Clear Sky Science · en
Complete defluorination of PFASs via photocatalytic reduction in water
Why breaking “forever chemicals” finally looks possible
For decades, a class of pollutants known as PFAS—often called “forever chemicals”—has built up in drinking water, wildlife, and even human blood because they barely break down in nature. This study describes a new light-powered material that can almost completely strip fluorine atoms from some of the most stubborn PFAS in water, turning them into harmless small molecules without needing harsh chemicals or expensive energy inputs. That makes it a promising blueprint for safer, more practical cleanup of contaminated water supplies.
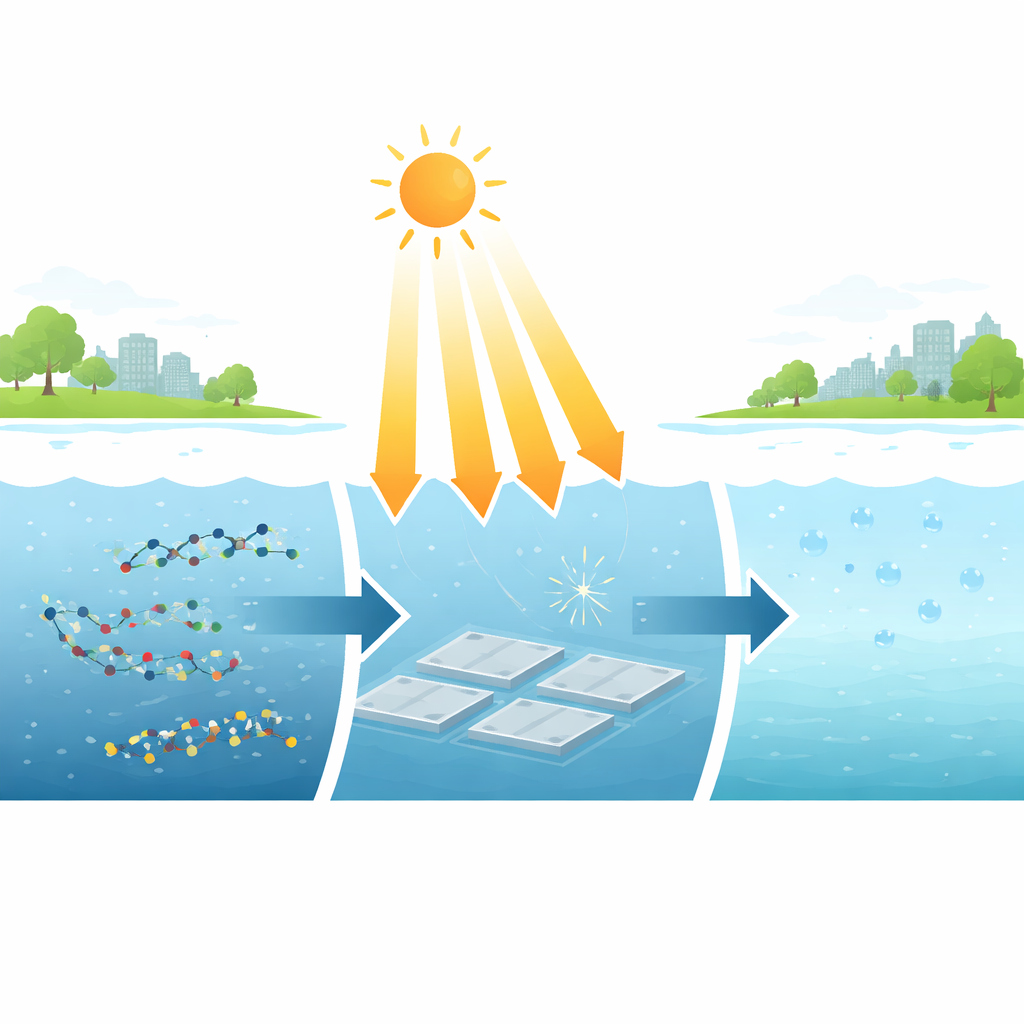
The problem with stubborn fluorinated pollutants
PFAS (per- and polyfluoroalkyl substances) are used in nonstick pans, firefighting foams, and stain-resistant coatings. Their carbon–fluorine bonds are among the toughest in chemistry, which is why PFAS linger in the environment and build up in living organisms. Two of the most widespread PFAS, PFOA and PFOS, are now classified as human cancer risks, and countries have set drinking water limits in the parts-per-trillion range. Unfortunately, most existing methods that can truly destroy PFAS—like intense ultrasound, high-temperature treatment, or strong chemical additives—work only at high concentrations and require large amounts of energy, making them hard to scale for real-world water treatment.
A new light-driven catalyst built for extreme bonds
The researchers designed a special organic material called TAPP, which self-assembles into ordered stacks of flat molecules. When illuminated with visible light, these stacks form a long-lived radical state—a version of the molecule with an unpaired electron—that remains stable for more than a week in air. Because electric charge is spread out across the molecule and its attached amino groups, this radical can absorb a second dose of light and push electrons to extremely high energies. Those energized electrons are strong enough to attack the ultra-stable carbon–fluorine bonds in PFAS, something ordinary light-based catalysts cannot do.
How the catalyst grabs and dismantles PFAS
The TAPP particles are positively charged in water, while PFOS and related PFAS carry negative charges at one end and fluorinated tails at the other. This contrast pulls the pollutants onto the catalyst surface: the negatively charged head groups interact with protonated amino sites, and the fluorinated tail lies along the flat aromatic surface through gentle attraction forces. After this “pre-concentration” step, visible light repeatedly excites TAPP. Its radical form sends high-energy electrons directly into antibonding regions of the PFAS carbon–fluorine bonds. That extra electron destabilizes the rigid helical structure of the PFAS chain, stretches the carbon backbone, and makes individual C–F bonds much easier to snap.
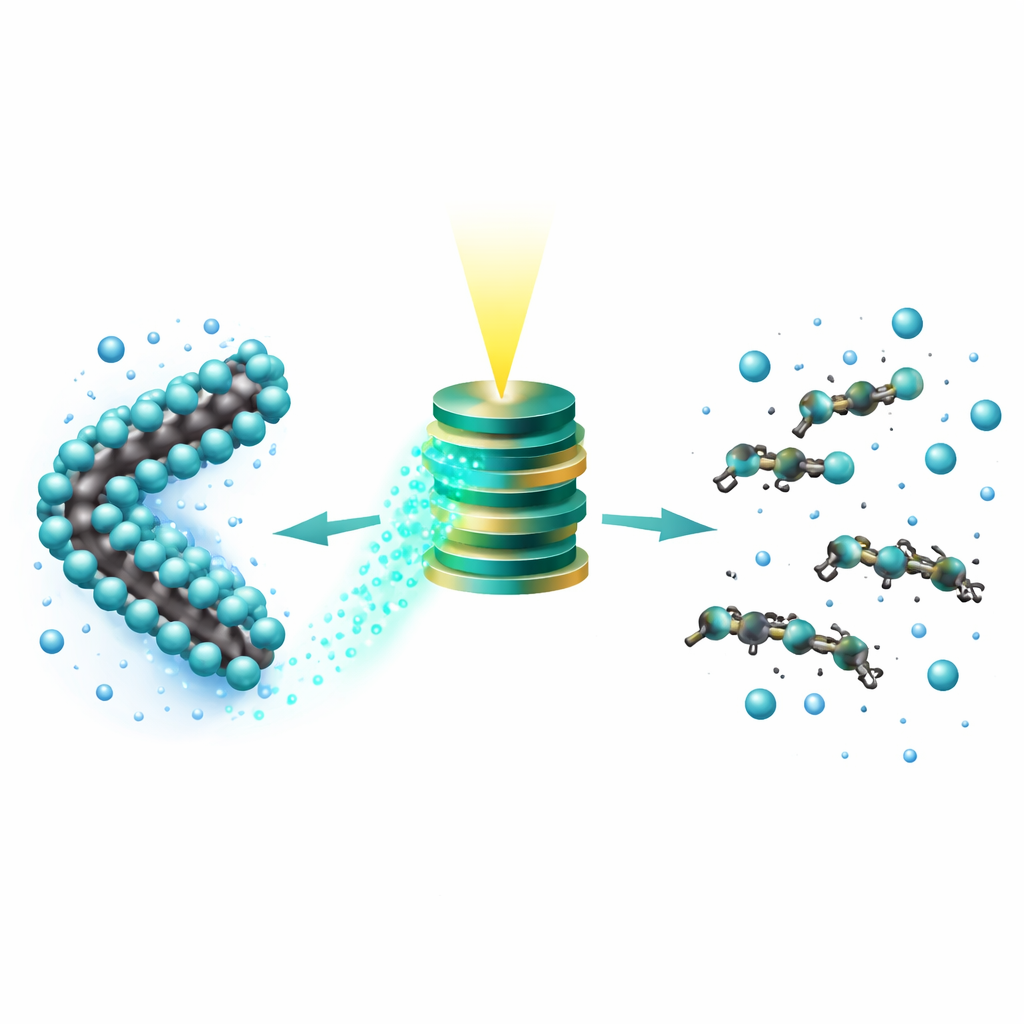
From deadly chains to harmless fragments
In carefully controlled water experiments with realistic contaminant levels (around 0.1 parts per million), TAPP removed PFOS from solution and, under light, converted nearly all of its fluorine into free fluoride ions within about two days. Detailed chemical analysis showed that the original PFOS signal disappeared and was replaced by simple organic acids such as formate, oxalate, and lactate, with no PFOS left on the catalyst surface. Early in the reaction, the team detected a series of shorter fluorinated fragments, supporting a picture in which electrons first knock fluorine off the chain, then the weakened carbon skeleton breaks apart into smaller pieces that are further oxidized to benign end products.
Performance in real water and energy savings
To test practicality, the scientists built a small outdoor reactor mimicking a treatment unit in a wastewater plant. With only natural sunlight, their TAPP-based system completely defluorinated PFOS-spiked water in three days. The catalyst worked well even in the presence of natural organic matter and common ions, though some salts slowed the process by competing for surface sites. Compared with other light-driven PFAS treatments, this approach consumed about 90–98% less energy per volume of water and avoided toxic metals or added oxidizing chemicals. The catalyst also stayed active for at least five repeated cycles with minimal loss of efficiency.
What this means for cleaning up “forever chemicals”
This work shows that carefully engineered organic materials can use ordinary visible light to generate electrons energetic enough to crack the toughest carbon–fluorine bonds in PFAS, all while operating in simple water with no sacrificial chemicals. By combining strong pollutant adsorption, a long-lived radical state, and multi-step photoexcitation, the TAPP catalyst turns persistent PFAS into fluoride and small, harmless organic molecules. While scaling up and tackling the full variety of PFAS will still take time, this study offers a realistic path toward solar-powered, low-cost destruction of “forever chemicals” in drinking water and wastewater systems.
Citation: Chong, M., Zhou, Q., Xu, J. et al. Complete defluorination of PFASs via photocatalytic reduction in water. Nat Commun 17, 3081 (2026). https://doi.org/10.1038/s41467-026-69933-9
Keywords: PFAS remediation, photocatalysis, water treatment, defluorination, environmental chemistry