Clear Sky Science · en
COMPASS subunit Bre2 regulates chromatin remodeler Arp9 to control Aspergillus flavus aflatoxin synthesis and virulence
Why a Food Mold Matters to Your Health
Aflatoxins are poisons made by certain molds that grow on crops such as peanuts and corn. The most dangerous of these, aflatoxin B1, is a powerful liver carcinogen that can contaminate food and feed. This study uncovers how the common crop-infecting fungus Aspergillus flavus turns on the machinery that makes aflatoxin and boosts its ability to infect plants and animals, pointing to new ways to prevent contamination before it reaches our plates.
A Hidden Control Switch Inside Fungal DNA
Inside every A. flavus cell, long strands of DNA are wrapped around proteins to form chromatin, which acts like a gatekeeper for gene activity. The authors focus on a protein called Bre2, part of a larger COMPASS complex that places small chemical marks on histone proteins, especially at a position known as H3K4. These marks do not change the DNA sequence itself, but they influence whether genes are switched on or off. The team shows that Bre2 is crucial for adding activating marks (H3K4me2 and H3K4me3), and that removing the bre2 gene sharply reduces these signals at key regions of the fungal genome.
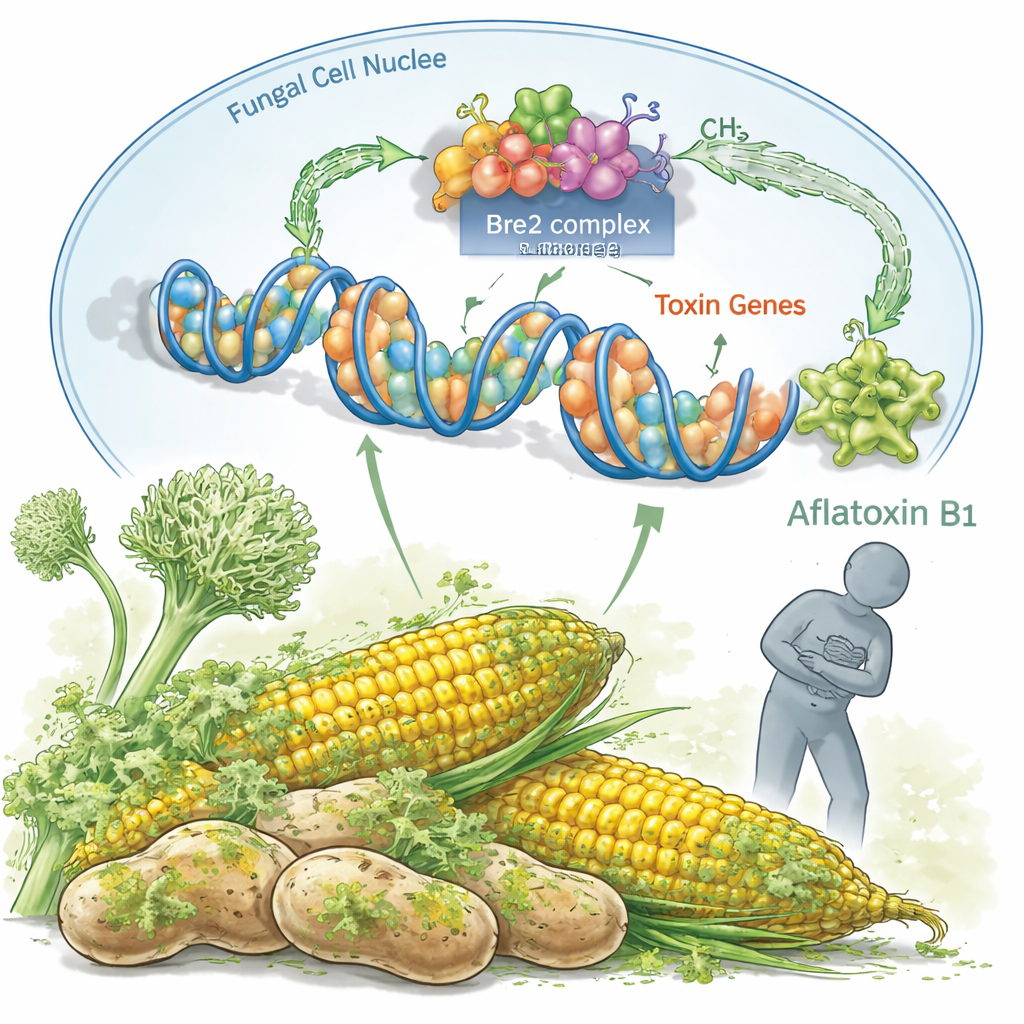
From Gene Marks to Mold Growth and Toxin Production
When Bre2 is deleted, the fungus becomes sluggish and less capable of reproducing. Colonies grow to less than half their normal size, and the number of spores, which spread the fungus, drops dramatically. The fungus also fails to form tough resting structures called sclerotia, which help it survive in soil and on stored crops. At the same time, production of aflatoxin B1 plummets, as measured by chemical tests. The study links this drop in toxin output to weaker activity in the aflatoxin gene cluster, a group of about 30 tightly coordinated genes. Without Bre2, the master switches aflR and aflS and several toxin-building genes are turned down, so the fungus simply cannot make as much poison.
Weaker Infection of Crops and Insects
The consequences of losing Bre2 extend from the lab dish into living hosts. When researchers infected peanut and maize kernels with the bre2-deficient fungus, they observed much sparser fungal growth and far fewer spores on the grain surface. The kernels also contained far less aflatoxin B1. Similar effects appeared in animal models: insect larvae infected with the mutant fungus survived longer and carried fewer spores and less toxin than those exposed to normal A. flavus. The work further shows that fungal genes linked to breaking down host defenses are less active without Bre2, suggesting that this protein helps the pathogen both grow and evade immunity in its hosts.
A Partnership with a Chromatin Remodeler
To understand how Bre2’s histone marks are translated into broad changes in gene activity, the authors looked for downstream players. Using genome-wide mapping, they found that Bre2 strongly marks and boosts expression of a gene called arp9, which encodes a component of a chromatin remodeling complex. This Arp9 protein helps slide and loosen nucleosomes, opening up stretches of DNA so genes can be read. When arp9 is deleted, A. flavus shows many of the same defects seen in the bre2 mutant: poor growth, low spore counts, lack of sclerotia, and nearly no aflatoxin production on media and crop kernels. Detailed protein studies reveal that Arp9 works together with other remodeler subunits (including RSC8, Arp7, and Sth1) to reshape chromatin.
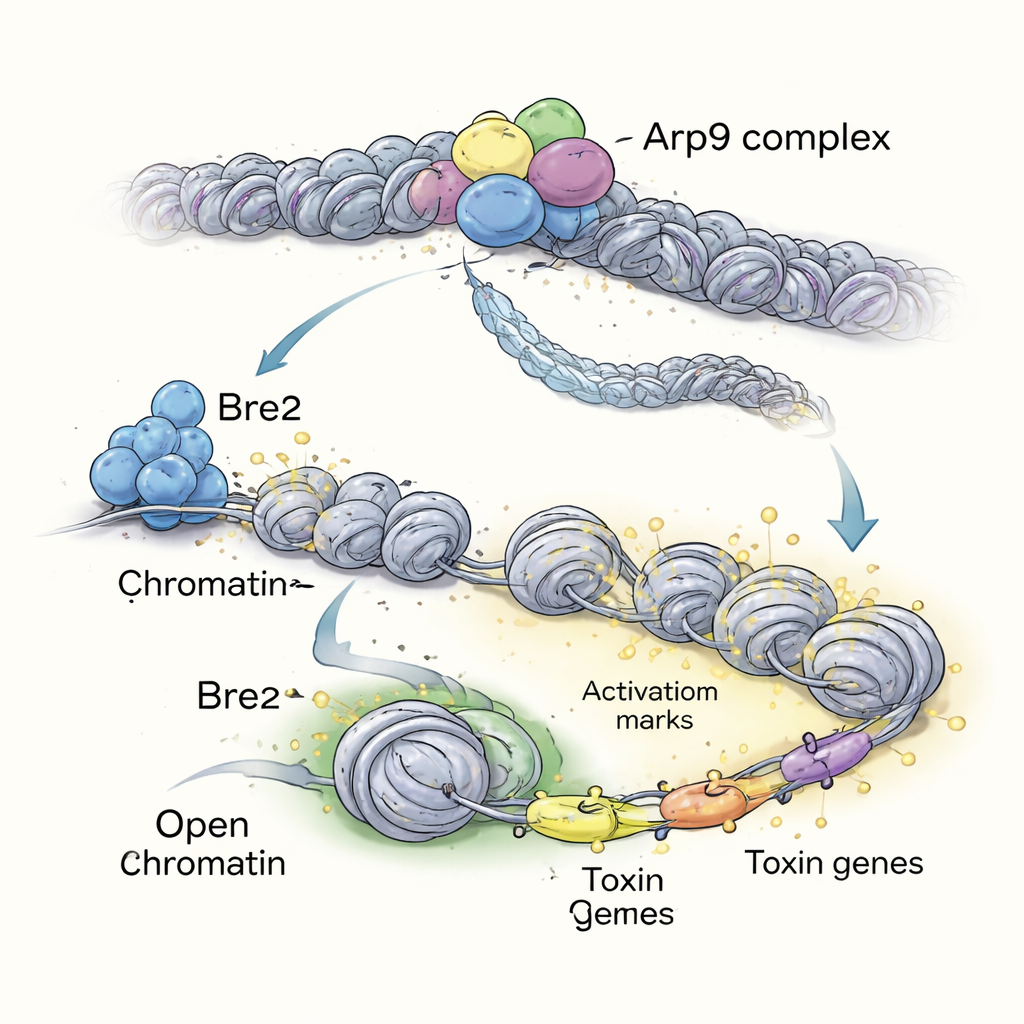
Downstream Genes That Tune Virulence and Toxins
By probing how accessible different DNA regions are in fungi lacking Arp9, the researchers identified more than 200 genes that depend on this remodeler to stay active. Many of these genes are involved in metabolism, stress responses, and the production of secondary chemicals like toxins. The team then deleted a panel of Arp9-controlled genes one by one. Several, including a membrane protein called SMAP and transporters such as CDR1 and UMF1, turned out to be important for colony growth, spore formation, sclerotia development, and aflatoxin B1 synthesis. In some mutants, toxin production was completely blocked. This places Bre2 at the top of an “epigenetic” pathway: Bre2 marks chromatin, activates arp9, and Arp9 in turn opens chromatin at many target genes that fine-tune fungal virulence and toxin output.
What This Means for Safer Food
In plain terms, the study maps a chain of command inside A. flavus that controls how aggressively it grows and how much aflatoxin it makes. Bre2 acts as an on-switch by decorating histones, which activates Arp9 and its partners to open or close stretches of DNA linked to development and toxin genes. Disrupting this Bre2–Arp9 axis, or some of its key targets, sharply weakens the fungus and nearly shuts down aflatoxin production. These insights suggest that future antifungal treatments or crop-protection strategies could aim at epigenetic and chromatin-remodeling components to keep this dangerous mold in check before it contaminates food.
Citation: Zhuang, Z., Sun, M., Wu, D. et al. COMPASS subunit Bre2 regulates chromatin remodeler Arp9 to control Aspergillus flavus aflatoxin synthesis and virulence. Nat Commun 17, 1862 (2026). https://doi.org/10.1038/s41467-026-69877-0
Keywords: aflatoxin, Aspergillus flavus, epigenetics, chromatin remodeling, mycotoxin control