Clear Sky Science · en
Optimizing spatial organization of FtsZ rings for large-scale constriction in synthetic cells
Why tiny rings matter for building synthetic cells
Imagine constructing a simple, artificial cell that can grow and split in two on its own. To do that, scientists must learn how to pinch a soft, soap-bubble‑like membrane in a controlled way, using only a few biological parts. This study shows how a bacterial protein can be helped to form large, stable rings on giant bubbles of fat, allowing these model “cells” to constrict almost to division. It reveals what kind of protein architecture is needed to bend and reshape a membrane at the scale of a whole cell.
Borrowing nature’s division toolkit
Many bacteria divide using a protein called FtsZ, a distant cousin of the tubulin that builds our own cells’ internal skeleton. FtsZ assembles into filaments that gather into a ring at the cell’s midpoint, marking where the cell will split. In living bacteria, a crowd of helper proteins attach this ring to the inner membrane, position it at mid‑cell, and link it to enzymes that remodel the cell wall. Previous laboratory attempts to re‑create this machinery in large, cell‑sized vesicles had managed to form rings and cause small dents in the membrane, but not a smooth, progressive constriction that reshapes a giant vesicle into two nearly separate halves.
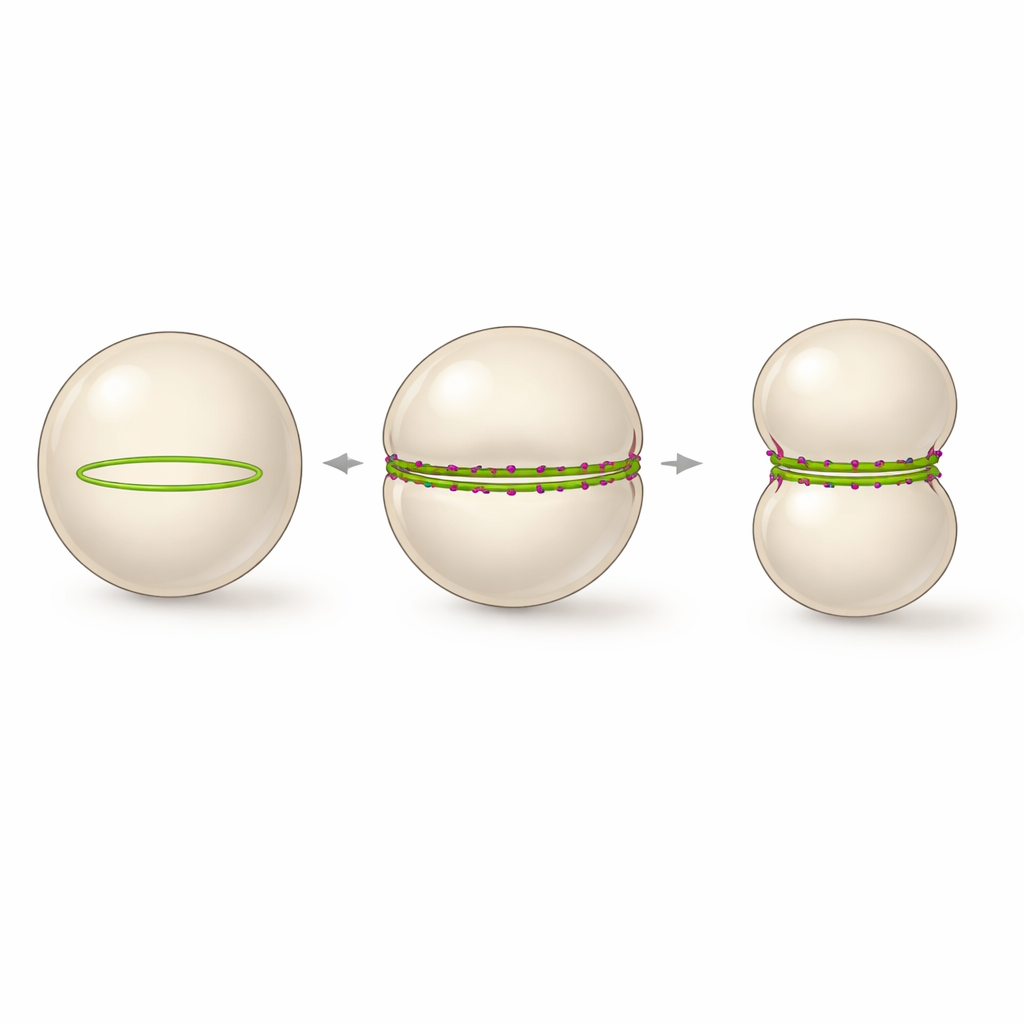
A minimalist approach to a dividing shell
The authors asked whether they could strip this system down to just two pieces: a version of FtsZ that can stick directly to membranes, and a short positively charged tail from another division protein, FtsN. They encapsulated these components inside giant unilamellar vesicles — hollow spheres of lipid resembling simplified cells — and supplied them with the energy molecule GTP and a slow‑acting GTP mimic to tune the protein dynamics. Without the FtsN tail, FtsZ formed many tiny rings and patches that only caused local dimples. With an excess of the FtsN fragment (called cytoFtsN), those small rings reorganized into a single, large ring that wrapped around the vesicle near its equator and was able to constrict over several hours.
From tangled network to single contractile ring
To understand how this small peptide changes behavior so dramatically, the team observed FtsZ filaments on flat, supported membranes with high‑resolution microscopy. Normally, FtsZ on such surfaces forms swirling, curved vortices. Adding growing amounts of cytoFtsN gradually erased these spirals and replaced them with straighter, more aligned bundles. Quantitative image analysis confirmed that the network became more coherent and ordered as peptide concentration rose. Fluorescence recovery experiments showed that subunits within these bundles exchanged more slowly, indicating that the peptide stabilizes the filaments and reduces their churning dynamics.

Electrostatic “Velcro” that locks filaments together
Biochemical measurements revealed that cytoFtsN and FtsZ interact mainly through opposite electric charges: FtsZ is overall negative, while the peptide carries clusters of positive charges. At lower salt levels, the peptide bound more strongly, promoted the formation of thicker FtsZ bundles in solution, and slowed the breakdown of filaments by dampening the protein’s GTP‑eating activity. When the researchers changed key positively charged residues in the peptide, or increased the salt concentration to screen electrostatic attraction, the bundling and reorganization effects disappeared. This points to a simple physical mechanism in which the positively charged peptide acts like molecular “Velcro,” reducing repulsion between FtsZ filaments so they can pack side by side into stiff bundles.
How a single ring reshapes a giant vesicle
Inside vesicles, this peptide‑stabilized bundling has a striking consequence. Instead of many small curved rings, the straightened FtsZ bundles are forced to follow the large curvature of the vesicle, assembling into one continuous ring that spans the entire circumference. Time‑lapse imaging showed that whenever such a fully closed ring formed, the vesicle reliably deformed from a sphere into a dumbbell shape, with a tight waist where the ring sat. The process could be followed in real time as the vesicle diameter shrank and the constriction deepened, nearly splitting the compartment in two, although final scission was not achieved. The authors propose that a stable, equator‑spanning FtsZ ring with slowed but still active turnover is the minimal architecture needed to drive large‑scale membrane constriction in a synthetic cell.
What this means for future synthetic life
By showing that a membrane‑anchored FtsZ variant plus a short cationic peptide can reshape giant lipid vesicles almost to division, this work defines a clear design rule for artificial cells: a single, coherent protein ring that matches the cell’s size and is mechanically stable over time is enough to pinch a soft boundary on the cellular scale. More complex factors may still be required to cut the membrane completely, but the study demonstrates that controlled large‑scale constriction does not demand a full cast of bacterial division proteins. Instead, careful tuning of filament organization and dynamics can turn a simple protein scaffold into an effective contractile ring, bringing truly self‑dividing synthetic cells a step closer.
Citation: Panevska, A., Šakanović, A., Paccione, G. et al. Optimizing spatial organization of FtsZ rings for large-scale constriction in synthetic cells. Nat Commun 17, 2320 (2026). https://doi.org/10.1038/s41467-026-70091-1
Keywords: synthetic cell division, FtsZ ring, membrane constriction, minimal divisome, cytoskeletal self-organization