Clear Sky Science · en
Prussian blue nanoparticles targeting multiple PANoptosome-mediated PANoptosis for myocardial ischemia-reperfusion injury therapy
Why protecting the healing heart matters
When a person has a heart attack, doctors rush to reopen the blocked artery and restore blood flow. This life-saving step, however, comes with a hidden cost: the sudden return of oxygen can itself injure the heart, killing cells and setting the stage for heart failure. This study explores an inventive nanomedicine that aims to protect the heart during this vulnerable window by blocking several intertwined cell-death routes at once, potentially leading to gentler recoveries after heart attacks.
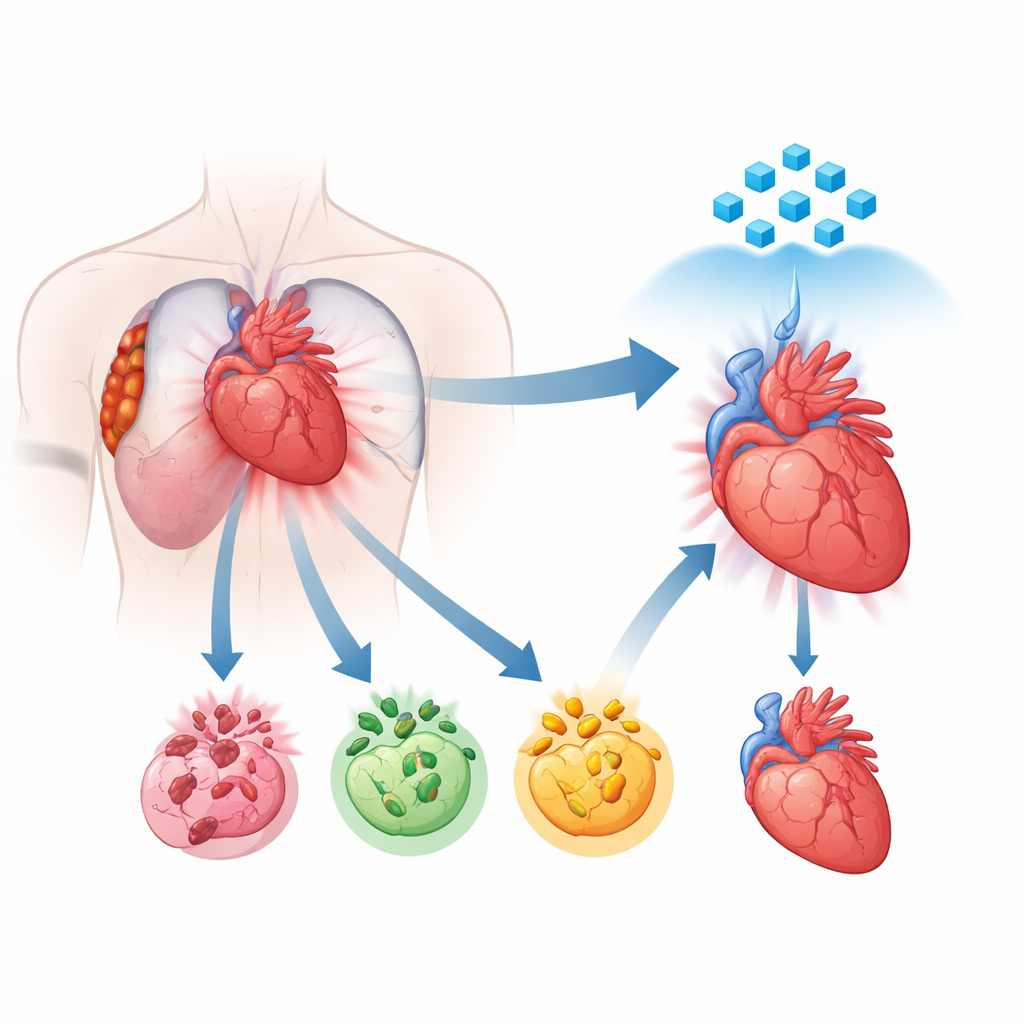
A new view of how heart cells die
For years, scientists have known that damaged heart cells can die through different programmed routes, including apoptosis, necroptosis, and pyroptosis. Each follows its own molecular script, but growing evidence shows that these routes do not operate in isolation. Instead, they talk to each other and can join into a combined, runaway process called PANoptosis. In this state, several death programs fire together, making therapies that block only one pathway frustratingly weak. The authors used single-nucleus RNA sequencing of human heart tissue from patients with recent heart attacks to map where and how strongly these death programs are switched on. They found that in the most damaged regions, heart muscle cells showed a powerful shift toward this combined death mode, marking PANoptosis as a central driver of injury.
Designing a tiny multi-tool for the injured heart
To tackle such a complex process, the team turned to Prussian blue, a long-approved medical compound best known as an antidote for certain metal poisonings. At the nanoscale, Prussian blue particles behave like tiny catalytic robots that can quench harmful molecules and influence cell behavior. Through computer simulations, the researchers showed that these nanoparticles could directly attach to three key protein hubs—RIPK1, ZBP1, and AIM2—that help assemble the molecular machinery of PANoptosis. By binding to these hubs in different ways, the particles were predicted to interfere with the construction of the death-inducing complexes that coordinate the three cell-death routes.
Guiding nanoparticles straight to damaged heart tissue
Nanoparticles only work if they reach the right place, so the team wrapped Prussian blue particles in natural membranes stripped from platelets, the blood cells that home in on injured vessels. This coating created PB@PM, a biomimetic carrier that circulates like a platelet but delivers a therapeutic payload. In mice subjected to a controlled heart attack and reopening of the artery, imaging showed that PB@PM accumulated far more strongly in the injured heart region than uncoated particles, while largely avoiding healthy organs. Importantly, the coated particles were well tolerated: blood tests and tissue examinations did not reveal obvious toxicity, supporting their potential for further development.
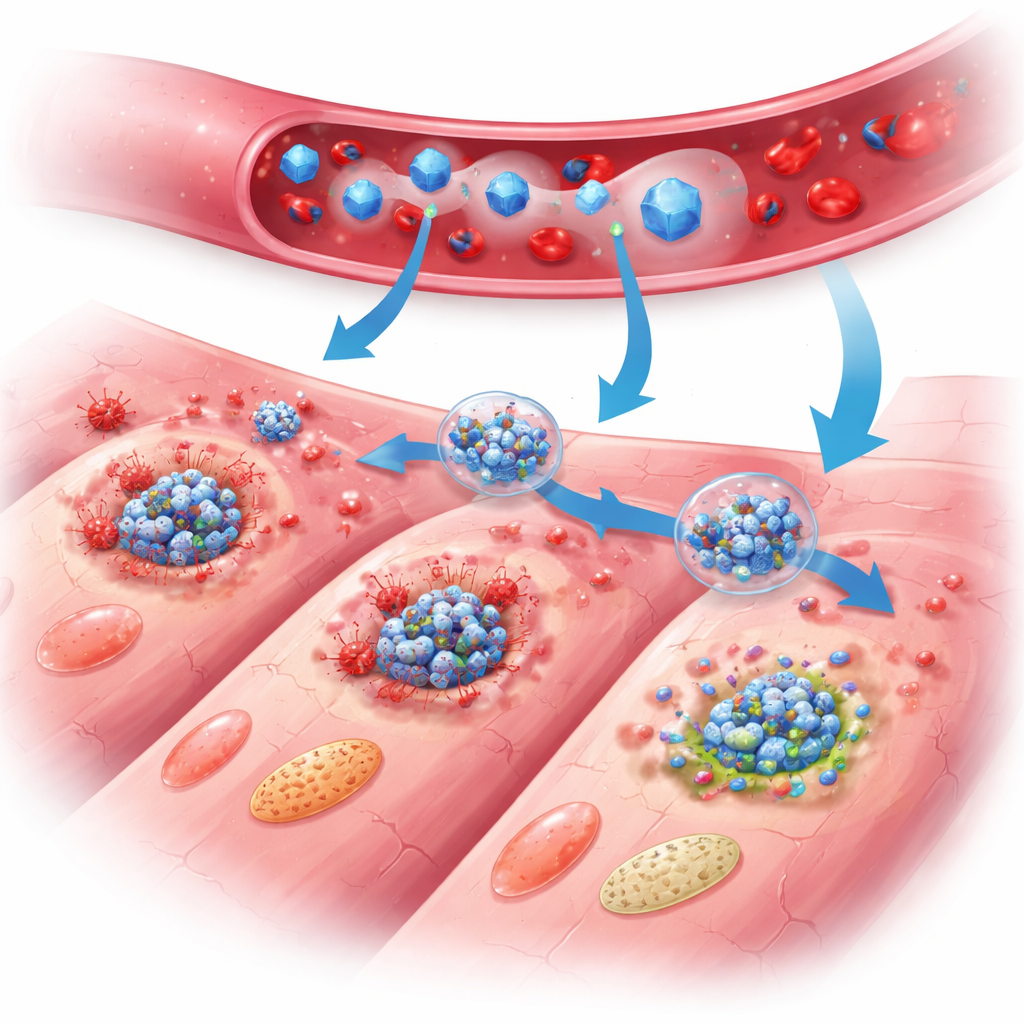
How the nano-shield protects the heart
In treated mice, the benefits of PB@PM became clear over days and weeks. Heart-pumping function improved, scar size shrank, and the heart wall thinned less compared with untreated animals. Detailed tissue studies revealed fewer dying heart cells, calmer inflammatory cells, and better-structured repair tissue with healthier blood vessel growth and less abnormal enlargement of surviving cells. At the molecular level, broad gene-expression profiling and protein measurements showed that PB@PM quieted the three major programmed-death pathways all at once, disrupted assembly of the multi-protein PANoptosis complexes, and reduced levels of signaling molecules that fuel inflammation. The particles also scavenged damaging reactive oxygen species, stabilized mitochondria—the cell’s power plants—and restored more normal energy metabolism, removing key triggers that would otherwise push cells toward self-destruction.
What this could mean for future heart attack care
Put simply, this work shows that a cleverly engineered nanoparticle can act like a multi-pronged shield for the heart, dampening a tangle of destructive death signals rather than chasing one pathway at a time. By homing in on injured heart tissue, binding core control proteins, and easing oxidative and inflammatory stress, PB@PM sharply reduced the chain reaction of cell loss and scarring that often follows reopening a blocked artery. While much testing remains before this strategy could reach patients, the study offers a proof of concept that targeting PANoptosis as a whole—and doing so with smart, biologically inspired particles—may open a new chapter in protecting the heart after a heart attack.
Citation: Xu, L., Jiang, L., Wu, R. et al. Prussian blue nanoparticles targeting multiple PANoptosome-mediated PANoptosis for myocardial ischemia-reperfusion injury therapy. Nat Commun 17, 2329 (2026). https://doi.org/10.1038/s41467-026-70012-2
Keywords: myocardial ischemia reperfusion, nanoparticle therapy, programmed cell death, cardiac inflammation, mitochondrial protection