Clear Sky Science · en
Targeting de novo pyrimidine synthesis confers vulnerability to copper-mediated ATR inactivation in PARP inhibitor-resistant ovarian cancer
Why this research matters
Many women with ovarian cancer are treated with drugs that sabotage tumor cells’ ability to fix damaged DNA. These medicines, called PARP inhibitors, can work well at first, but tumors often adapt and return. This study uncovers how a copper-carrying drug and a key metabolic weakness may help push resistant ovarian cancers past their limits, pointing toward smarter combination treatments and more durable responses.
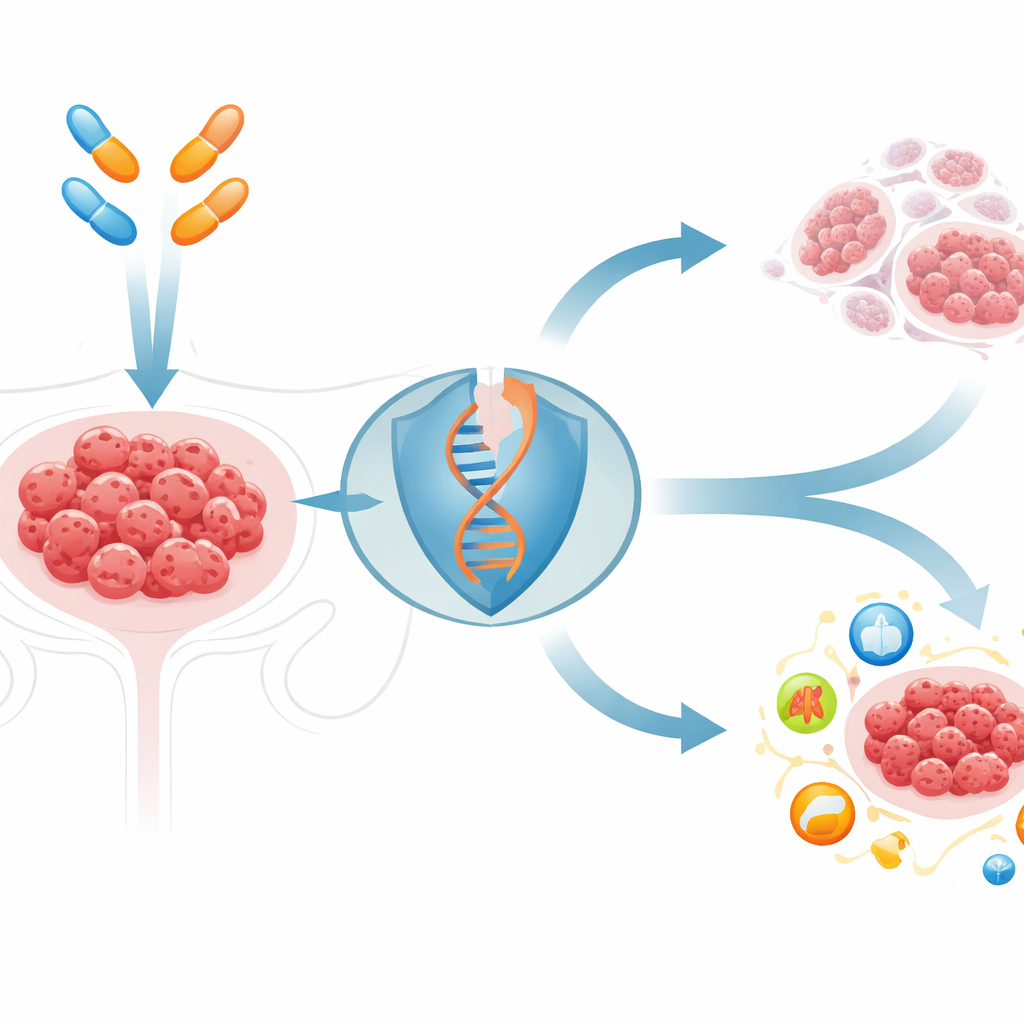
Cracking stubborn tumor defenses
PARP inhibitors exploit a flaw in how some cancers repair broken DNA. They work best in tumors with inherited defects in BRCA genes, but most ovarian cancers have intact BRCA and respond poorly or only briefly. The researchers screened 144 cell‑death–related compounds alongside a standard PARP inhibitor and found that one drug, elesclomol, stood out. Elesclomol ferries copper into cells. When combined with PARP inhibitors in BRCA‑normal ovarian cancer cells and mouse tumors, this copper‑boosting drug sharply increased DNA damage and shrank tumors far more than either drug alone, without obvious toxicity in healthy organs.
Copper jams a DNA repair switch
To understand why copper made PARP blockade more lethal, the team looked at a major DNA‑damage signaling route centered on a protein called ATR. This pathway helps cells survive when their DNA replication is stressed—precisely the situation created by PARP inhibitors. In tumor cells that had survived initial drug exposure, ATR and its partner protein CHK1 were strongly switched on, while a related pathway (ATM‑CHK2) stayed quiet. Detailed biochemical tests and computer‑guided structural modeling showed that copper binds directly to ATR’s helper protein, ATRIP, at specific cysteine sites. This binding distorts ATRIP’s shape, breaks its contact with ATR, and shuts down ATR‑CHK1 signaling, leaving damaged DNA unrepaired and making PARP‑treated cancer cells far more likely to die.
The hidden role of nucleotide fuel
Even with ATR and PARP both crippled, some cancer cells and residual tumors managed to hang on. To learn how, the researchers profiled hundreds of small molecules inside drug‑adapted cells. They found a striking surge in building blocks for DNA known as pyrimidines, particularly those made through the “de novo” pathway, in which cells synthesize these components from scratch. Tracer experiments confirmed that resistant cells channeled more nitrogen from glutamine into fresh pyrimidines, while purine building blocks were not similarly boosted. Adding extra pyrimidine components like uridine or thymidine to cultures blunted the killing power of PARP plus ATR or copper‑based treatment, suggesting that an abundant DNA‑building supply helps tumors tolerate otherwise lethal DNA damage.
Striking a metabolic weak spot
The team then tested whether blocking this pyrimidine supply line could close the escape hatch. They used BAY‑2402234, an experimental drug that inhibits DHODH, a key enzyme in de novo pyrimidine synthesis. In ovarian cancer cell lines and patient‑derived organoids, adding the DHODH inhibitor restored sensitivity to PARP plus ATR or copper treatment, wiping out previously drug‑tolerant cells. In mouse tumors and in eight patient‑derived xenograft models, tumors that were resistant to PARP alone—and even to PARP combined with ATR or copper blockade—showed strong growth suppression when pyrimidine synthesis was also blocked. Tumors with naturally high pyrimidine metabolite levels were the hardest to treat with PARP‑based approaches but responded when this metabolic pathway was targeted.
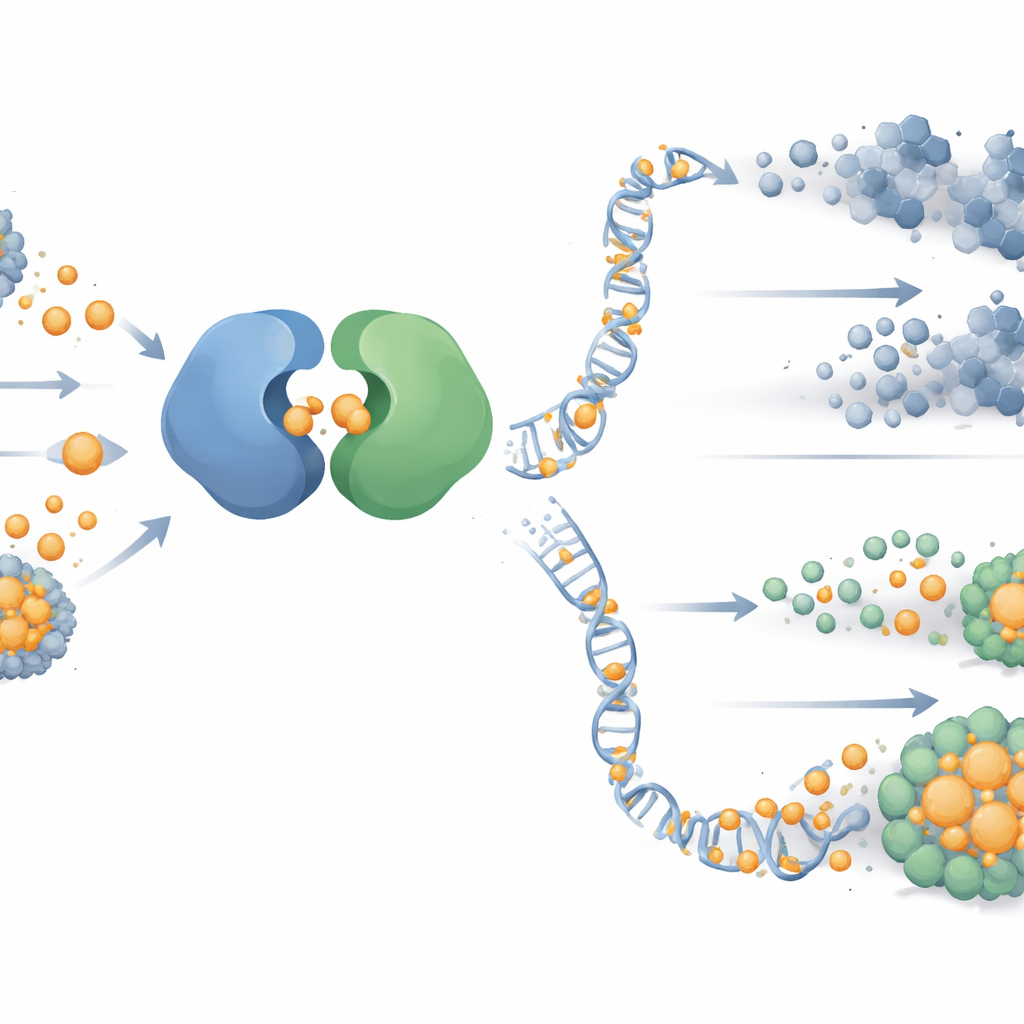
What this could mean for patients
This study reveals two linked vulnerabilities in PARP inhibitor‑resistant ovarian cancer. First, copper can be used as a precise wrench to disable a key DNA repair switch, ATR, by prying apart its partnership with ATRIP, making standard DNA‑targeting drugs more potent. Second, tumors that adapt by ramping up pyrimidine production become dependent on this metabolic route, and blocking it can re‑sensitize them to treatment. In practical terms, the findings argue for tailored combination therapies: PARP inhibitors plus ATR‑targeting agents for tumors with low pyrimidine reliance, and a triple approach that also blocks pyrimidine synthesis for tumors that are metabolically primed to resist. While further clinical testing is needed, the work charts a clearer map for overcoming one of the toughest forms of drug resistance in ovarian cancer.
Citation: Nan, Y., Wang, K., Hu, M. et al. Targeting de novo pyrimidine synthesis confers vulnerability to copper-mediated ATR inactivation in PARP inhibitor-resistant ovarian cancer. Nat Commun 17, 3142 (2026). https://doi.org/10.1038/s41467-026-70001-5
Keywords: ovarian cancer, PARP inhibitors, copper therapy, DNA repair, pyrimidine metabolism