Clear Sky Science · en
The dynamic distribution of genetic tandem amplifications in a heteroresistant Escherichia coli population revealed by ultra-deep long read sequencing
Hidden pockets of survival
When doctors treat bacterial infections with antibiotics, they assume the whole swarm of microbes will respond in the same way. But sometimes, a tiny fraction of the population quietly resists, survives treatment, and sets the stage for failure or relapse. This study uncovers how such rare survivors arise and shift over time in Escherichia coli, and introduces a powerful way to watch these genetic changes unfold cell by cell.
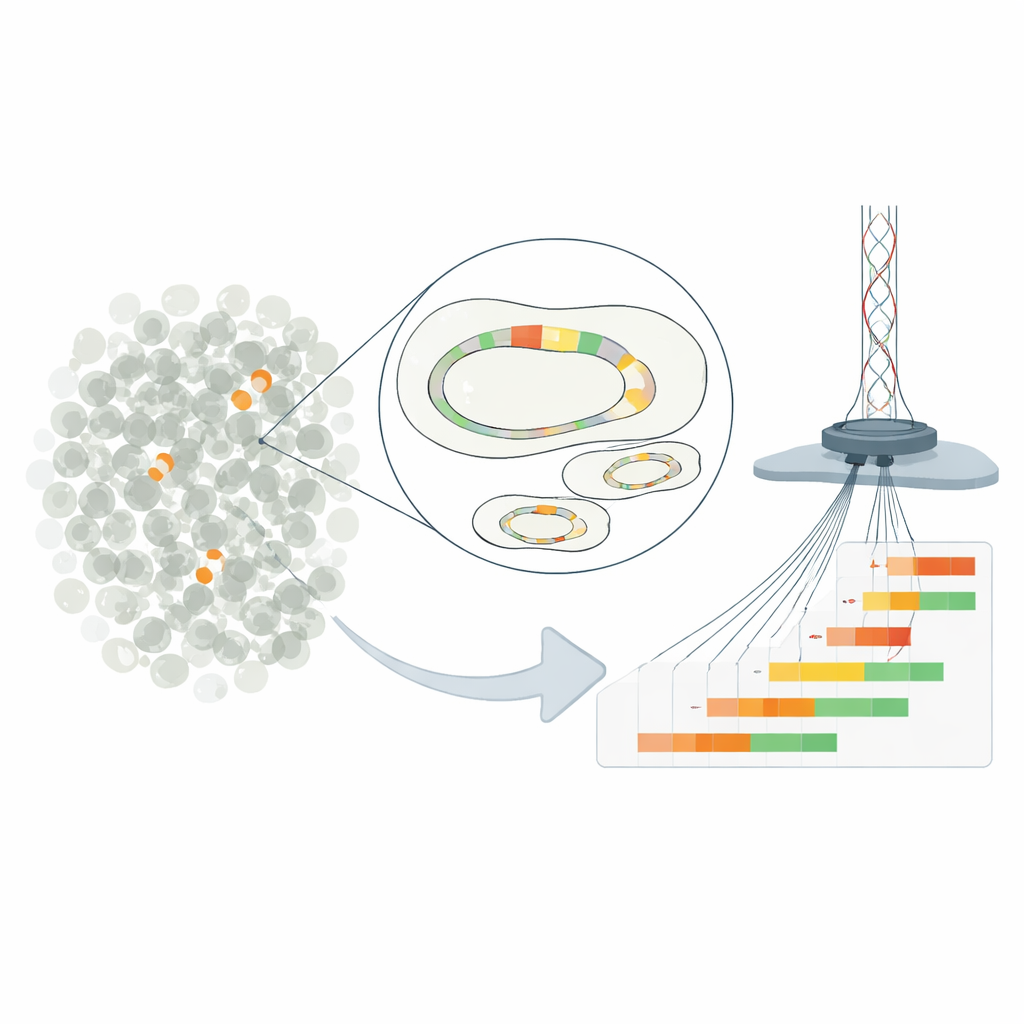
Small genetic repeats, big consequences
The work focuses on “heteroresistance,” a situation where most bacteria in a sample appear susceptible to a drug, yet a small minority can withstand much higher doses. In the E. coli strain studied, this behavior comes from repeated copies of a resistance gene carried on a plasmid—a small DNA circle separate from the main chromosome. By duplicating a short DNA segment containing a beta-lactamase gene, the bacteria can boost the production of an enzyme that breaks down the antibiotic piperacillin-tazobactam. The more repeats a cell carries, the more enzyme it makes and the higher its survival chances in the presence of the drug.
Reading DNA circles in unprecedented detail
Traditional methods can only estimate the average number of gene copies across an entire bacterial population, masking the rich diversity between individual cells. To overcome this, the researchers engineered the clinical plasmid so it could be cut at a precise spot and then isolated from the rest of the genome. They used ultra-deep Nanopore long-read sequencing to read full plasmid molecules end to end, counting exactly how many repeated resistance units each one contained. This approach reached a resolution down to one cell in 100,000, revealing plasmids with zero to more than a dozen gene copies coexisting in the same culture. Checks with other techniques confirmed that, on average, the new method reported accurate copy numbers.
How antibiotics reshape the population
With this tool in hand, the team followed what happened when they exposed E. coli cultures to increasing doses of piperacillin-tazobactam and then removed the drug. As antibiotic levels rose, the overall distribution shifted toward plasmids carrying more copies of the resistance gene, but cells with fewer copies never fully disappeared. When the drug was withdrawn and the bacteria were grown for hundreds of generations, the population only gradually drifted back toward lower copy numbers and never completely returned to its starting state. Separate growth tests showed that each extra gene copy gave a measurable step up in drug tolerance, tightly linking the observed genetic distributions to the visible heteroresistance pattern in standard lab assays.

Protection from neighbors and quiet costs
To explain why low-copy cells persisted even under strong treatment, the authors built a mathematical model that combined bacterial growth, antibiotic breakdown, and the chance events that add or remove gene copies. The simulations showed that “indirect resistance” plays a key role: highly resistant cells pump out enough beta-lactamase to lower the drug concentration in the shared environment, incidentally shielding less resistant neighbors. As long as the extra gene copies impose only a small growth cost when no drug is present, this protective effect helps maintain a wide spread of copy numbers, slowing the return to a fully susceptible population. The model also showed how ordinary adaptation to the growth medium—unrelated mutations that improve growth—can lock in particular copy-number states by making some subpopulations fitter than others.
Why this matters for patients and beyond
For non-specialists, the message is that a bacterial culture is not a uniform enemy: it is a constantly shifting mix of individuals with different capacities to survive antibiotics. By directly counting gene copies on thousands of single DNA molecules, this study links that hidden diversity to treatment-relevant resistance in a quantitative way. Such insights could improve how genome sequencing is used to predict hard-to-detect heteroresistance, help identify bacterial strains that are especially likely to cause treatment failure, and guide better diagnostic tests. Because tandem gene amplifications also drive rapid adaptation in viruses, tumors, and other organisms, the same strategy could be adapted far beyond E. coli, offering a new window into how repeated bits of DNA help life outmaneuver threats.
Citation: Jonsson, S., Guliaev, A., Berryhill, B.A. et al. The dynamic distribution of genetic tandem amplifications in a heteroresistant Escherichia coli population revealed by ultra-deep long read sequencing. Nat Commun 17, 2113 (2026). https://doi.org/10.1038/s41467-026-70044-8
Keywords: antibiotic heteroresistance, gene amplification, Escherichia coli, long-read sequencing, beta-lactamase