Clear Sky Science · en
Systematic identification of variant-specific RNA structure-small molecule interactions exemplified by RNA G-quadruplexes
Why tiny changes in RNA matter for future medicines
Most of us inherit small differences in our DNA that make us unique. These tiny changes can subtly reshape the folds of RNA molecules inside our cells. The new study introduces a way to see how such genetic tweaks alter the grip of experimental drugs that target RNA. This matters because the same medicine might behave very differently from one person to another, especially in cancer or genetic disease.
New way to see where drugs touch RNA
Many drug developers are now looking at RNA, not just proteins, as targets for therapy. Small molecules can latch onto special RNA shapes and influence how genes are read, but until now most methods have only worked on a single “reference” RNA sequence. They largely ignored the countless single-letter changes found in real patients. The authors created a high-throughput approach called BIVID-MaP that can simultaneously test thousands of RNA variants and pinpoint where a small molecule binds each one, down to single-nucleotide resolution. 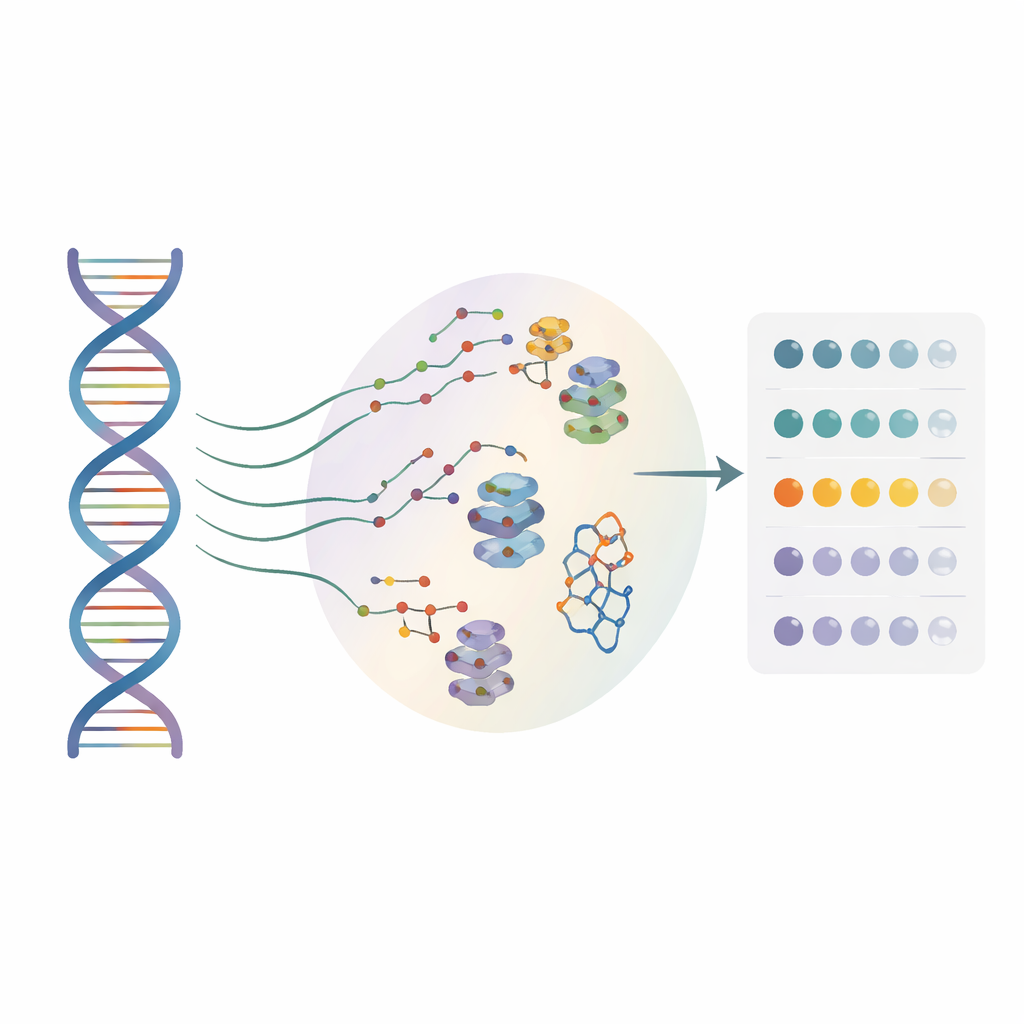
Turning chemical marks into a readable signal
BIVID-MaP works in three main steps. First, the team attaches a reactive tag to an RNA-binding small molecule. When this tagged compound nestles into its preferred RNA structure, such as a compact G-quadruplex formed by guanine-rich sequences, the tag forms a permanent chemical bond with a nearby RNA base. Second, a special enzyme copies the modified RNA into DNA. When it encounters one of these chemical marks, it tends to “skip” a single base, creating a tiny deletion in the copy. Third, deep sequencing reads out these deletion signatures across millions of molecules. Because almost all of the surrounding sequence is preserved, the deletions can be accurately assigned to specific genetic variants in a mixed pool.
Proving the method on a controlled test case
To confirm that BIVID-MaP truly detects variant-specific binding, the researchers studied an RNA segment from the CD44 gene that normally forms a G-quadruplex. A single G-to-A change disrupts this structure. In a mixture containing both versions, only the intact form showed strong deletion signals near the folded region when exposed to a berberine-based probe, and independent assays verified stronger binding there. The mutant, which cannot form the same shape, produced far fewer deletions and weaker binding. Other common mapping techniques either missed this difference or produced confusing background, underscoring the improved sensitivity and specificity of the new approach.
Scanning cancer-linked mutations across many genes
The team then scaled up. They built an RNA library of 5′ untranslated regions (5′ UTRs) from 283 cancer-related genes, each paired with real somatic mutations reported in tumors. Using a probe that recognizes G-quadruplex-like structures, BIVID-MaP uncovered numerous mutations that either strengthened or weakened small-molecule binding. In genes such as DAXX and ING2, single-letter changes in guanine-rich stretches markedly altered binding, in line with independent biophysical tests of G-quadruplex formation and direct binding measurements by mass spectrometry. In some cases, mutations outside the obvious G-tracts still shifted binding by subtly changing how likely nearby bases were to pair, reshaping the structural “ensemble” without grossly altering the predicted overall fold. 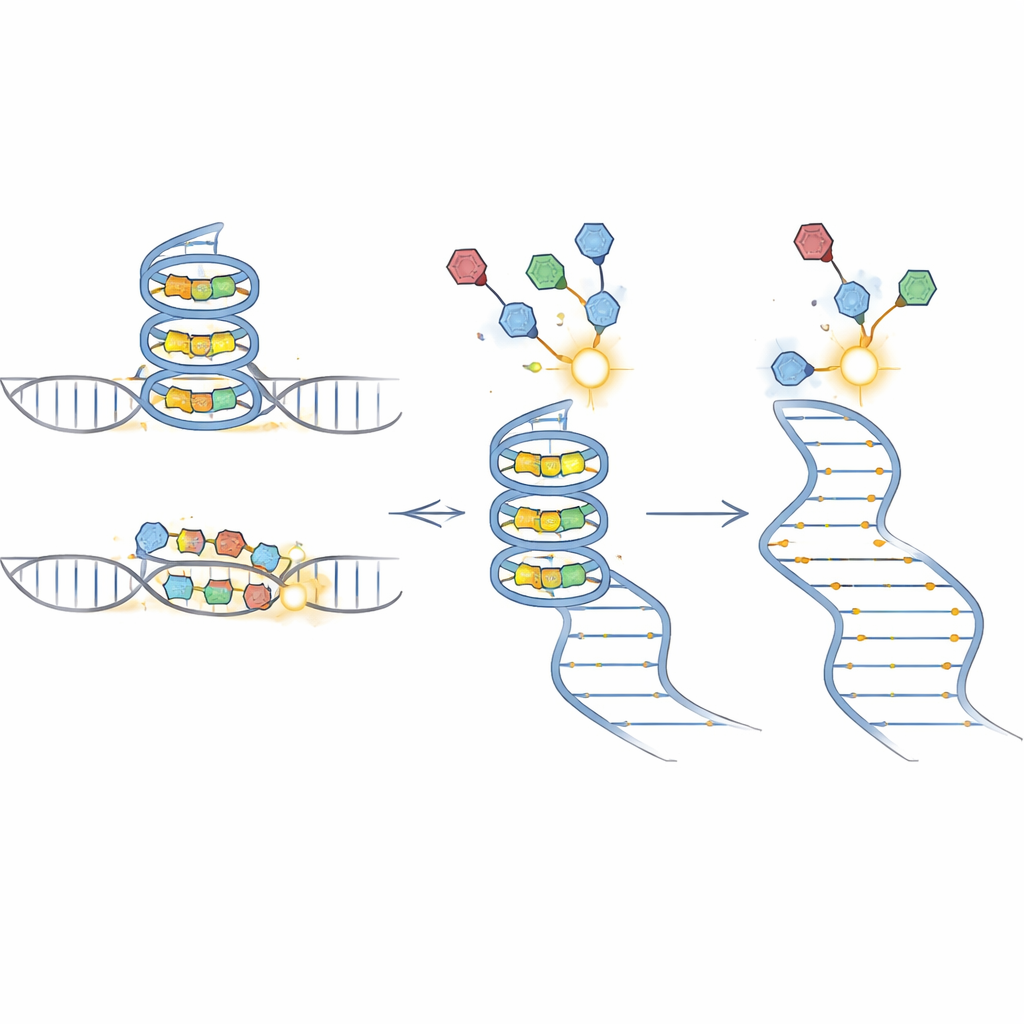
Seeing what computer predictions miss
Importantly, existing RNA structure and G-quadruplex prediction tools often failed to foresee the changes that BIVID-MaP revealed. Some variants that barely changed computational scores produced large real-world differences in binding. Others seemed to affect binding across a family of G-quadruplex-targeting compounds, while certain mutations had drug-specific effects. This shows that single-nucleotide variants can tune the landscape of RNA folds and drug contacts in ways that are hard to infer from sequence alone.
What this means for personalized RNA drugs
The authors conclude that BIVID-MaP offers a powerful route to map how individual genetic differences reshape RNA structures and their interactions with small-molecule drugs. By converting local chemical modifications into precise deletion signatures, the method can survey thousands of variants in parallel and highlight those that meaningfully change drug binding. In the long term, such maps could guide the design of RNA-targeting medicines that are more effective and safer across diverse patients, and reveal previously hidden RNA structures that might serve as new drug targets.
Citation: Miyashita, E., Onizuka, K., Chen, Y. et al. Systematic identification of variant-specific RNA structure-small molecule interactions exemplified by RNA G-quadruplexes. Nat Commun 17, 2243 (2026). https://doi.org/10.1038/s41467-026-70097-9
Keywords: RNA-targeting drugs, G-quadruplex, genetic variants, small-molecule binding, cancer mutations