Clear Sky Science · en
TRIM13 in situ engineering boosts anti-inflammatory capacity of CAR-Ms for liver fibrosis therapy
Reprogramming the Liver’s Cleanup Crew
Liver fibrosis, a scarring process that can lead to cirrhosis and liver failure, affects millions of people and currently has few effective treatments. This study explores a way to harness and reprogram the liver’s own immune cells so they not only remove scar‑forming cells but also calm harmful inflammation, potentially offering a safer and more powerful therapy for chronic liver disease.
Why Liver Scarring Is So Hard to Stop
When the liver is injured again and again—by viruses, alcohol, or metabolic disease—it tries to heal by laying down scar tissue. Specialized cells called hepatic stellate cells switch into an “activated” state and pump out tough fibers, much like builders pouring concrete. Over time this scarring, known as fibrosis, distorts the organ and impairs its function. Immune cells called macrophages sit at the center of this process. In a damaged liver, they can behave like a double‑edged sword: in one mode they drive inflammation and activate stellate cells, while in another, more healing mode, they help break down scar tissue and restore balance.
Turning Harmful Signals into Helpful Ones
The researchers focused on a key danger signal in damaged livers: mitochondrial DNA that leaks from injured cells. Macrophages sense this DNA through a pathway known as cGAS‑STING, which pushes them into a pro‑inflammatory, pro‑scarring state. A protein called TRIM13 normally helps rein in STING by tagging it for destruction. The team reasoned that boosting TRIM13 inside liver macrophages could flip them back toward a calming, tissue‑repairing mode. At the same time, they wanted these cells to directly hunt down and remove the scar‑forming stellate cells. To do that, they used the chimeric antigen receptor, or CAR, concept borrowed from cancer therapy: a synthetic receptor that allows immune cells to recognize a chosen target—in this case, a protein called FAP that is abundant on activated stellate cells.
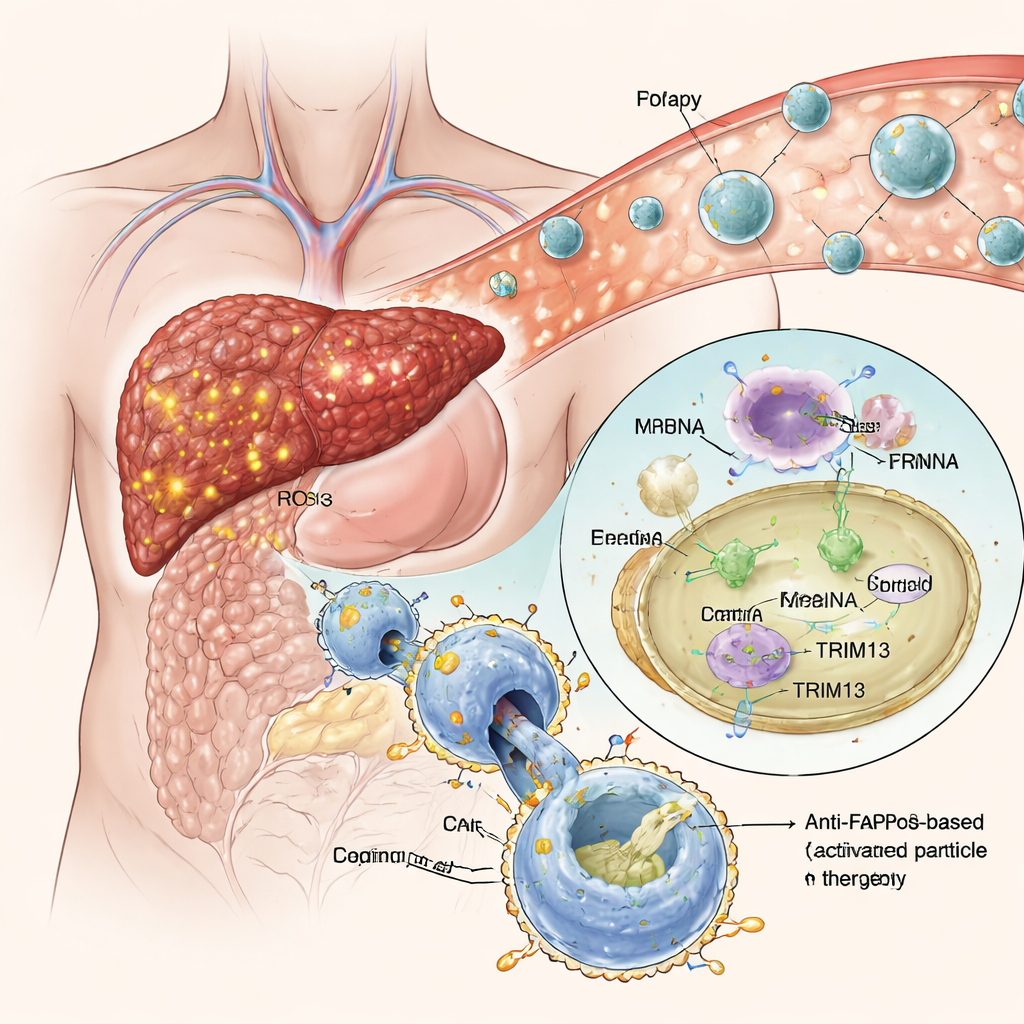
Smart Nanoparticles That Home to Diseased Tissue
Rather than engineering macrophages outside the body, the team built “efferocytosis‑sparked” lipid nanoparticles—tiny fat‑based spheres that carry genetic instructions as messenger RNA (mRNA). These particles are coated with a disguised form of phosphatidylserine, a natural “eat‑me” signal that dying cells display to attract macrophages. In the high‑oxidative environment of fibrotic liver tissue, the disguise is removed and the signal is revealed, prompting nearby macrophages to engulf the nanoparticles. Once inside, the mRNA is released and read by the cell, leading to production of both TRIM13 and the anti‑FAP CAR. In cell cultures, this approach successfully pushed macrophages toward an anti‑inflammatory state while giving them strong, selective appetite for FAP‑positive scar‑forming cells, without disturbing their normal ability to clear other debris.
From Diseased to Regenerating Livers in Mice
The team then tested this strategy in mouse models of liver fibrosis caused either by a toxic chemical or by a fatty, nutrient‑deficient diet. Systemically injected nanoparticles efficiently reached the liver and, especially in fibrotic animals, were taken up mainly by macrophages. There, the engineered cells showed reduced activity of the STING pathway and lower production of inflammatory molecules, along with enhanced killing of activated stellate cells. Tissue analyses revealed striking drops in collagen and other scar markers, finer and more normal extracellular matrix structure, and improved blood tests of liver function. The livers of treated mice showed more proliferating cells and regained healthier architecture, suggesting genuine organ repair rather than just a temporary dampening of inflammation.
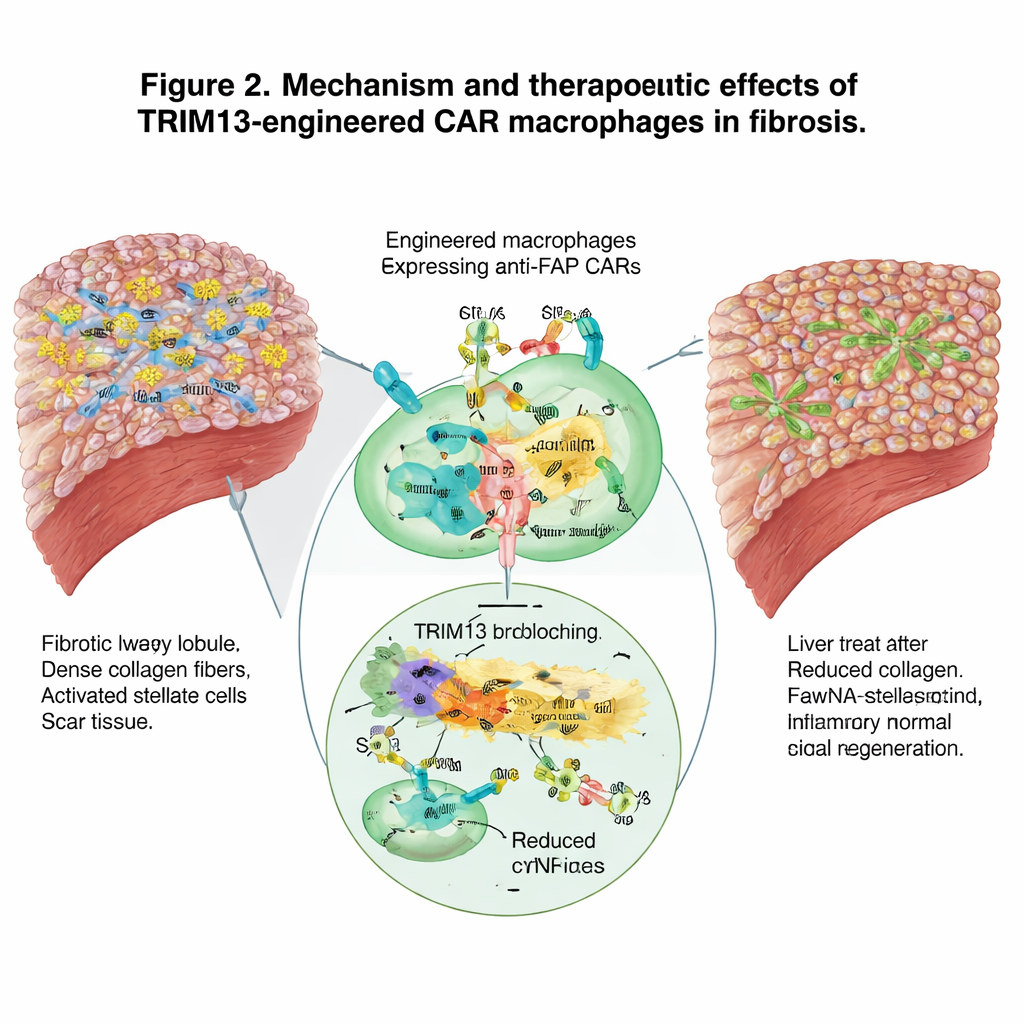
Enlisting the Wider Immune System
Beyond direct scar removal, the engineered macrophages reshaped the broader immune landscape. They shifted from an aggressive, damage‑promoting profile to a more resolving, healing one and reduced inflammatory signals in the blood. At the same time, by devouring scar‑forming cells, they appeared to act as teachers for the adaptive immune system, drawing in neutrophils, dendritic cells, and T cells and encouraging a coordinated response against the fibrotic tissue. Single‑cell RNA sequencing of liver immune cells confirmed a move away from pro‑inflammatory gene programs and toward enhanced capacity for engulfing and clearing harmful cells and debris.
What This Could Mean for Patients
To a lay reader, the takeaway is that this work points to a way of “re‑educating” the liver’s own garbage‑collecting cells so they both dismantle scar tissue and switch off the chronic alarm signals that keep damage going. By using short‑lived mRNA instructions packaged in smart nanoparticles, the therapy avoids permanently altering cells and limits activity to diseased regions, which may reduce side effects. While much more testing is needed before it could be tried in humans, this strategy outlines a promising blueprint for treating liver fibrosis—and potentially other fibrotic diseases—by turning the body’s own immune system from a driver of scarring into an active partner in healing.
Citation: Gao, J., Yang, Z., Song, Y. et al. TRIM13 in situ engineering boosts anti-inflammatory capacity of CAR-Ms for liver fibrosis therapy. Nat Commun 17, 2077 (2026). https://doi.org/10.1038/s41467-026-69858-3
Keywords: liver fibrosis, macrophage therapy, nanoparticle mRNA delivery, chimeric antigen receptor, TRIM13 STING pathway