Clear Sky Science · en
Maximizing energy utilization and lithium leaching efficiency via sequential electrochemical dual-oxidation and soaking-relaxation
Why old car batteries still matter
Lithium-ion batteries power our phones, laptops and, increasingly, our cars. But as millions of electric vehicle packs reach the end of their lives, they create a new kind of waste — and a new opportunity. The lithium and other metals locked inside spent batteries are valuable but energy‑hungry to recover. This study explores a smarter way to pull lithium back out using electricity more efficiently, cutting costs and environmental impact while keeping up with the world’s growing appetite for battery materials.
Turning waste batteries into a resource
Today, most industrial recycling of lithium-ion batteries relies on harsh chemicals or high‑temperature furnaces. These methods can recover metals, but they often waste energy and make it hard to separate lithium cleanly from nickel, cobalt, and manganese in so‑called NCM batteries. The researchers focused on a newer, cleaner route: using an electrical current in salty water to draw lithium out of the used cathode material. They asked a simple but crucial question: can we rearrange when and how we use electricity so that almost every watt helps remove lithium rather than being lost in side reactions?
A two-step dance: power, then quiet
The team designed a two‑stage process that combines an active “push” with a quiet “soak.” In the first stage, called electrochemical dual‑oxidation, a steady voltage is applied across a cell containing a spent NCM cathode and a sodium‑chloride solution. The current pulls lithium ions out of the solid and into the liquid while also creating powerful oxidizing species in the solution. The scientists found that most of the useful work happens in the first hour: lithium leaves the crystal structure quickly at the beginning, but later much of the power is wasted on side reactions such as bubbling oxygen gas.
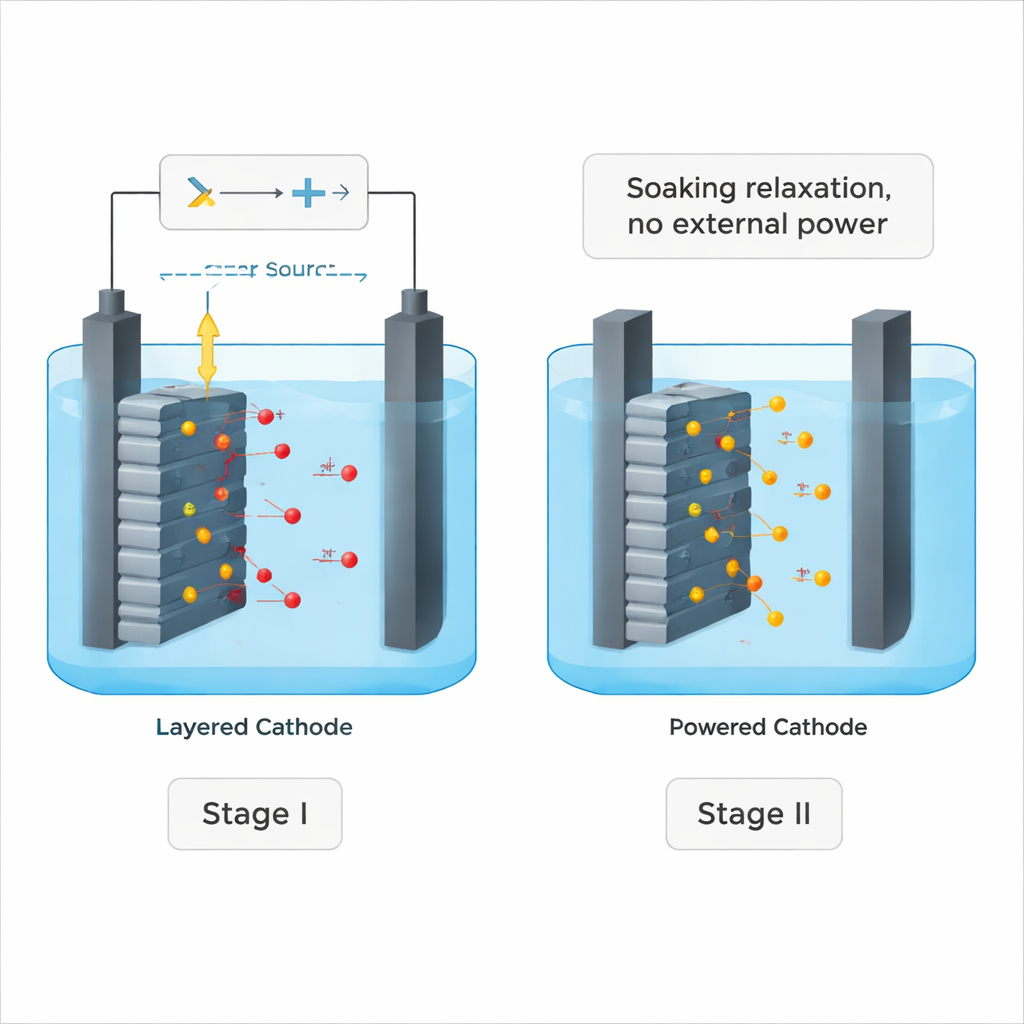
Letting chemistry finish the job
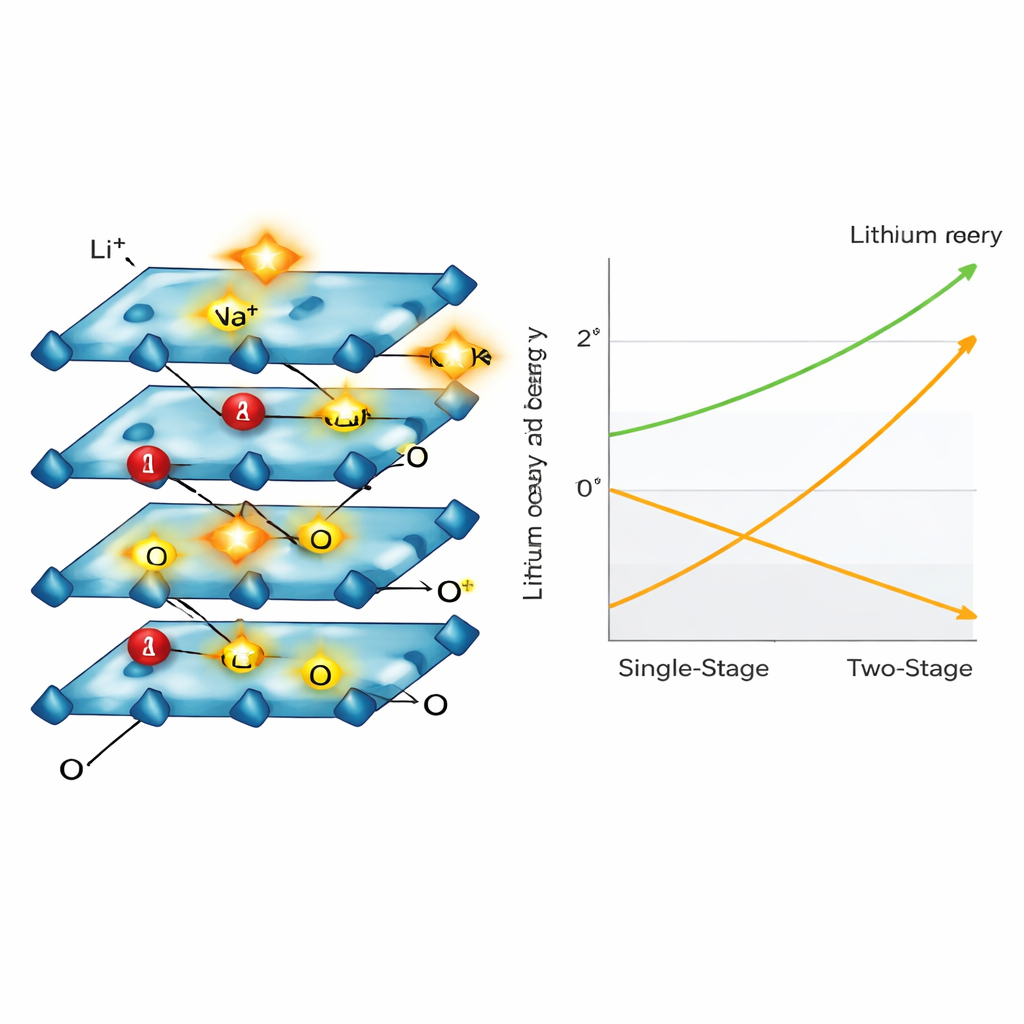
Instead of running the power longer and longer, the researchers simply switched it off and left the electrode soaking in the now‑oxidizing salt solution. Surprisingly, lithium continued to leach out until almost all of it was removed — reaching about 99% recovery for fresh NCM material and around 98% for real spent cathodes. Detailed measurements showed that oxygen atoms inside the crystal, temporarily pushed into a more reactive state during the powered stage, became the hidden driver in this second, quiet stage. These “activated” oxygen species promoted a slow swap: lithium ions diffused out into the liquid while sodium (or potassium) ions from the solution slipped into the empty spaces, all without further electrical input.
How the crystal structure rearranges
Using electron microscopes, X‑ray diffraction and spectroscopy, the team watched the cathode particles crack, thin and change their internal stacking as lithium departed. The material shifted through several known layered arrangements as it went from lithium‑rich to lithium‑poor, ending in a sodium‑rich form that held the structure together but no longer contained much lithium. Throughout this process, nickel and cobalt atoms changed their charge states to keep the overall material electrically balanced, while manganese stayed mostly unchanged, helping stabilize the framework. The researchers also showed that potassium ions, which shed their water molecules more easily than sodium, could speed up the ion‑swap step even further.
From lab bench to industry floor
To test whether this idea could work outside the lab, the team built a pilot system able to treat half a kilogram of real battery waste per batch. Using their two‑stage method, they recovered over 98% of the lithium as high‑purity lithium carbonate, suitable for making new batteries. Crucially, because the power is turned off once the “smart chemistry” is set in motion, the process used about half as much electrical energy as a standard one‑stage electrochemical method, saving more than a fifth of the total operating profit per ton of recycled cathode material.
What this means for future batteries
In plain terms, the study shows that we do not always need to keep pumping in electricity to recover valuable materials from old batteries. A well‑timed burst of power can prepare the material and the solution so that the rest of the work happens on its own, driven by internal chemical forces. If adopted at scale, this two‑stage approach could make recycling lithium from spent NCM batteries cheaper, cleaner and more attractive to industry, helping close the loop on the battery life cycle and easing pressure on new lithium mining.
Citation: Zhong, W., Gu, X., Feng, X. et al. Maximizing energy utilization and lithium leaching efficiency via sequential electrochemical dual-oxidation and soaking-relaxation. Nat Commun 17, 2050 (2026). https://doi.org/10.1038/s41467-026-69834-x
Keywords: lithium recycling, battery waste, energy efficient leaching, NCM cathodes, electrochemical recovery