Clear Sky Science · en
Identification of ephrin-A1–EphA2 signalling as a potential target for fracture prevention
Why protecting fragile bones matters
As people age, their bones can become thin and brittle, turning everyday mishaps like tripping on a rug into serious fractures. These osteoporotic fractures are not only painful but also costly for health systems and can steal years of healthy life. Current medicines help, but they are usually reserved for people with established osteoporosis and can carry rare but worrying side effects. This study asks a forward‑looking question: can we use genetics and protein measurements in large populations to uncover entirely new ways to strengthen bone and prevent fractures before they happen?
Hunting for fracture clues in the bloodstream
The researchers began by treating the body’s circulating proteins as a giant clue book. Many drugs already work by targeting proteins in the blood, so finding proteins that truly influence fracture risk could point directly to new therapies. Using samples from the UK Biobank, they looked at genetic variants that raise or lower the levels of 1,615 different blood proteins. They then linked these genetic “protein fingerprints” to data from a huge genetic study of more than 50,000 forearm fracture cases. A statistical method called Mendelian randomization helped them ask: when people are born with genes that push a protein level up or down, does their lifelong fracture risk shift in a consistent way?
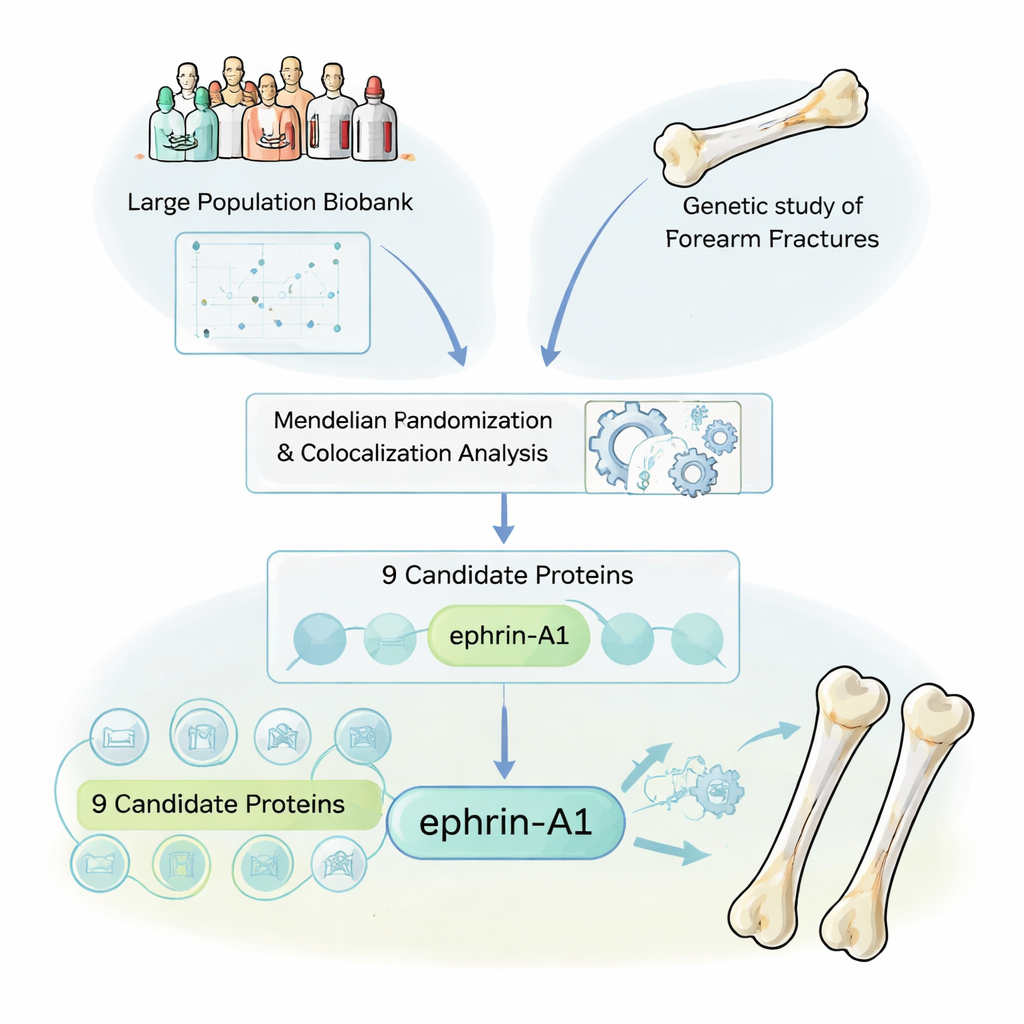
Separating real signals from genetic noise
Because nearby genes often travel together, the team used a second layer of analysis, called colocalization, to ensure they were seeing true cause‑and‑effect rather than misleading genetic neighbors. Out of the 1,615 proteins, nine stood out as being causally linked to forearm fractures. Importantly, several of these were already known bone actors: sclerostin and osteoprotegerin, which are targeted by existing osteoporosis drugs, and other proteins long tied to bone mineral density. Their re‑discovery provided a crucial quality check, showing that the pipeline could reliably rediscover proven drug targets while also bringing fresh candidates into view.
A surprising new protector: ephrin-A1
Among the newly highlighted proteins, one in particular drew attention: ephrin‑A1. People who were genetically predisposed to have higher blood levels of ephrin‑A1 had denser bones and fewer forearm fractures. Ephrin‑A1 is normally attached to cell surfaces and can be shed into the bloodstream. It binds to a receptor called EphA2, which the team found is highly expressed on bone‑forming cells called osteoblasts. By examining both human and mouse tissues with single‑cell sequencing, in situ RNA imaging, and an advanced 3D “DeepBone” clearing technique, they saw a consistent pattern: ephrin‑A1 sits on the lining cells of blood vessels inside bone, while EphA2 sits on osteoblasts along the bone surface, often just a few cell diameters away. This arrangement makes direct cell‑to‑cell communication between vessel cells and bone‑forming cells not only possible but likely.
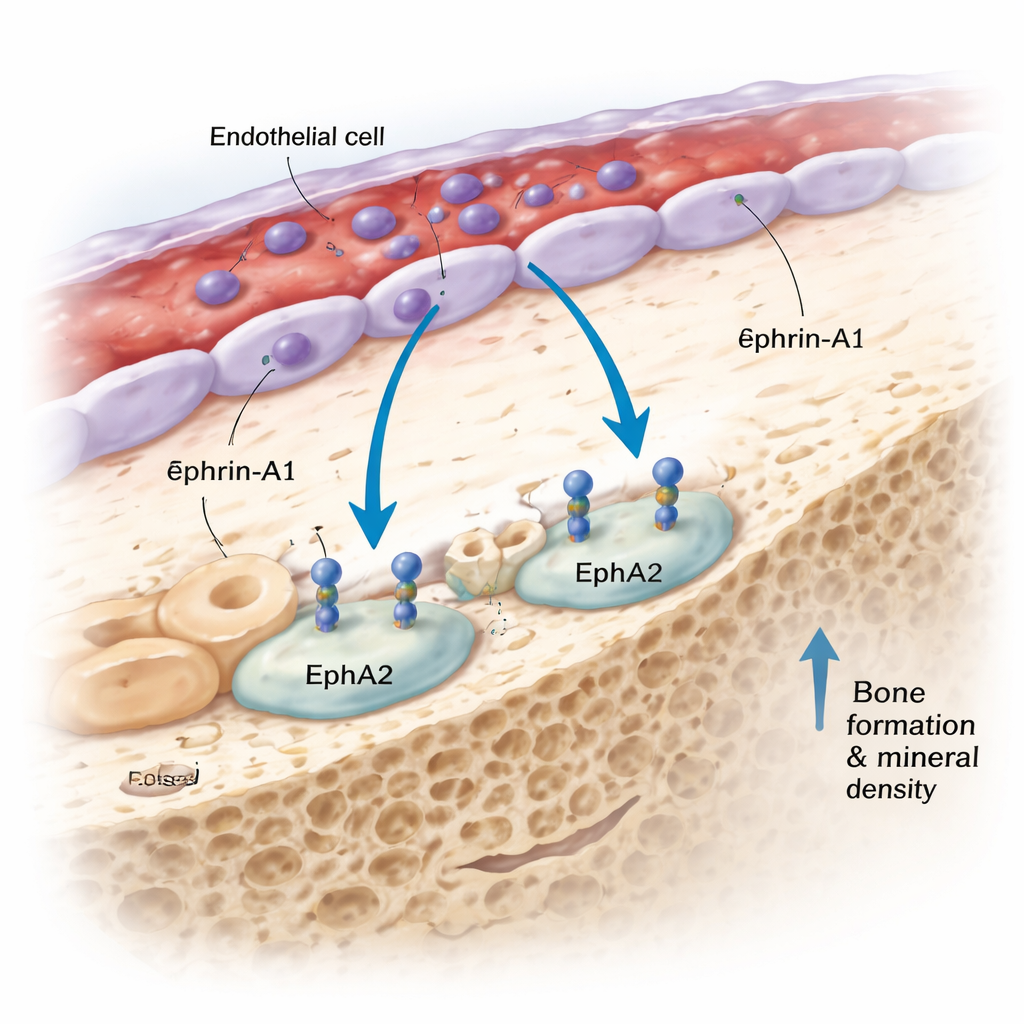
Testing the signal in living bones
To find out whether this ephrin‑A1–EphA2 conversation truly matters for bone strength, the researchers turned to mouse genetics. Mice engineered to lack either ephrin‑A1 or its receptor EphA2 had significantly lower total body bone mineral density than their normal littermates, in both sexes. The team then looked at how this signaling pair behaved in different mouse models that mimic human bone loss and gain. Inflammation, aging, and high‑dose vitamin A—each known to weaken bone—were all associated with reduced expression of ephrin‑A1 and/or EphA2 in bone tissue. By contrast, mechanical loading, which mimics the beneficial effect of weight‑bearing exercise, boosted ephrin‑A1 expression. Notably, these changes did not mirror those seen with current osteoporosis drugs, hinting that ephrin‑A1–EphA2 may operate through a distinct route that could complement existing therapies.
What this means for future fracture prevention
Altogether, the study shows that a sophisticated genetics‑plus‑proteins approach can rediscover known bone drug targets and uncover new ones. It points to ephrin‑A1 interacting with EphA2 on osteoblasts as a key signal that helps keep bones dense and resilient. For a lay reader, the take‑home message is that our blood and genes hold detailed blueprints for how bones stay strong—and that ephrin‑A1 is now a promising new switch in that blueprint. If future work can safely turn this switch up, perhaps using drugs that enhance ephrin‑A1–EphA2 signaling, it may offer a new, potentially safer way to prevent fractures, especially in the many people who are currently left untreated.
Citation: Movérare-Skrtic, S., Nethander, M., Li, L. et al. Identification of ephrin-A1–EphA2 signalling as a potential target for fracture prevention. Nat Commun 17, 1988 (2026). https://doi.org/10.1038/s41467-026-69863-6
Keywords: osteoporosis, bone mineral density, fracture prevention, ephrin-A1 EphA2, Mendelian randomization