Clear Sky Science · en
Single-molecule dynamics reveal ATP binding alone powers substrate translocation by an ABC transporter
Why tiny cellular pumps matter
Every cell in your body depends on microscopic machines that move molecules across membranes, shaping everything from nutrient uptake to immune defense. One major class of these machines, called ABC transporters, uses the cell’s chemical fuel ATP to push cargo uphill, against natural gradients. Yet for decades, scientists have argued about a basic question: does the simple act of ATP binding power the transport step, or is the energy only released when ATP is broken down? This study zooms in on a single transporter, molecule by molecule, to settle that debate.
Watching one transporter at a time
Instead of measuring the combined behavior of millions of molecules, the researchers built a setup that lets them follow individual transport events in real time. They focused on TmrAB, a bacterial cousin of the human TAP transporter that loads immune peptides for display to killer T cells. TmrAB sits in a membrane and ferries short protein fragments (peptides) from one side to the other using ATP. To see each transport step, the team trapped a single TmrAB molecule in an artificial membrane bubble (a liposome) and enclosed inside that bubble a specially engineered "sensor" protein that changes its fluorescence when it catches a peptide. Using a highly sensitive microscope, they could then see, as brief jumps in the light signal, the exact moments when one peptide after another arrived inside the bubble.
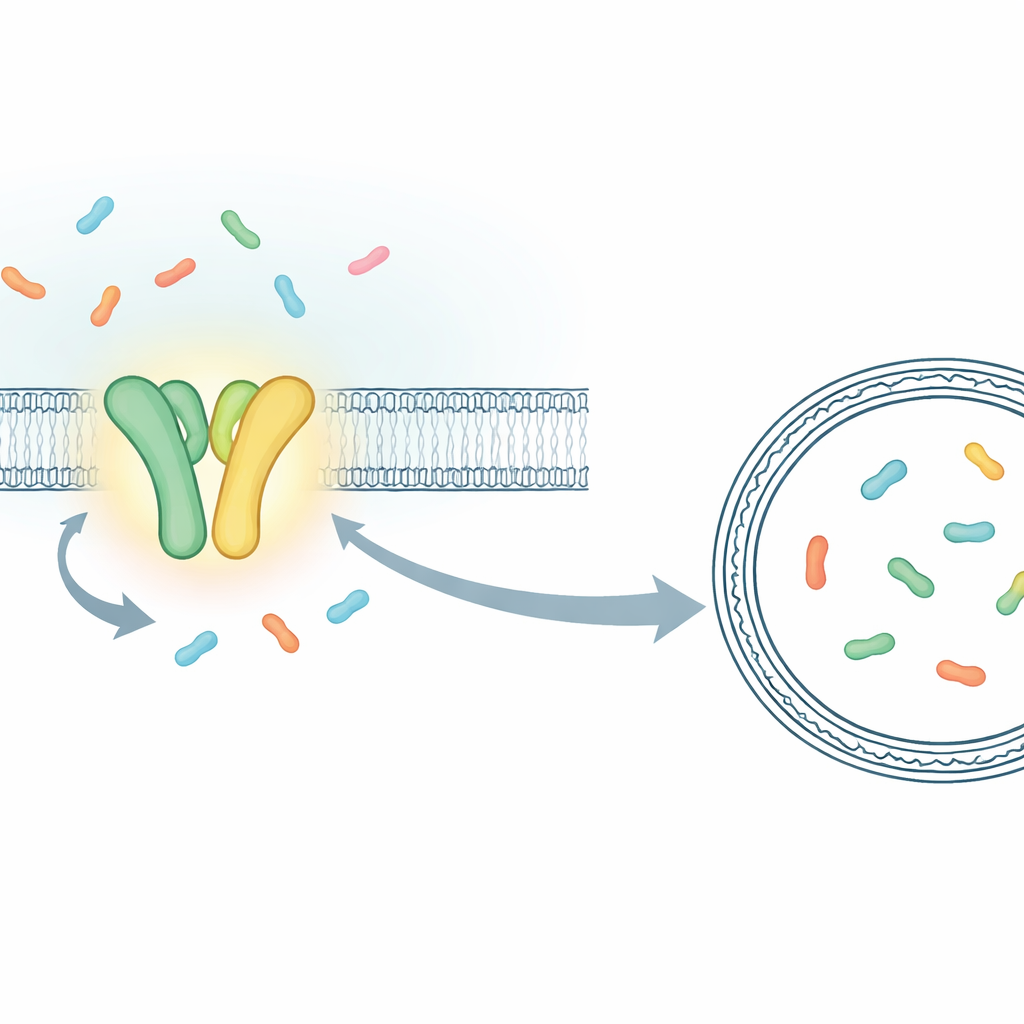
Proving that one ATP binding moves one peptide
With this single-molecule assay in hand, the researchers compared normal TmrAB with a modified, slow-turnover version whose cycles are stretched out in time. The slow variant made it possible to distinguish individual events that would otherwise blur together. They showed that when ATP and peptides were present together, the sensor inside the liposome shifted from its "empty" to its "loaded" state in discrete steps, each consistent with a single peptide being transported. By analyzing how fast the sensor closed around peptides and estimating the tiny volume inside each liposome, they could even translate these light changes into approximate peptide concentrations. The numbers matched what you would expect if one, then two peptides accumulated in bubbles of about 100 nanometers in diameter, confirming that they were now counting transport events one by one.
ATP binding alone flips the molecular switch
The key controversy concerns magnesium ions (Mg2+), which usually accompany ATP in cells and are known to be required for efficient ATP breakdown. Some models suggested Mg2+ might also be needed for ATP to bind or for the transporter to close. To tease these roles apart, the researchers removed Mg2+ using a chelating agent but still supplied ATP. Under these conditions, both the normal and slow transporter could carry out exactly one peptide transport step per molecule, but then stopped. That behavior indicates that ATP can still bind and force the transporter to switch from an inward-facing to an outward-facing shape and move a peptide, even without Mg2+. However, without Mg2+ the transporter cannot efficiently split ATP, and therefore cannot reset itself for another round.
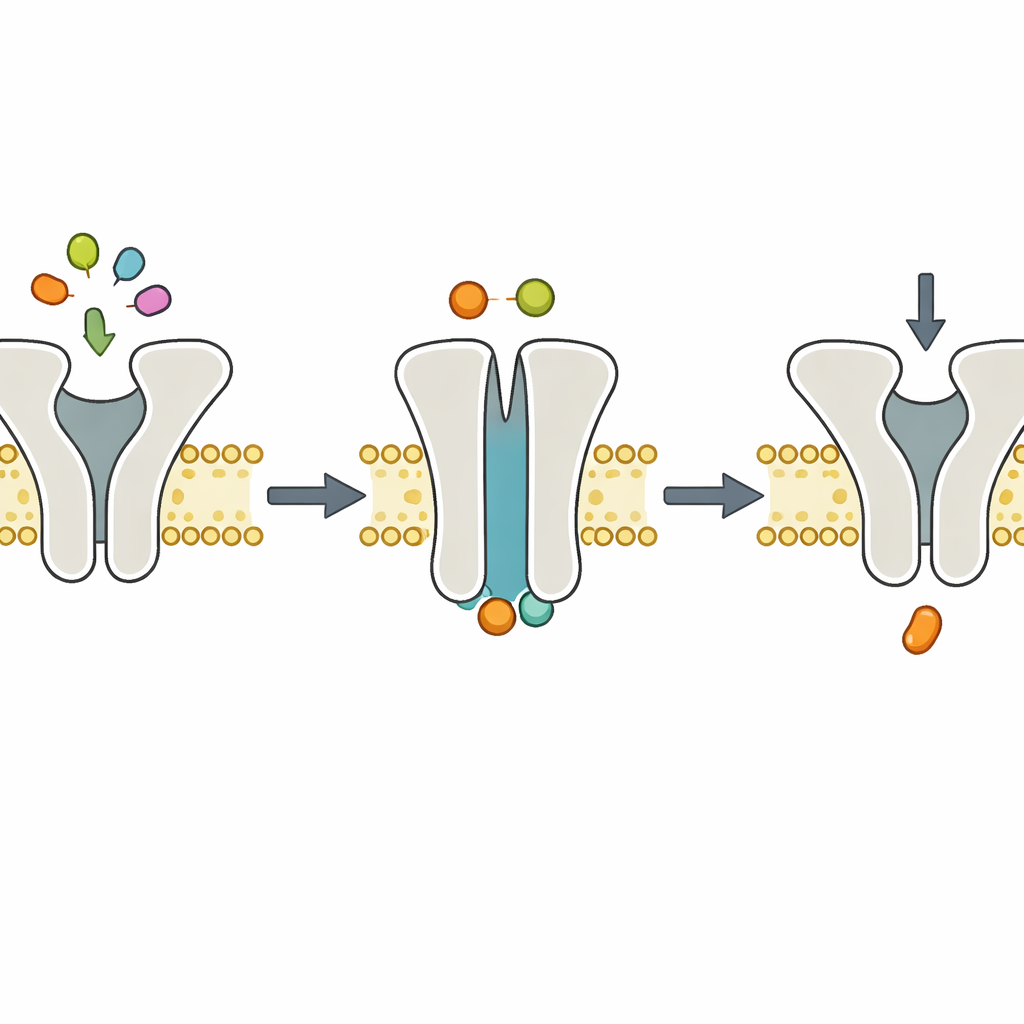
Structural snapshots back up the dynamics
To see how the machine looks during this Mg2+-free step, the team turned to cryo–electron microscopy, which images flash-frozen molecules at near-atomic detail. They reconstituted TmrAB in lipid nanodiscs, added ATP without Mg2+, and froze the complexes. The resulting structures showed TmrAB in an outward-facing, "occluded" conformation with ATP wedged into both of its internal binding pockets, but with no Mg2+ visible. These snapshots were essentially indistinguishable from earlier structures obtained with ATP and Mg2+ present. Together with biochemical assays showing that ATP breakdown completely stalls without Mg2+, the pictures support a simple division of labor: ATP binding drives the conformational switch that moves the peptide, while Mg2+-dependent ATP hydrolysis carries out the recovery step.
A clearer picture of how cellular pumps work
By watching single transporters and solving their structures, this work resolves a long-standing question about ABC transporters: the mechanical power stroke that moves a peptide is triggered by ATP binding itself, not by the later chemical breakdown of ATP. Mg2+ enters the story mainly to allow ATP to be split, which in turn resets the transporter so it can work again. Beyond clarifying a fundamental piece of cell biology, this single-molecule approach offers a blueprint for dissecting other membrane machines that underlie immunity, drug resistance, and many diseases—one molecule and one transport step at a time.
Citation: Nocker, C., Pečak, M., Nocker, T. et al. Single-molecule dynamics reveal ATP binding alone powers substrate translocation by an ABC transporter. Nat Commun 17, 3038 (2026). https://doi.org/10.1038/s41467-026-70021-1
Keywords: ABC transporters, ATP binding, single-molecule FRET, membrane transport, TAP and TmrAB