Clear Sky Science · en
Th17 cells require the DNA repair sensor xeroderma pigmentosum complementation Group C to control oxidative DNA damage in a murine model
Guardians of Our Immune Defenders
T helper 17 cells, or Th17 cells, are a special kind of white blood cell that patrols our body’s barriers, such as the gut and skin, to fend off bacteria and fungi. But they are also involved in autoimmune diseases when their responses go awry. This study asks a deceptively simple question: how do these highly active cells keep their own DNA safe from the chemical stress they generate while fighting infection? The answer centers on a DNA damage sensor called XPC, best known for protecting skin cells from sunlight. Here, researchers show that XPC is also critical for keeping Th17 cells healthy, energetic, and effective.
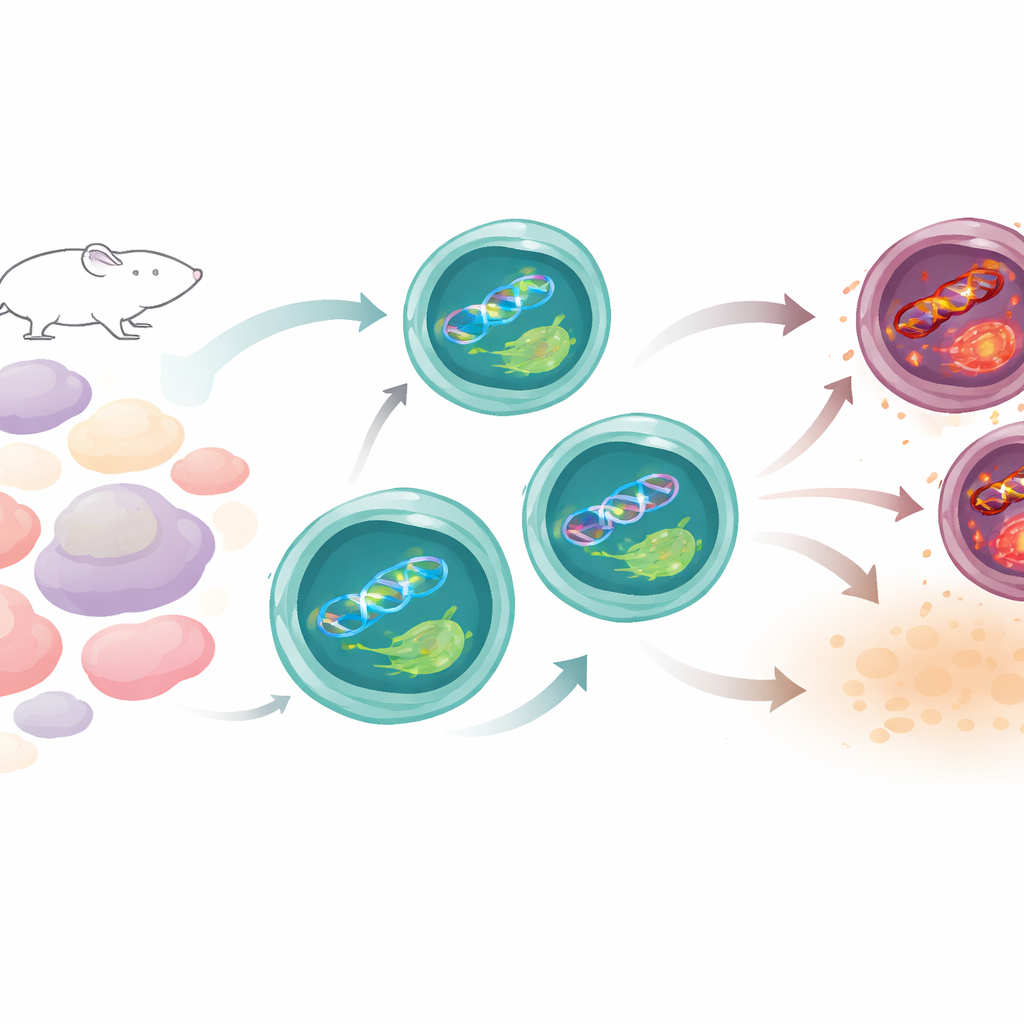
How Th17 Cells Normally Keep Balance
Th17 cells live a dangerous life. To function, they ramp up their metabolism and produce reactive oxygen species—chemically reactive molecules that can injure DNA. The team compared Th17 cells with other T cell types in mice and found that, despite their intense activity, Th17 cells accumulate less DNA damage than many of their cousins. A key clue was that genes involved in DNA repair, especially those from a pathway that usually fixes sun-induced lesions, were switched on more strongly in Th17 cells. Among these was XPC, a protein that scans DNA for structural damage. As Th17 cells matured, XPC levels rose and localized to damaged sites in the nucleus, suggesting it was on constant patrol to maintain their genetic integrity.
What Happens When the DNA Sensor Is Missing
To see how important XPC really is, the researchers used mice engineered to lack this protein. At first glance, their immune systems looked mostly normal: overall T cell numbers and many T cell subtypes were unchanged. But when the team focused on Th17 cells, a striking defect emerged. Without XPC, far fewer cells produced the hallmark Th17 molecule IL-17, and key identity markers and signaling switches that drive the Th17 program were dialed down. In dishes, naive T cells from these mice struggled to become fully fledged Th17 cells. In a standard model where transferred T cells trigger intestinal inflammation, XPC-deficient T cells failed to induce colitis, reflecting a dampened inflammatory capacity. At the same time, regulatory T cells, which calm immune responses, became more abundant, hinting at a shifted balance between aggression and restraint.
DNA Damage, Energy Factories, and Chemical Stress
Digging deeper, the team found that Th17 cells without XPC carried more broken DNA strands and more oxidized DNA bases, a signature of attack by reactive oxygen species. Genes that usually sense DNA damage and halt the cell cycle were also reduced, suggesting a weakened alarm system. The cells’ energy metabolism was reshaped: they lost some of their ability to use quick-burning sugar pathways and became more dependent on their mitochondria, the tiny power plants inside cells. Yet these mitochondria leaked more reactive oxygen species and showed signs of strain, creating a vicious cycle of rising chemical stress and mounting DNA injury. Remarkably, treating the cells with an antioxidant restored IL-17 production and reduced DNA damage, directly linking oxidative stress to the observed functional defect.
A Team Effort in Repairing Damaged DNA
Beyond its classic role, XPC also worked hand in hand with another repair protein called OGG1, which recognizes a common oxidized DNA base. The researchers showed that these two proteins physically associate in Th17 cells, helping coordinate the removal of damaged bases. When XPC was absent, OGG1’s cutting activity actually increased, as if trying to compensate for the excess damage. But this overactivity occurred in a background of high oxidative stress and disrupted repair networks, and it was not enough to restore normal Th17 function. Genetic and structural analyses suggested that XPC acts as a hub connecting different repair pathways and redox control, ensuring that damage is not only detected but processed in a way that allows the cell to keep dividing and producing protective molecules.
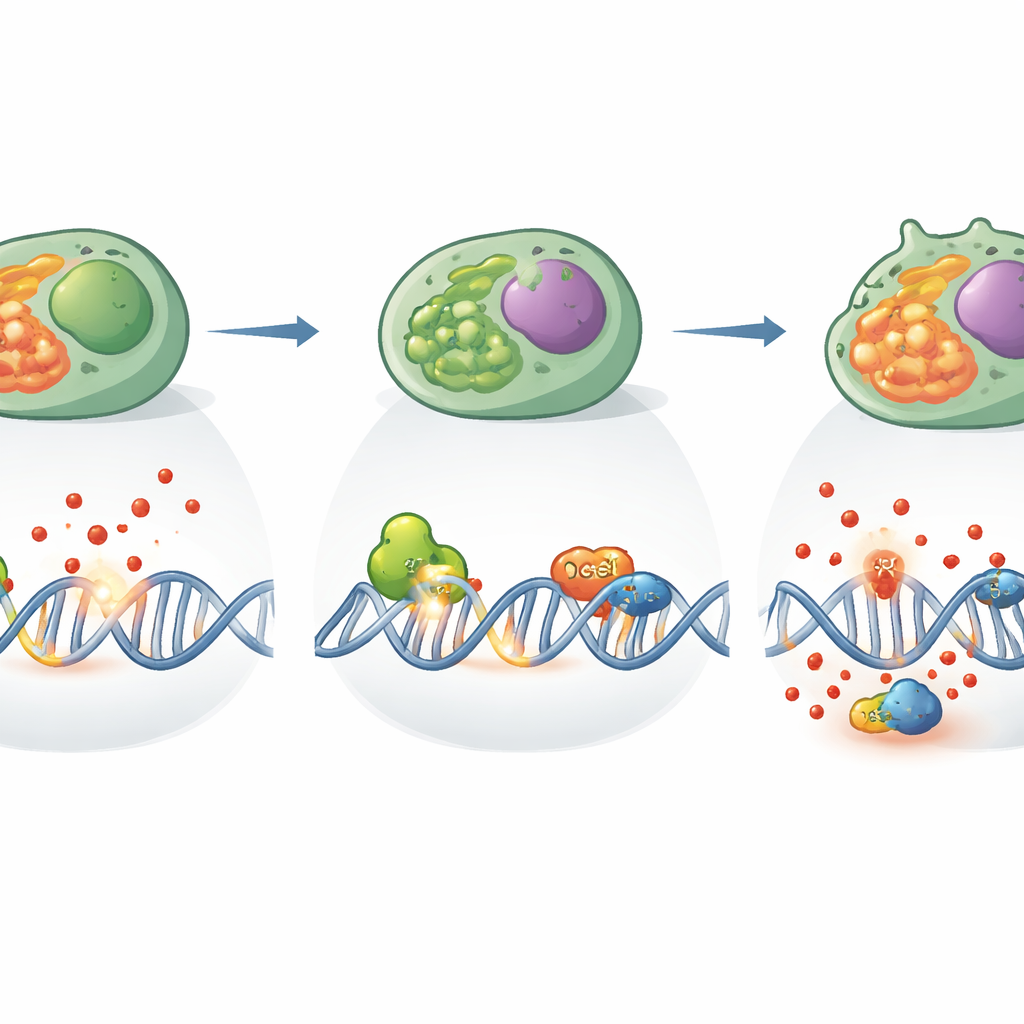
Why This Matters for Disease and Therapy
To a lay reader, the key message is that immune cells need their own internal bodyguards to stay fit while they protect us. This study reveals that XPC, previously famous for shielding skin cells from UV light, is also a vital guardian of Th17 cells, keeping their DNA intact and their energy systems in balance under oxidative stress. When XPC is missing, Th17 cells become genetically and metabolically fragile, produce less inflammatory output, and may shift the immune balance toward regulation rather than attack. Because Th17 cells are central in conditions such as inflammatory bowel disease, psoriasis, and multiple sclerosis, pathways controlled by XPC could become new targets for tuning immune responses—either calming harmful inflammation or boosting defense where it is too weak—by modulating how these cells repair their DNA and handle oxidative stress.
Citation: Leite, J.A., Bos, N.N., Menezes-Silva, L. et al. Th17 cells require the DNA repair sensor xeroderma pigmentosum complementation Group C to control oxidative DNA damage in a murine model. Nat Commun 17, 3157 (2026). https://doi.org/10.1038/s41467-026-69914-y
Keywords: Th17 cells, DNA repair, oxidative stress, XPC protein, autoimmune disease