Clear Sky Science · en
Survival of cyanobacteria and mitigation of Fe(II) toxicity effects in a silica-rich Archean ocean
Ancient seas and the air we breathe
Billions of years before animals or plants existed, tiny photosynthetic microbes called cyanobacteria began releasing oxygen into Earth’s oceans. Yet it took hundreds of millions of years before oxygen finally built up in the atmosphere. This study asks why that delay was so long and whether the chemistry of early seas—rich in dissolved iron and silica—helped or hindered those microbes that ultimately made our planet breathable.

Iron-rich oceans and a toxic side effect
Early oceans contained large amounts of dissolved iron, especially in coastal regions where deep waters welled up toward the surface. When this iron met the oxygen given off by cyanobacteria, it rusted, forming iron minerals that later became banded iron formations—striped rocks that are some of our oldest geological archives. But the same reactions can also create “reactive oxygen species,” highly aggressive forms of oxygen that can damage DNA, proteins, and cell membranes. Previous work suggested that this iron-driven chemical backlash might have poisoned cyanobacteria, slowing their spread and delaying the rise of oxygen in the air.
Silica as an unexpected bodyguard
The authors focused on another abundant ingredient of ancient seawater: dissolved silica, the same basic material found in glass. Geological evidence suggests that early oceans held silica at concentrations far higher than today. In laboratory experiments, they grew a marine cyanobacterium (Synechococcus sp. PCC 7002) under carefully controlled conditions with different amounts of dissolved iron and silica. They tracked how fast the cells grew, how much oxygen they produced, how quickly iron was oxidized, and how much reactive oxygen formed. At low iron levels, cells thrived regardless of silica. But when iron was very high—similar to conditions expected in some Archean coastal waters—cultures without added silica faltered, while those with high silica remained active, produced more oxygen, and reached larger cell numbers.
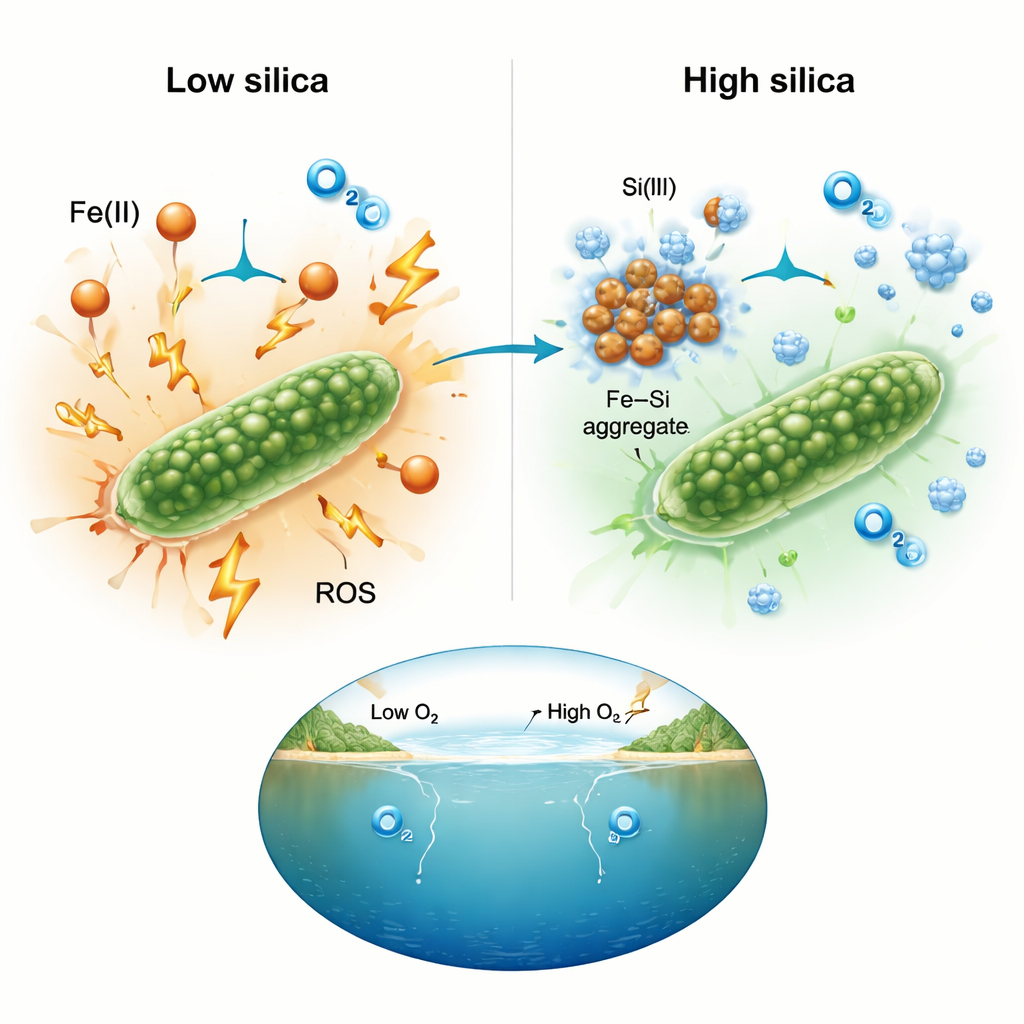
Chemical teamwork that tames harmful reactions
To understand why silica made such a difference, the team measured reactive oxygen directly using fluorescent dyes. At high iron levels, cultures lacking silica showed strong signals of reactive oxygen, consistent with stressful chemistry occurring around the cells. In silica-rich setups, those signals stayed close to background levels even when iron concentrations were extreme. The researchers interpret this as silica binding to dissolved iron and forming iron–silica aggregates. Once tied up in these clusters, iron is less available to fuel the chain reactions that generate damaging oxidants. The net effect is to soften the chemical environment so that cyanobacteria can keep photosynthesizing instead of being crippled by oxidative stress.
Day–night rhythms and ocean-scale impacts
The experiments also ran under realistic day–night light cycles rather than constant illumination. Under these alternating conditions, cyanobacteria oxidized iron faster and tolerated high-iron waters better, suggesting that nightly “rest” periods reduce long-term stress. Using measured oxygen production rates from the lab, the authors then built a simple numerical model of an early ocean water column. For plausible cyanobacterial abundances and realistic rates of deep-water upwelling, they found that surface waters could become oxygen-rich while deeper layers remained iron-laden. In many scenarios, oxygen concentrations in the sunlit zone reached or exceeded modern seawater saturation levels, implying that local oxygen “oases” above iron-rich depths could have been common.
Rethinking why oxygen took so long
Taken together, the results argue that reactive oxygen formed from iron and oxygen was probably not a show-stopping problem for early cyanobacteria, provided silica was abundant and natural day–night cycles prevailed. Instead of being chronically poisoned, these microbes likely found many coastal settings where silica buffered iron’s harmful side effects, allowing them to grow, oxidize vast amounts of iron, and leak excess oxygen into the atmosphere. The lingering mystery of why Earth’s atmosphere oxygenated so late must therefore lie more in large-scale factors—such as how quickly oxygen was consumed by volcanic gases and rocks—than in the local chemistry around individual microbial cells.
Citation: Dreher, C.L., Cirpka, O.A., Schad, M. et al. Survival of cyanobacteria and mitigation of Fe(II) toxicity effects in a silica-rich Archean ocean. Nat Commun 17, 1987 (2026). https://doi.org/10.1038/s41467-026-69826-x
Keywords: early Earth oxygen, cyanobacteria, banded iron formations, reactive oxygen species, silica-rich oceans