Clear Sky Science · en
Prospective multicenter study of ctDNA versus tumor tissue guiding FGFR-targeted therapy in metastatic urothelial cancer
Why this research matters
For people living with advanced bladder cancer, time is precious. Powerful new drugs can target specific weaknesses in cancer cells, but today those weaknesses are usually found by testing pieces of tumor removed during surgery or biopsy. Those samples are often old, small, or hard to get, and they may not fully represent all the cancer growing in the body. This study explores whether a simple blood test that reads bits of tumor DNA floating in the bloodstream can reliably guide the use of a key targeted drug, potentially making treatment faster, less invasive, and more accurate.
A new way to read cancer’s fingerprints
The work focuses on a family of switches in cells called FGFRs that, when altered, can drive bladder cancer growth. A drug called erdafitinib can shut down tumors with the right FGFR changes, but patients only benefit if those changes are correctly identified. Traditionally, doctors rely on tumor tissue testing, which can fail because there is not enough tissue, the sample is old, or different parts of the cancer carry different mutations. The researchers asked whether fragments of tumor DNA circulating in blood (known as circulating tumor DNA, or ctDNA) could act as a real-time, whole-body snapshot of a patient’s cancer, revealing FGFR alterations that tissue tests might miss.
Putting blood and tissue tests head-to-head
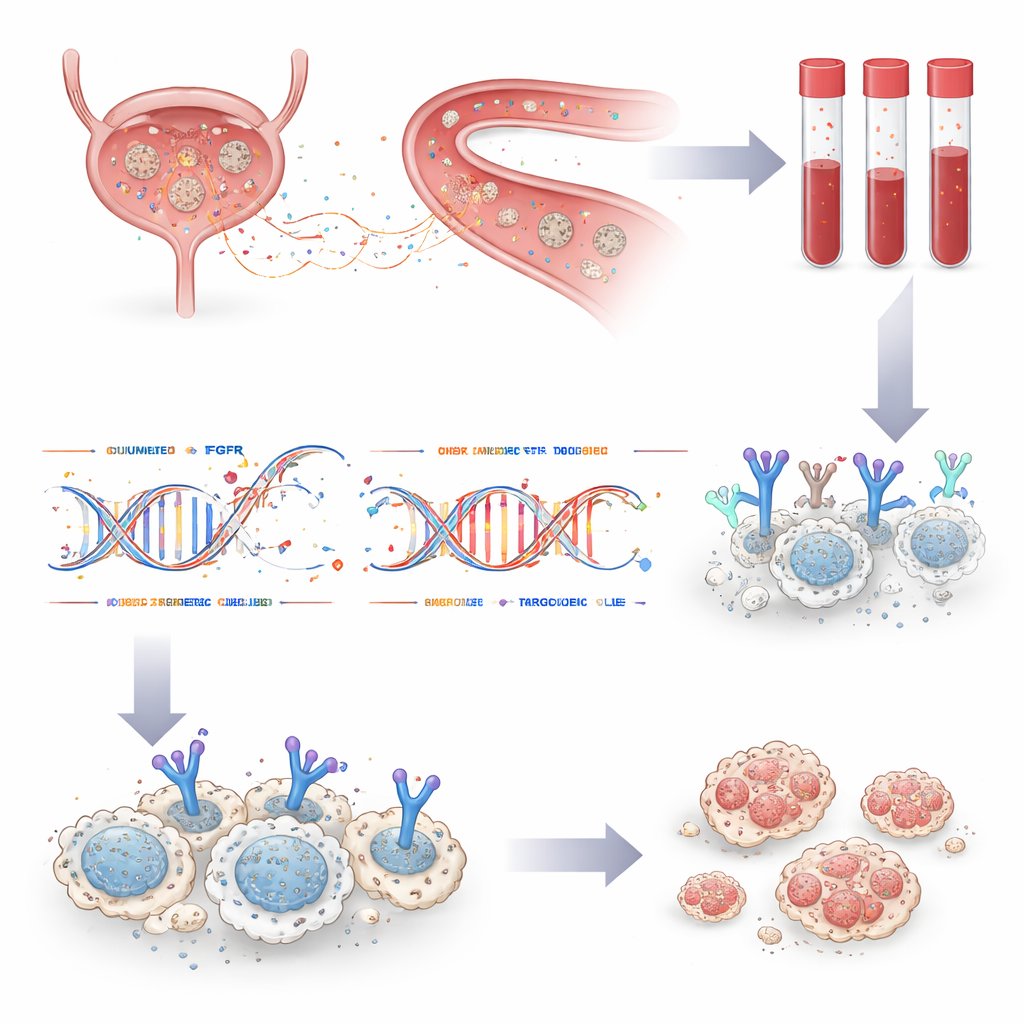
In a pan-Canadian study across 12 centers, the team enrolled 208 people with metastatic urothelial carcinoma—an advanced form of bladder and urinary tract cancer—who were being considered for erdafitinib. Every participant had standard clinical FGFR testing on previously collected tumor tissue, and also provided blood samples for ctDNA analysis using a custom DNA sequencing panel. By comparing results from both sources in the same patients, the researchers could directly assess how well the blood test matched tissue testing and whether it added new, actionable information.
Strong agreement and hidden targets revealed
Among 125 patients whose blood contained enough tumor DNA for evaluation and who also had usable tissue results, FGFR status matched between the two methods in 90 percent of cases. The blood test detected 84 percent of FGFR alterations seen in tissue, and tissue detected 79 percent of those found in blood. Crucially, ctDNA testing uncovered FGFR changes in seven patients whose tumors had been labeled normal by tissue testing. One such patient, treated based on the blood test result alone, experienced a remarkably long benefit from erdafitinib—remaining on therapy for nearly three years. The study also showed that repeat blood draws over time could clarify results for patients whose first sample had too little tumor DNA, and could track changes in FGFR status as the disease evolved or spread.
Watching cancer change and resist treatment
For 21 patients who ultimately received erdafitinib, the researchers followed outcomes and, for many, collected additional blood at the time the drug stopped working. On average, patients went about seven and a half months before their cancer progressed, similar to or better than earlier trials. In most cases with progression samples, the original FGFR alteration was still visible in ctDNA, confirming that the cancer remained dependent on the same pathway. In some patients, new FGFR mutations appeared that are known to interfere with drug binding—molecular escape routes that allowed cancer cells to grow despite treatment. In one person, several distinct resistance mutations arose in parallel, along with a new FGFR fusion, illustrating the diverse ways tumors can adapt under drug pressure.
How this could change care
The authors propose a practical testing strategy that starts with the blood test: if ctDNA is detectable, it can often identify FGFR alterations quickly and may even find targets that tissue testing misses. If the blood sample does not contain enough tumor DNA, doctors would then fall back on tissue testing where possible. Modeling with their data suggests that this “ctDNA first, reflex tissue” approach could modestly improve the overall sensitivity of FGFR testing compared with using tissue alone, while also sparing some patients from repeat invasive biopsies and delays. Although not every patient sheds enough DNA into the blood for this to work, and larger treatment studies are still needed, the message is clear. Blood-based FGFR testing is reliable, complementary to tissue analysis, and has real potential to make precision treatment more accessible and timely for people with advanced bladder cancer.
Citation: Müller, D.C., Murtha, A.J., Bacon, J.V.W. et al. Prospective multicenter study of ctDNA versus tumor tissue guiding FGFR-targeted therapy in metastatic urothelial cancer. Nat Commun 17, 3263 (2026). https://doi.org/10.1038/s41467-026-69927-7
Keywords: metastatic urothelial cancer, circulating tumor DNA, FGFR-targeted therapy, liquid biopsy, precision oncology