Clear Sky Science · en
Eukaryote initiation factor 6 modulates small-cell lung carcinoma plasticity via the integrin-FAK signaling axis
Why this lung cancer research matters
Small-cell lung carcinoma (SCLC) is one of the deadliest forms of lung cancer. It usually responds well to chemotherapy at first, but almost always comes back, quickly and aggressively. This study asks a central question: what allows SCLC cells to “change personality” and become drug-resistant, and can that shape-shifting be slowed or blocked?
Shape-shifting cancer cells
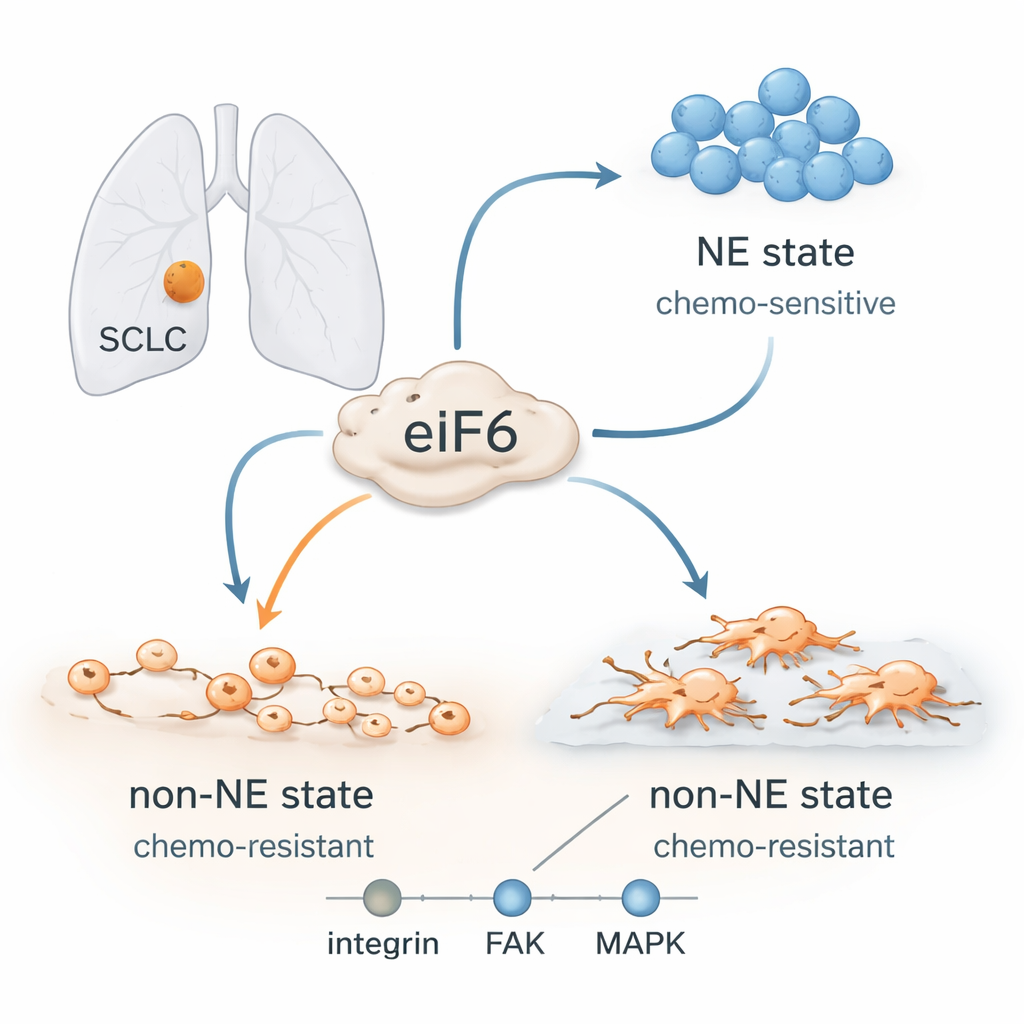
Doctors used to think SCLC was a uniform disease, but it is now clear that these tumors contain different cell types. Some cells have a strong “neuroendocrine” identity: they grow in tight floating clusters, respond well to platinum-based chemotherapy, and tend to be rich in certain nerve-like proteins. Others lose this identity and adopt a “non-neuroendocrine” state: they spread out, attach to surrounding tissue, show features of more mobile, invasive cells, and are much harder to kill with drugs. Patients’ tumors often evolve from the sensitive state to the resistant one during or after treatment, even though their DNA does not change much. That points to flexible cell programs rather than new mutations as the engine of relapse.
A translation factor with a double life
The authors homed in on a protein called eIF6, best known for helping assemble ribosomes, the cell’s protein factories. By studying patient samples, mouse models, and several human SCLC cell lines, they found that eIF6 levels consistently rose when cells shifted into the drug-resistant, non-neuroendocrine state. Surprisingly, this increase came alongside an overall drop in bulk protein production, hinting that eIF6 might be doing more than its textbook job. Detailed analyses of how messenger RNAs attach to ribosomes showed that the basic ribosome parts did not change much. Instead, the way messages were selected and processed for translation was remodeled as cells switched identities.
Breaking away to wire into survival signals
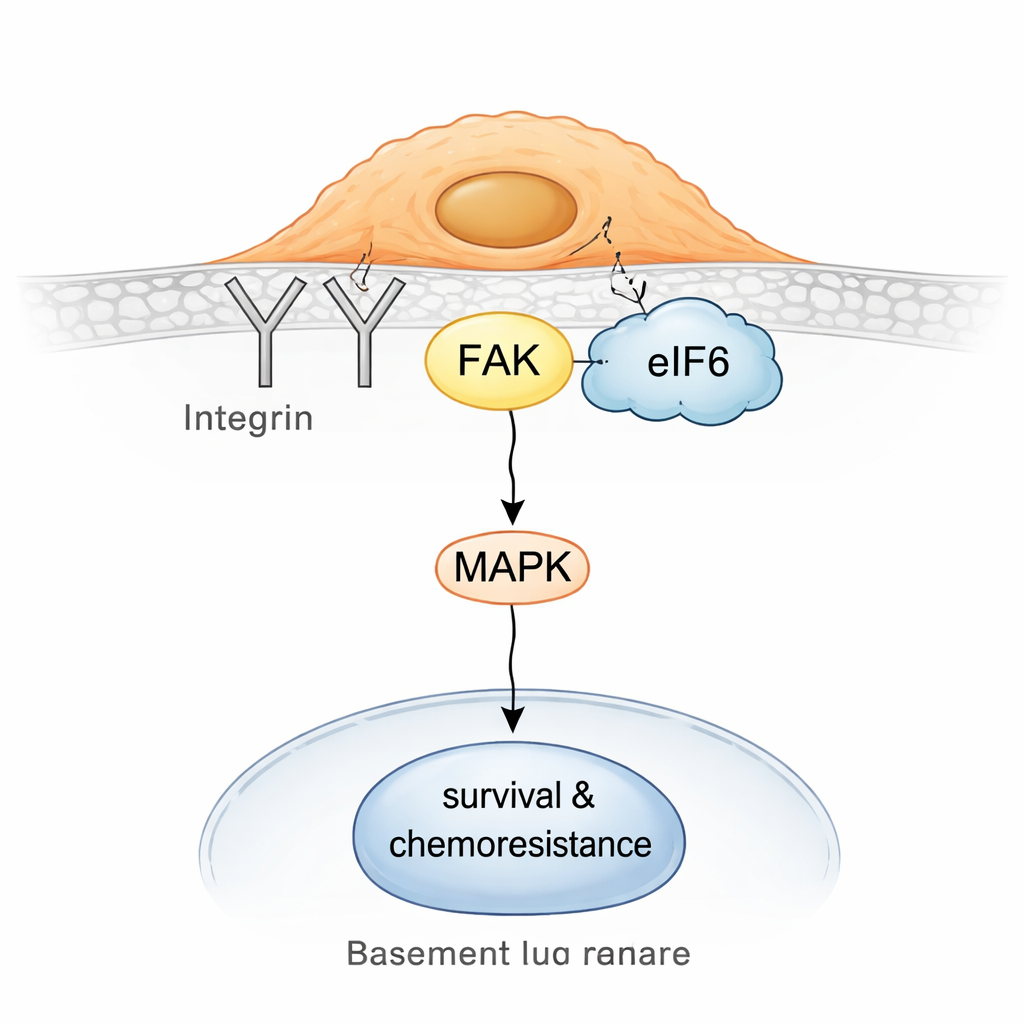
Digging deeper, the team discovered that in resistant cells eIF6 partly detaches from ribosomes and associates with a surface signaling hub built around integrins and a protein called focal adhesion kinase (FAK). Integrins help cells sense and cling to their surroundings; FAK relays those signals inward, often triggering pathways that support movement, survival, and drug tolerance. Biochemical pull-downs and in-cell proximity assays showed that eIF6 physically interacts with the integrin partner CD104 and with FAK, and that these complexes are more abundant in non-neuroendocrine cells. This eIF6–integrin–FAK assembly in turn boosts activity in the MAPK signaling pathway, a well-known driver of growth and stress resistance.
Making resistant tumors vulnerable again
The researchers then asked whether manipulating eIF6 could change how SCLC behaves. When they reduced eIF6 levels in resistant cells, genes linked to a mobile, scar-forming (epithelial–mesenchymal) program were dialed down, while classic neuroendocrine genes were partially restored. These changes did not fully turn back the clock, but they slowed the transition toward the resistant state. Crucially, cells with less eIF6 became more sensitive to standard carboplatin–etoposide chemotherapy in culture and in mouse tumors, leaving fewer surviving colonies and more lasting shrinkage. Because eIF6 itself currently lacks specific drugs, the team tested FAK inhibitors that are already in clinical development. Blocking FAK signaling re-sensitized resistant cells to chemotherapy and, in mice, the combination treatment produced the strongest tumor control.
From laboratory insight to future patient tools
Finally, the authors examined tumor samples from patients with limited-stage SCLC who had undergone surgery. Using a sensitive staining method that lights up proteins only when they are close together, they measured how often eIF6 sits in complex with CD104 or FAK inside tumor cells. High levels of these complexes—more so than eIF6 alone—were linked to earlier recurrence and shorter survival, marking them as promising prognostic markers. Taken together, this work reveals that eIF6 can “moonlight” away from ribosomes to help wire cancer cells into powerful survival circuits. By mapping this eIF6–integrin–FAK axis, the study suggests ways to identify patients at higher risk of relapse and supports combining FAK inhibitors with chemotherapy to counteract SCLC’s dangerous plasticity.
Citation: Peng, H., Wang, Z., Wang, M. et al. Eukaryote initiation factor 6 modulates small-cell lung carcinoma plasticity via the integrin-FAK signaling axis. Nat Commun 17, 2048 (2026). https://doi.org/10.1038/s41467-026-69899-8
Keywords: small-cell lung cancer, drug resistance, cell plasticity, eIF6, integrin FAK signaling