Clear Sky Science · en
Chiral peptidoglycan mimics target bacterial wall biosynthesis for pathogen intervention
A New Way to Hunt Harmful Germs
Antibiotic-resistant infections are rising around the world, yet doctors still lack tools that can precisely spot and attack dangerous bacteria without harming our own cells. This study introduces a clever molecular "decoy" that imitates a key building block of bacterial cell walls. These designer molecules can both light up infections for imaging and ferry antibiotics directly to the microbes, offering a fresh strategy to fight hard-to-treat pathogens.
Imitating the Bacterial Armor
Bacteria are wrapped in a tough mesh called the cell wall, built from sugars and unusual mirror-image amino acids that our own cells largely do not use. The researchers took advantage of this difference by creating long sugar-based chains decorated with a specific mirror-form of alanine, a common amino acid. These chains, called chiral peptidoglycan mimics, are designed to closely resemble the natural ingredients bacteria use to build their walls, but are absent from mammalian tissues. By fine-tuning how much of this D-shaped alanine was attached, the team produced versions that strongly favored bacteria over human cells.
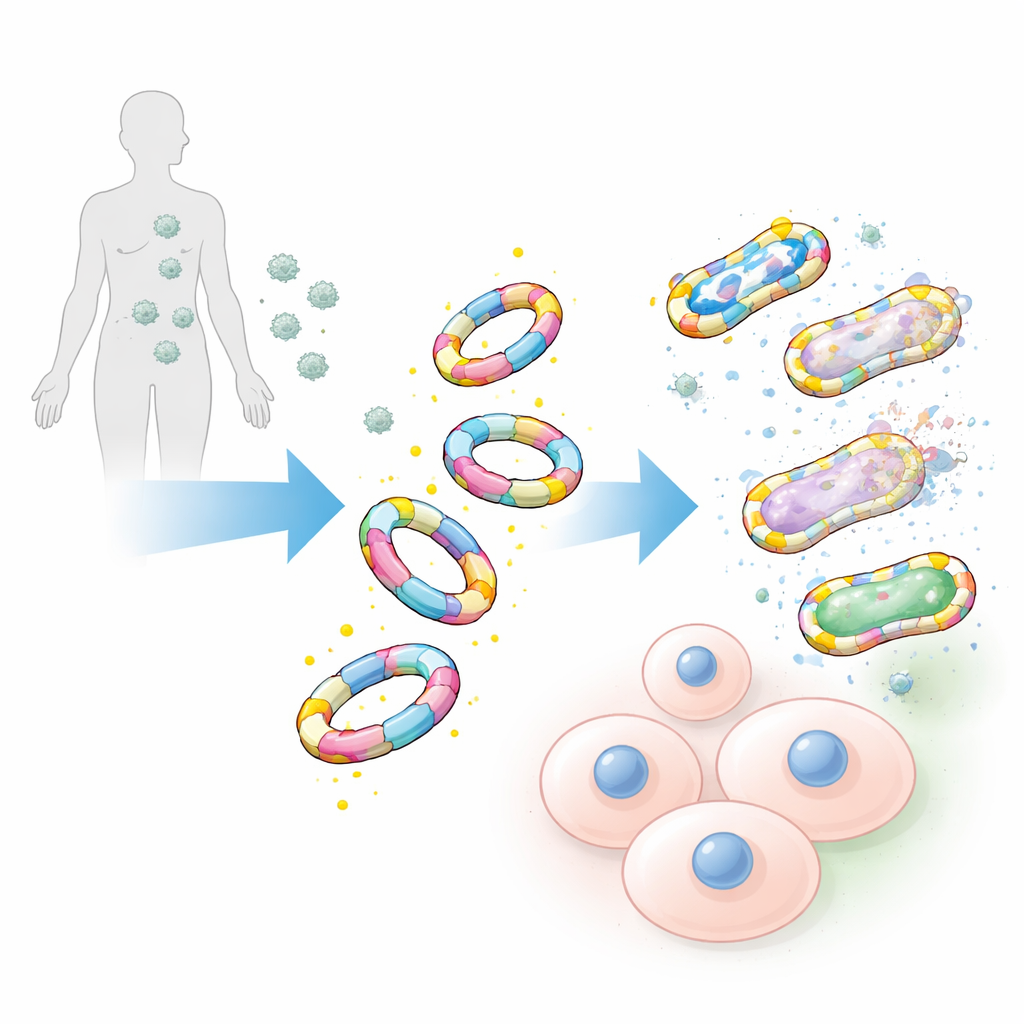
Locking Onto Bacteria, Not Our Cells
When tested against a wide range of pathogens, including notorious hospital strains such as methicillin-resistant Staphylococcus aureus and other ESKAPE bacteria, the D-alanine–decorated mimics bound to bacterial surfaces with striking efficiency. Their mirror-image counterparts built from the L-form of alanine, as well as the original sugar chains alone, showed only weak or negligible binding. The team also compared these large mimics with traditional small chemical probes based on single D-alanine molecules. Even when the small probes were made extremely bright, they still labeled far fewer bacteria and gave weaker signals, underscoring the power of the larger, more lifelike design.
Hijacking the Cell Wall Assembly Line
To understand how recognition becomes so strong, the scientists examined how the mimics interact with the bacterial wall-building machinery. They found that the D-alanine–based chains are not just sticking to the outside; they are taken up as if they were genuine building blocks and fed into the same enzymatic steps that normally join cell wall components together. Chemical analysis detected new intermediate molecules that arise only when these mimics are present, and computer simulations showed that key wall-building enzymes grip the mimics even more tightly than the natural substrates. Over several rounds of bacterial growth, the mimics accumulated in the wall, gradually distorting its structure and making it leaky.
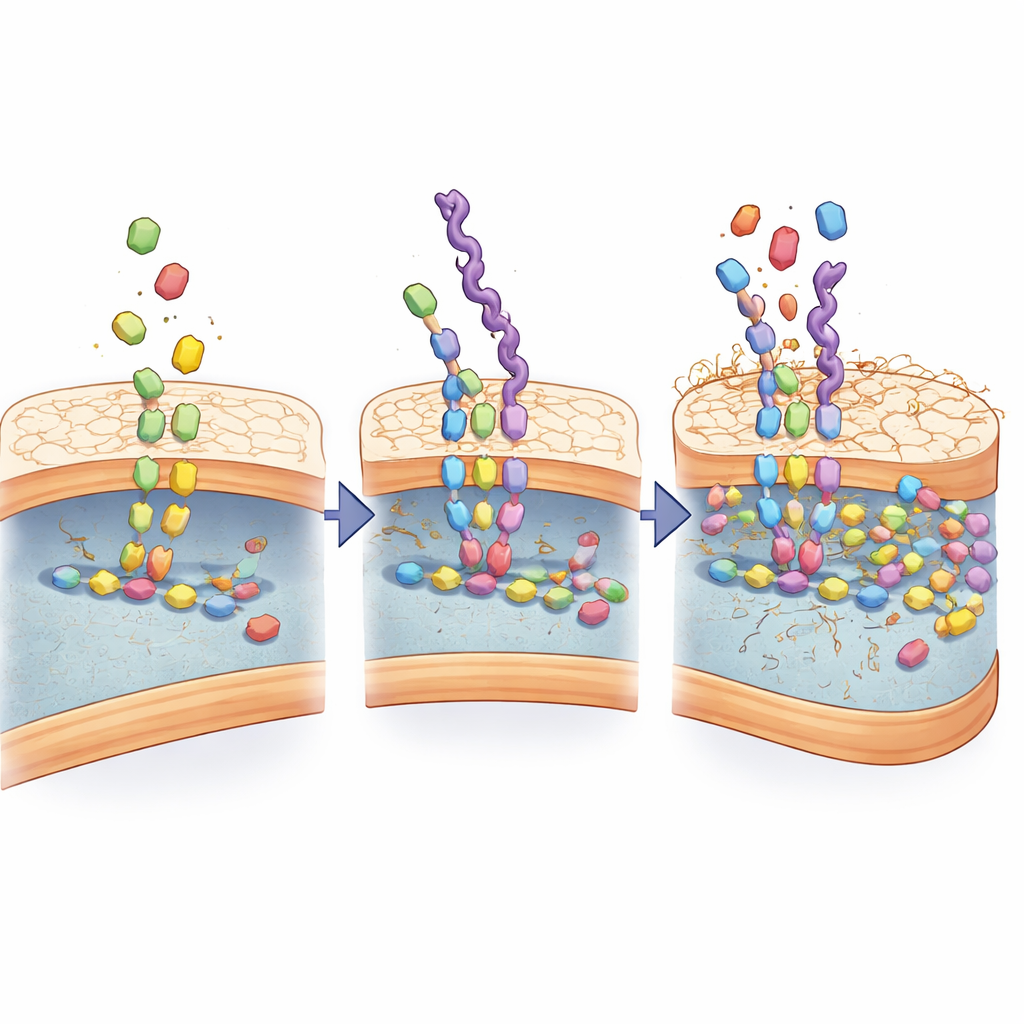
Turning Mimics into Smart Drug Carriers
Because these chains naturally gather into tiny particles in water and carry many chemical handles, the team used them as scaffolds for imaging dyes and antibiotics. Fluorescent versions clearly outlined bacteria in cell cultures, while scarcely interacting with mammalian cells grown alongside. The researchers then loaded the antibiotic tetracycline into the mimic-based particles. In lab tests, this packaging made tetracycline several times more effective, killing even resistant strains at much lower doses than the free drug. Microscopy revealed that antibiotic-loaded mimics crowded onto bacteria, penetrated their envelopes, and triggered extensive cell death compared with untargeted formulations.
Fighting Infections Inside the Body
The most demanding tests took place in mouse models of localized skin wounds and life-threatening abdominal infections. In infected wounds, the fluorescent mimics homed in on bacteria deep in the tissue and, when carrying tetracycline, almost completely cleared the microbes. This led to faster healing, thicker new skin, more orderly collagen deposition, and richer growth of small blood vessels. In systemic infection models, the mimics accumulated specifically in infected organs such as the spleen and lungs, where they sharply reduced bacterial counts when loaded with antibiotic. Importantly, high doses of the mimics alone showed good safety, with no obvious damage to major organs or blood chemistry.
What This Could Mean for Future Treatments
By building a close chemical look-alike of the bacterial cell wall’s own ingredients, this work demonstrates a new way to “blend in” with a pathogen’s metabolism instead of just attacking from the outside. These chiral mimics serve as both high-precision beacons for imaging and smart vehicles that steer antibiotics directly to their microbial targets, including resistant strains, while sparing mammalian cells. If translated into clinical tools, such biomimetic platforms could help doctors see exactly where infections lurk and treat them more effectively with lower drug doses, offering a promising weapon against the growing challenge of antimicrobial resistance.
Citation: Deng, K., Zou, D., Zeng, Z. et al. Chiral peptidoglycan mimics target bacterial wall biosynthesis for pathogen intervention. Nat Commun 17, 3258 (2026). https://doi.org/10.1038/s41467-026-69967-z
Keywords: antibiotic resistance, bacterial cell wall, targeted drug delivery, molecular imaging, nanomedicine