keep vessels open and healthy. The authors focused on a small hormone called C‑type natriuretic peptide (CNP), which these endothelial cells release in the lungs. CNP acts locally on a receptor protein called guanylyl cyclase‑B on the same cells, triggering a chemical messenger that helps blood vessels relax and resist injury. By examining mice exposed to two different triggers of pulmonary hypertension— a toxic chemical and low‑oxygen air— the researchers found that lungs developing the disease had much lower levels of both CNP and its receptor than healthy lungs.
Switching off the signal worsens disease in animals
To test whether this loss of CNP signaling actually drives disease, the team engineered mice in which either CNP itself, or its receptor GC‑B, was removed only from endothelial cells. When these mice were exposed to the same disease triggers, their lung arteries narrowed more severely, pressure in the right side of the heart rose higher, and the heart muscle thickened more than in normal mice. In contrast, deleting the receptor only from the surrounding smooth muscle cells did not worsen the disease. This pinpointed the critical role of CNP acting on endothelial cells themselves rather than directly on the muscle layer.
How the signal keeps vessel walls calm
Looking more closely, the researchers discovered that the CNP–GC‑B pathway in endothelial cells acts as a brake on several harmful responses. Without it, these cells produced more endothelin‑1, a powerful vessel‑tightening molecule, and higher levels of inflammatory messengers such as interleukin‑6, CCL2, and TGF‑β1. In cell culture experiments, restoring CNP dampened these signals, but only if the GC‑B receptor was present. CNP also helped rebalance two competing growth‑control systems inside the cells, known as SMAD2/3 and SMAD1/5/9, which together decide whether the vessel wall remains stable or grows thicker. When CNP signaling was intact, genes linked to healthy, non‑overgrown arteries stayed active; when it was absent, the balance shifted toward overgrowth and scarring.
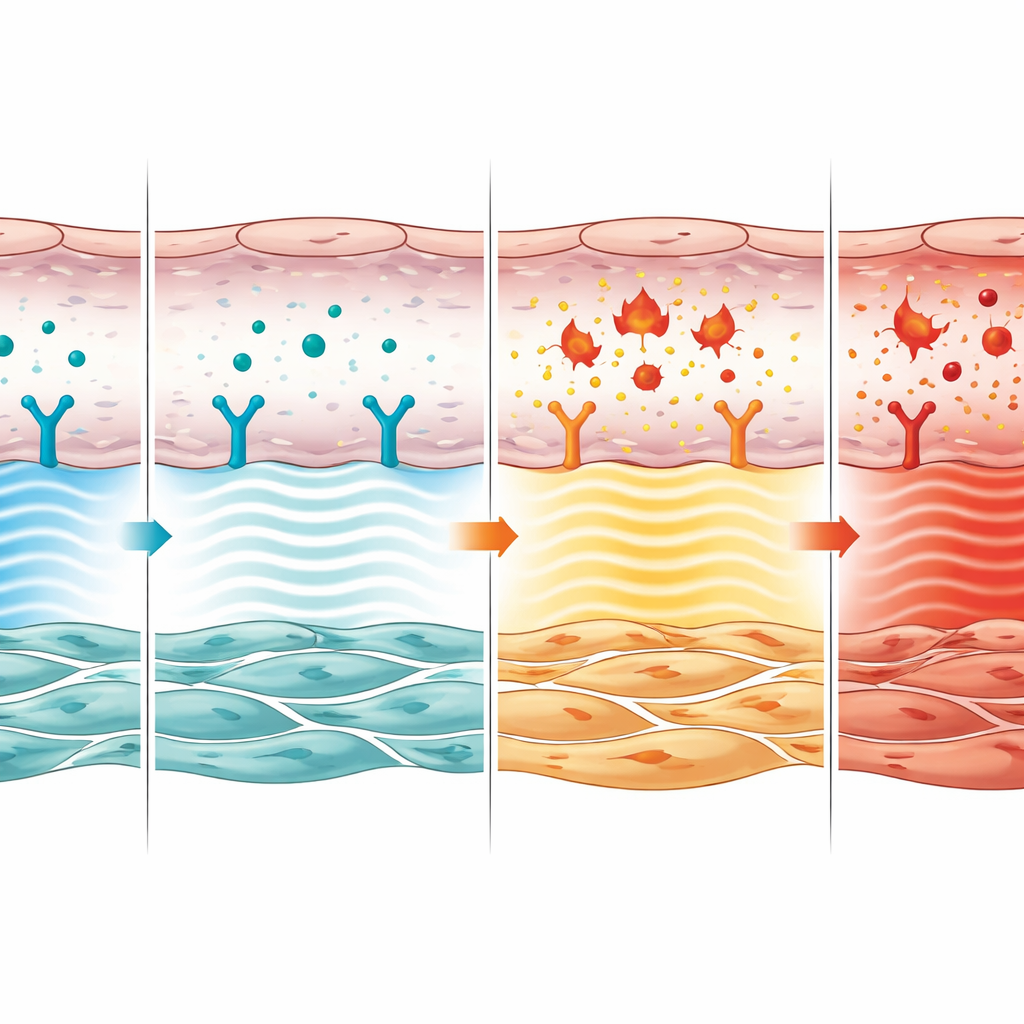
From cell talk to vessel remodeling
The team then asked how this endothelial signal affects the neighboring smooth muscle cells that actually thicken the vessel wall. They showed that chemical cues released from healthy, CNP‑responsive endothelial cells slowed the growth and migration of smooth muscle cells in the lab. When the CNP receptor was removed from endothelial cells, these cues instead encouraged smooth muscle cells to multiply and move, mimicking the vessel wall remodeling seen in disease. In mouse lungs, arteries lacking endothelial CNP signaling contained more dividing smooth muscle cells and fewer dying ones, confirming that this pathway normally restrains the overgrowth that chokes off blood flow.
Testing a drug‑like CNP in severe models
Encouraged by these findings, the authors tested a longer‑lasting form of CNP, called CNP‑53, delivered continuously by tiny pumps in rodents. In mice, giving CNP‑53 during exposure to disease triggers prevented rises in lung blood pressure and artery wall thickening— but only when endothelial cells still carried the GC‑B receptor. In rats and mice with an especially severe, human‑like form of pulmonary hypertension, starting CNP‑53 treatment after disease was established lowered lung pressures and improved heart measures. When combined with two existing drugs— one that blocks endothelin and another that targets a related growth pathway— CNP‑53 provided additional benefit without lowering normal blood pressure.
What this means for future treatments
Altogether, the study reveals that a natural hormone made by lung vessel linings acts as a guardian against pulmonary arterial hypertension. When the CNP–GC‑B signal is reduced, inflammation, vessel‑tightening chemicals, and growth pathways spiral out of control, thickening the artery wall and overworking the heart. Restoring or enhancing this signal with CNP‑based medicines, especially variants designed to last longer in the body, could open a new treatment route that targets the disease process itself rather than just relaxing blood vessels. While more work is needed to translate these findings into human therapies, the results suggest that helping the vessel lining talk properly may be key to protecting patients with this devastating lung‑heart disorder.
Citation: Yanagisawa, H., Kuwahara, K., Nakagawa, Y. et al. Endothelial C-type natriuretic peptide/guanylyl cyclase-B signaling prevents pulmonary arterial hypertension. Nat Commun 17, 2331 (2026). https://doi.org/10.1038/s41467-026-70139-2
Keywords: pulmonary arterial hypertension, endothelial signaling, C-type natriuretic peptide, vascular remodeling, lung circulation