Clear Sky Science · en
VAMP7-dependent late endosomal secretion of ER and mitochondrial proteins impacts the tumor microenvironment and macrophage engagement
How Stressed Cells Signal for Help
Cancer cells live hard lives. Starved of oxygen and nutrients, they accumulate damage in the tiny power plants (mitochondria) and protein factories (endoplasmic reticulum, or ER) inside them. This study uncovers an unexpected way in which cells under pressure jettison damaged pieces of these inner structures to the outside world. In brain tumors called glioblastomas, this cellular "trash export" system, driven by a protein named VAMP7, turns out to influence how immune cells sense and enter the tumor.
Hidden Shipping Routes Inside Cells
Most proteins that leave a cell use a classic route: they enter the ER, pass through a central sorting station called the Golgi apparatus, and are then packaged into small bubbles that fuse with the cell surface. But cells also rely on unconventional routes that bypass the Golgi. One such route uses late endosomes—internal sacs that can merge with the surface and release their contents outside. The authors focus on a molecular "zipper" protein, VAMP7, already known to help late endosomes fuse with the outer membrane and release small extracellular vesicles, tiny packages that carry cargo between cells.
Moving Worn-Out Parts to the Exit
Using engineered rat kidney and glioma cells lacking VAMP7, the researchers tracked what happens to proteins normally found in the ER and mitochondria. They showed that without VAMP7, cells released far less of a short form of an ER-shaping protein called RTN3 and of VDAC, a channel on the mitochondrial surface. At the same time, late endosomes became mispositioned and small vesicles rich in the marker CD63 were poorly secreted. High-resolution imaging and proximity assays revealed that RTN3 and mitochondrial material physically associate with CD63-positive late endosomes before being released, and that this handoff depends on VAMP7 and its partner fusion proteins. These findings support a model in which stressed ER and mitochondria bud off small vesicles that fuse with late endosomes, which then export their cargo out of the cell. 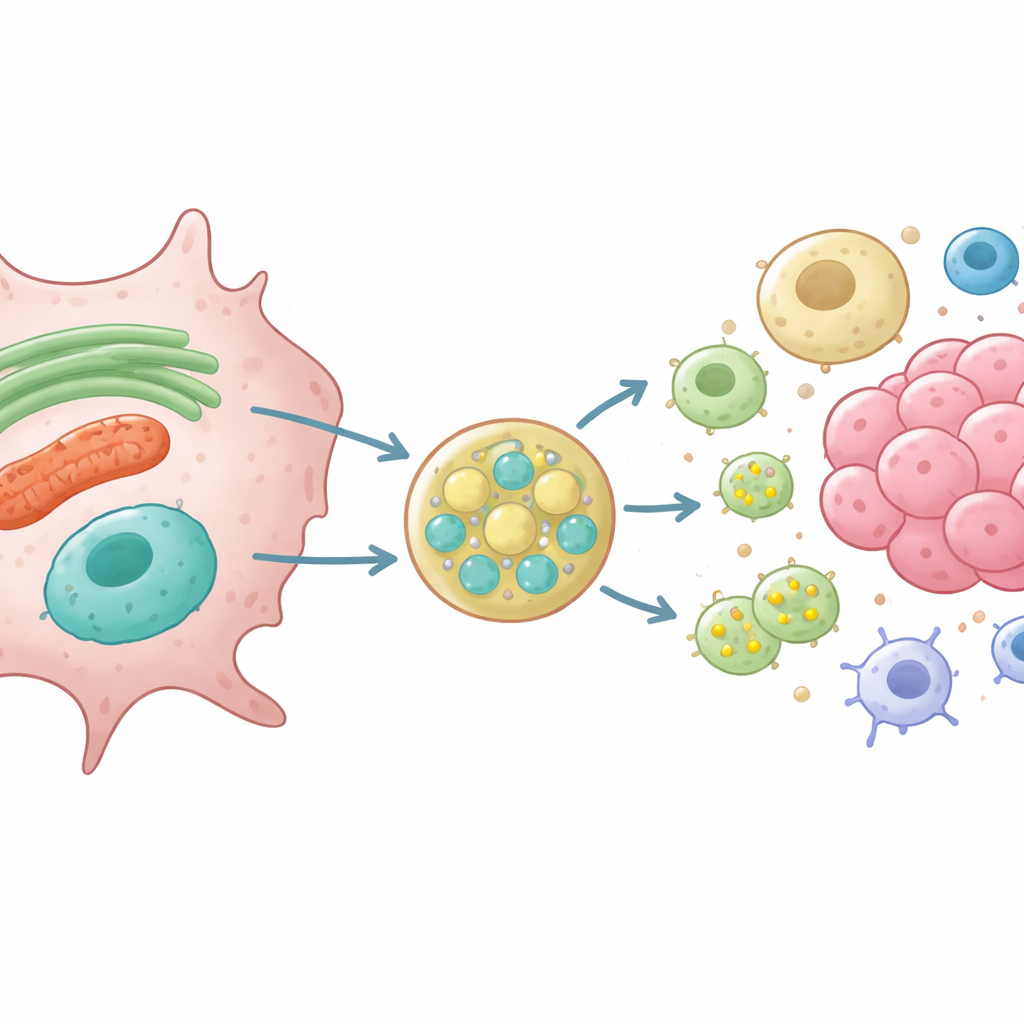
Stress Relief and Quality Control
Cells missing VAMP7 did not lose the general recycling pathway known as autophagy, but they did show clear signs of strain. Gene activity patterns pointed to activation of stress responses in the ER and changes in mitochondrial-related genes. Microscopy revealed abnormal "whorls" of ER membrane and excessively fused, elongated mitochondria, both hallmarks of organelles struggling to maintain balance. Measurements of oxygen consumption showed that, despite having more mitochondrial mass, cells lacking VAMP7 or a key autophagy protein (ATG5) had weaker energy production. When the scientists deliberately provoked ER or mitochondrial stress with drugs, cells with intact VAMP7 ramped up secretion of RTN3 or VDAC, respectively. This boost was lost when VAMP7 was removed, implying that late endosomal secretion acts as a pressure valve to help dispose of damaged components and broadcast the cell’s internal state.
Shaping the Tumor Neighborhood
To test the importance of this pathway in disease, the team turned to a rat model of glioblastoma, a highly aggressive brain tumor. They implanted normal, VAMP7-deficient, or ATG5-deficient glioma cells into rat brains. Tumors arising from VAMP7-lacking cells grew large and contained much more dead tissue, yet were infiltrated by fewer macrophages—the immune cells that engulf debris and can influence whether a tumor grows or stalls. In contrast, tumors lacking ATG5 were smaller overall but did not show the same selective drop in macrophage entry into the tumor core. Analyses of human brain tumor datasets further revealed that lower VAMP7 (and RTN3) expression is linked with poorer patient survival, suggesting that this stress-linked secretion route may naturally act to restrain tumor progression by shaping the immune landscape. 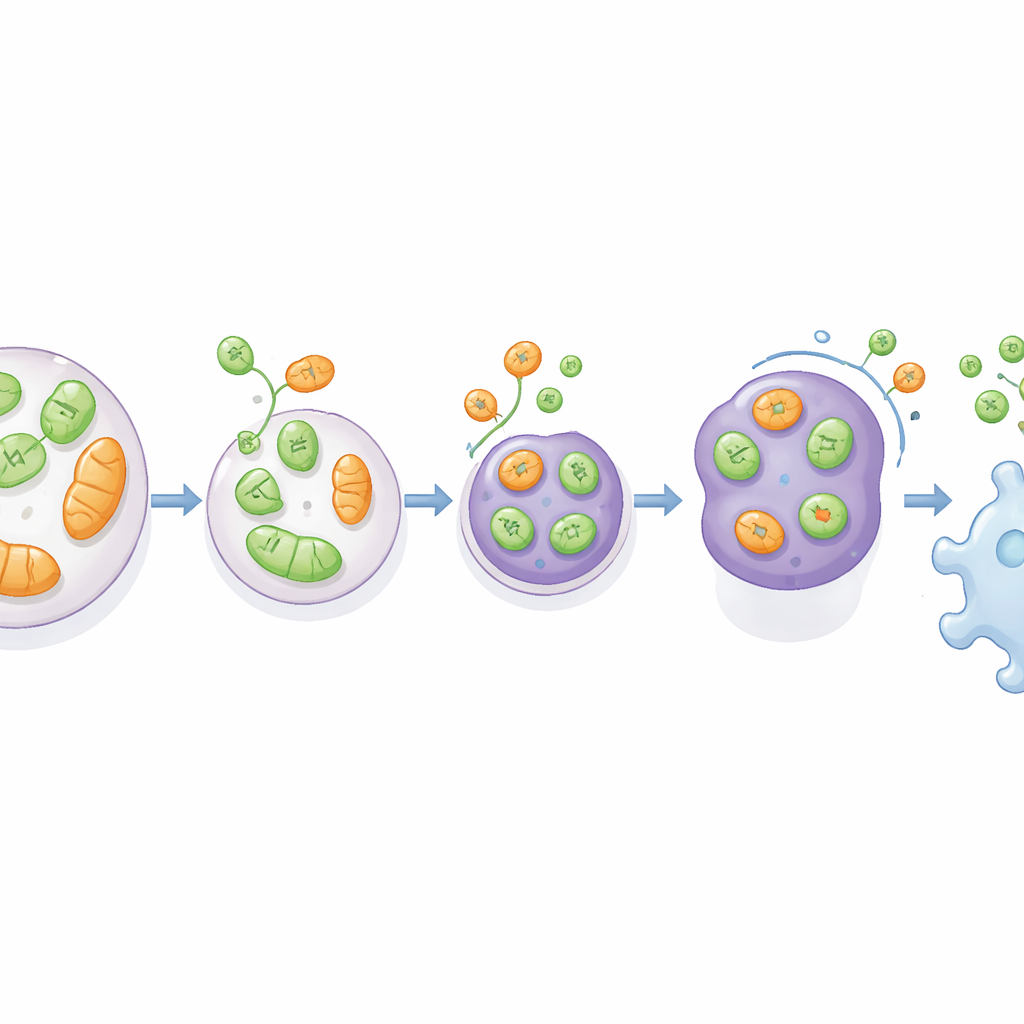
Why This Matters for Cancer and Immunity
Put simply, the study proposes that VAMP7 helps cells package and export damaged pieces of their internal machinery via late endosomes, especially under stress. In glioblastoma, this outward flow of ER- and mitochondrial-derived material appears to serve as a distress signal that encourages macrophages to enter the tumor and clear debris, contributing to a more tumor-suppressive environment. When VAMP7 is missing, the signal weakens, organelle damage accumulates, tumors become more necrotic, and macrophage engagement falls. Understanding and potentially tuning this secretory quality-control system could open new ways to make stubborn tumors more visible and responsive to the immune system.
Citation: Vats, S., Dionisio, P., Lemercier, Q. et al. VAMP7-dependent late endosomal secretion of ER and mitochondrial proteins impacts the tumor microenvironment and macrophage engagement. Nat Commun 17, 3012 (2026). https://doi.org/10.1038/s41467-026-69900-4
Keywords: unconventional protein secretion, VAMP7, glioblastoma, tumor microenvironment, extracellular vesicles