Clear Sky Science · en
Olfactory cleft biopsy analysis of Alzheimer’s disease pathobiology across disease stages
Why the Nose May Hold Clues to Memory Loss
Many people notice a declining sense of smell long before memory problems appear, and doctors have long suspected a link to Alzheimer’s disease. This study asks a simple but powerful question: if smell-related tissue in the nose is both easy to reach and closely connected to the brain, could it provide an early, living window into Alzheimer’s biology—years before dementia sets in?
A Gentle Brush Inside the Nose
The researchers focused on a small patch high inside the nasal cavity called the olfactory epithelium, which houses the nerve cells that detect odors. Using a thin, flexible brush guided by a nasal endoscope, they gently scraped this area in awake volunteers during an outpatient visit, collecting thousands of living cells per person. They enrolled three kinds of participants: people with normal thinking and normal spinal fluid tests for Alzheimer’s; people whose spinal fluid quietly signaled early Alzheimer’s but who still tested cognitively normal ("pre-clinical" stage); and people with both abnormal spinal fluid and memory or thinking problems (clinical Alzheimer’s).
Reading Cell-by-Cell Activity
From these tiny brush samples, the team performed single-cell RNA sequencing, a technique that reads which genes are switched on in individual cells. This allowed them to catalog not just the smell neurons themselves, but also local immune cells such as T cells and myeloid cells (including microglia-like cells and macrophages) that patrol the tissue. In total they analyzed more than 220,000 cells. By comparing gene activity patterns across the three groups, they looked for changes that emerged early and grew stronger as disease progressed, focusing on signs of inflammation and stress in both nerve and immune cells.
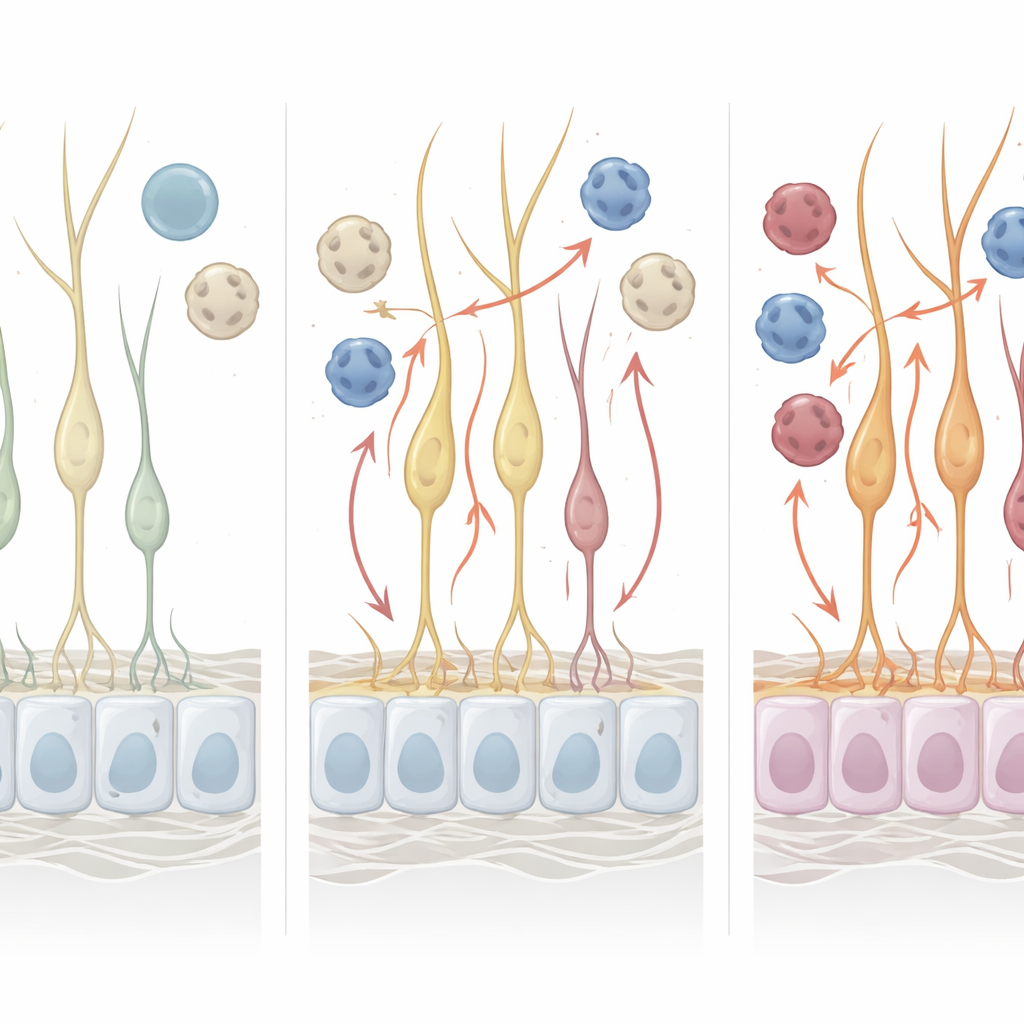
Immune Cells Sound an Early Alarm
One of the clearest signals came from T cells, a key arm of the body’s immune system. In the fluid surrounding the brain and spinal cord, previous work had shown unusually active CD8 memory T cells in people with Alzheimer’s. This study found that similar T cells in the nasal smell tissue were already abnormally activated in the pre-clinical group, before symptoms appeared. Myeloid cells in the same tissue also showed emerging inflammatory programs that became stronger in clinical Alzheimer’s, including increased activity of genes linked to brain immune risk factors. These immune shifts suggest that the olfactory lining mirrors, and may even help drive, brain-related inflammation seen in the disease.
Stressed Smell Neurons and Cross-Talk with Immunity
The smell neurons themselves showed gene changes that point to injury and altered communication with nearby immune cells. Some genes that can promote oxidative stress and inflammatory signaling were turned up, while others that normally help manage fat-like molecules and may protect against amyloid buildup were turned down. When the team modeled how neurons and immune cells might signal to each other, they found stronger predicted pathways for inflammatory molecules in both pre-clinical and clinical stages. This pattern paints a picture of ongoing, two-way cross-talk: immune cells becoming more activated, and neurons exhibiting stress signatures that may make them more vulnerable.
A Nasal Snapshot of Early Disease
To translate these complex patterns into something usable, the scientists combined the most informative genes from both immune and neuronal cells into a single “module score” for each person’s biopsy. This composite score distinguished people with either pre-clinical or clinical Alzheimer’s from healthy controls with good accuracy, and it tracked with the amount of amyloid-related change in spinal fluid. Because the test uses accessible tissue and can be repeated over time, it offers a promising way to monitor disease biology at an earlier, potentially more treatable phase than is possible with brain tissue itself.
What This Could Mean for Patients
The study suggests that a quick brush biopsy inside the nose can capture the same kinds of inflammatory and neuronal stress signals that are unfolding deep within the brain in Alzheimer’s disease—and that these signals are detectable even before memory problems arise. While larger, long-term studies are needed to confirm and refine this approach, the work opens the door to using the nasal olfactory tissue as an early warning system and research platform. In the future, such biopsies might help identify at-risk individuals, guide selection for preventive treatments, and reveal new targets to calm harmful neuro-immune activity before lasting damage is done.
Citation: D’Anniballe, V.M., Kim, S., Finlay, J.B. et al. Olfactory cleft biopsy analysis of Alzheimer’s disease pathobiology across disease stages. Nat Commun 17, 2245 (2026). https://doi.org/10.1038/s41467-026-70099-7
Keywords: Alzheimer’s disease, sense of smell, olfactory epithelium, neuroinflammation, early detection