Clear Sky Science · en
Photocatalytic four-component reaction to access covalent organic frameworks for photocatalysis
Light-Powered Building Blocks
Chemists are constantly searching for cleaner, milder ways to make complex materials that can, in turn, drive useful reactions such as making drug-like molecules. This study introduces a way to use visible light—similar to the blue light in LED lamps—to gently stitch together small organic building blocks into highly ordered, sponge-like materials called covalent organic frameworks. One of these new frameworks then acts as a reusable catalyst, again under visible light, to assemble benzimidazoles, a family of ring-shaped molecules important in medicine.
Why These Sponge-Like Solids Matter
Covalent organic frameworks are crystalline networks made only from light elements such as carbon, nitrogen, and oxygen. They resemble rigid sponges with uniform pores, offering large internal surface areas and high stability. These features make them attractive for tasks ranging from gas storage and separation to catalysis and sensing. However, many common ways to make these materials require high temperatures and pressures, which limit the types of building blocks that can be used and make the process less environmentally friendly.
Bringing Four Pieces Together at Once
The team set out to merge two powerful ideas: multicomponent reactions, where several ingredients are combined in a single pot, and photocatalysis, where light provides the driving energy. Instead of the usual two- or three-component approaches, they developed a four-component route that joins together aromatic aldehydes, hydrazines, aromatic double bonds, and boronic acids. Under blue LED light and at room temperature, these four ingredients are guided by a separate organic photocatalyst into forming extended, highly ordered frameworks with permanent pores. This one-pot strategy allows four different kinds of building blocks to be woven into a single, well-defined architecture, greatly expanding the possible structures and functions of these materials.
Proving the New Frameworks Are Robust
To show that the new materials are genuinely ordered frameworks and not random polymers, the researchers used several characterization tools. X-ray diffraction revealed sharp patterns consistent with layers of hexagonally arranged pores stacked in an orderly fashion. Gas adsorption experiments showed that the pores are open and accessible, with a substantial internal surface area. Electron microscopy images confirmed a crystalline internal lattice, while thermal and chemical tests demonstrated that at least one framework, named Cp-tBu-N3-COF, withstands heating to about 200 °C and remains intact in both strong acid and base, as well as under prolonged light exposure. Measurements of light absorption and electrical behavior indicated that this framework behaves like an n-type semiconductor, able to separate and move charges when illuminated.
Using the Framework as a Light-Driven Catalyst
The authors then turned Cp-tBu-N3-COF from product into tool by testing it as a catalyst for forming benzimidazoles. They combined a simple diamine and an aldehyde in ethanol and shone blue light on the mixture in the presence of the framework. Under these mild conditions, the solid material converted the starting compounds into a benzimidazole in very high yield, and it did so repeatedly over at least five cycles with almost no loss of performance. Control experiments showed that removing the framework, the light, or oxygen essentially shuts down the reaction, pointing to a truly light- and framework-dependent process. By varying the aldehyde and diamine, the team prepared a wide range of benzimidazoles, showing that the method is broadly useful.
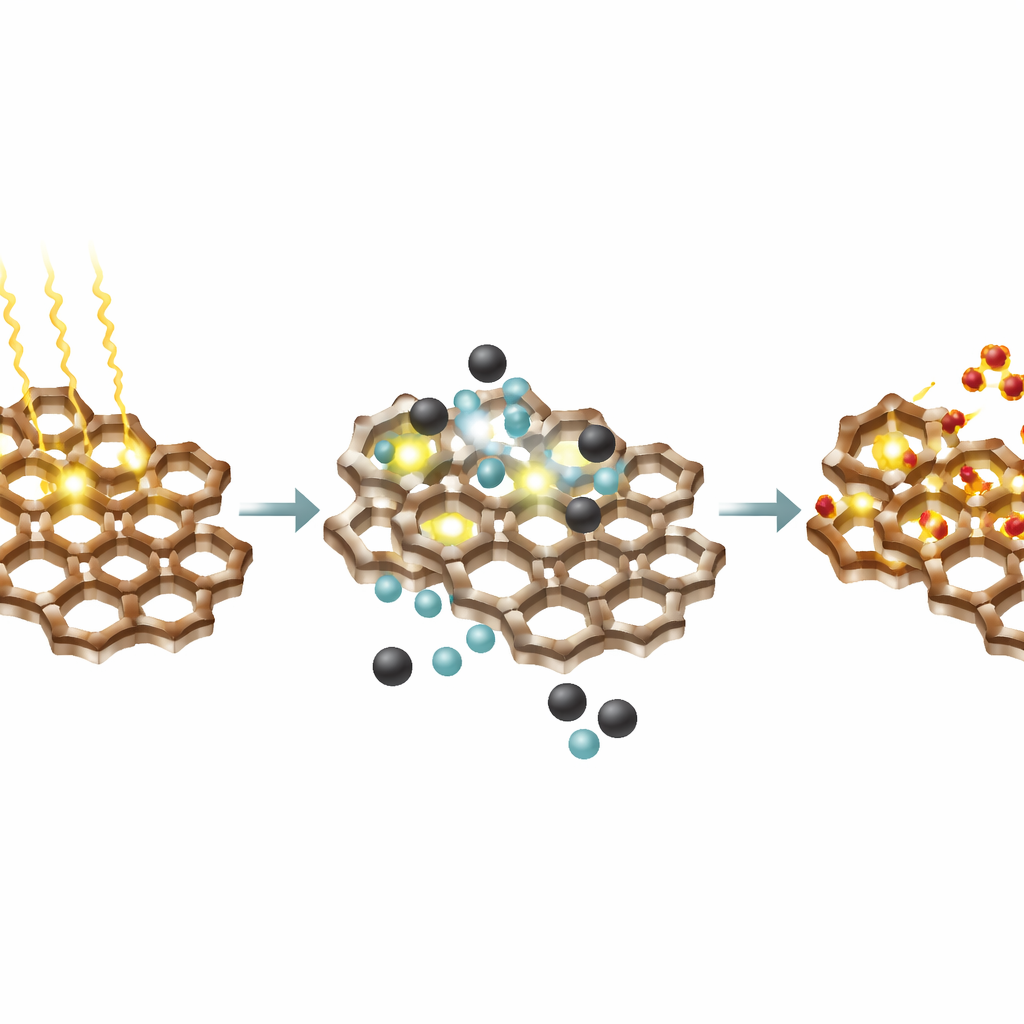
How Light, Oxygen, and the Framework Work Together
Mechanistic experiments and calculations suggest that, once light is absorbed, the framework transfers an electron to an intermediate formed from the starting molecules, and then passes the electron on to oxygen from the air. This step creates a reactive oxygen species—essentially an energized form of oxygen—that helps drive the final bond-forming and bond-breaking steps leading to the benzimidazole product, while the framework itself is regenerated. The internal arrangement of electron-rich and electron-poor regions in the framework appears to favor this light-triggered charge flow.
A Gentler Road to Designer Catalysts
In plain terms, this work shows that visible light can both build and power sophisticated porous materials under conditions gentle enough to tolerate delicate chemical groups. By combining four building blocks at once, the authors unlock far more design flexibility than traditional routes, while avoiding high heat and pressure. Their demonstration that one of these frameworks is an efficient, reusable catalyst for making medically relevant molecules highlights the promise of this strategy for creating next-generation, light-driven materials for green chemistry.
Citation: Wu, CJ., Li, TR., Liang, WJ. et al. Photocatalytic four-component reaction to access covalent organic frameworks for photocatalysis. Nat Commun 17, 3028 (2026). https://doi.org/10.1038/s41467-026-69824-z
Keywords: covalent organic frameworks, photocatalysis, multicomponent synthesis, visible light chemistry, benzimidazole formation