Clear Sky Science · en
Harnessing a germ‑free mouse gut bioreactor for directed evolution of probiotics to combat non-alcoholic fatty liver disease
Why this gut story matters for your liver
Non-alcoholic fatty liver disease (NAFLD) now affects hundreds of millions of people and is tightly linked to diet and gut health. This study explores an unusual idea: using the gut itself as a kind of living factory to “train” beneficial bacteria so they become better medicines. By letting probiotics evolve inside germ-free mice under a high-fat diet, the researchers shaped a bacterial strain that more efficiently handles bile acids—detergent-like molecules that connect what we eat to how our liver stores fat. The work suggests a new way to craft next-generation probiotics that are naturally tuned to our bodies rather than engineered only in test tubes.
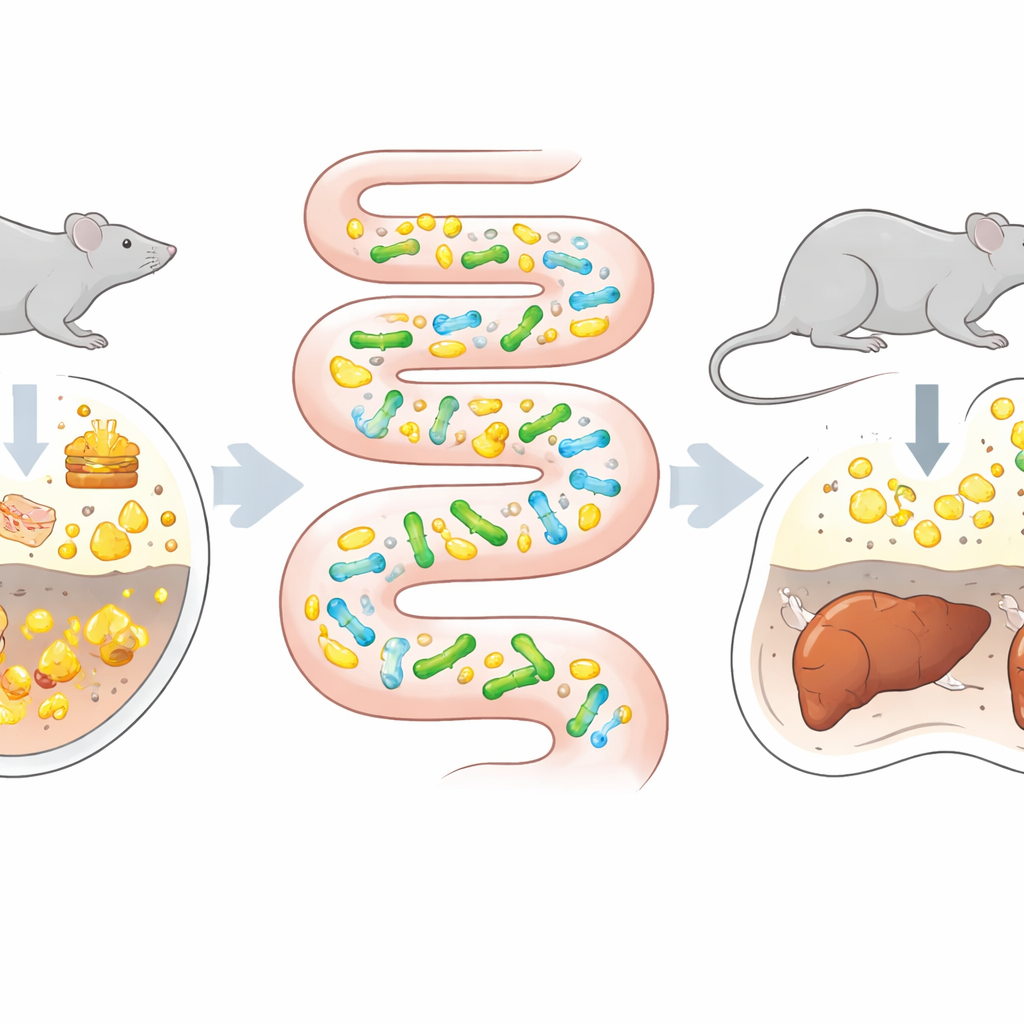
Turning the gut into an evolution chamber
Traditional directed evolution—mutating microbes and selecting the best performers—usually happens in lab flasks. That works well for enzymes or industrial microbes but often fails for probiotics, which must navigate the complex chemistry, immune signals, and physical forces of the intestine. The authors wondered: what if the gut itself, with all its natural pressures, were used as the selective environment? They chose a probiotic species, Bifidobacterium animalis subsp. lactis, that already has some ability to break down bile acids. Germ-free mice, which carry no other microbes, were colonized with this strain and then fed a stepwise higher-fat, high-cholesterol diet, known to boost bile acid levels in the intestine. In parallel, the same strain was pushed to adapt in standard lab medium containing bile acids, allowing a head-to-head comparison of in vitro versus in vivo evolution.
A gut-trained probiotic outperforms its lab-trained cousin
After repeated passaging in flasks, the lab-evolved bacteria showed no meaningful improvement in bile acid–processing activity. In striking contrast, isolates taken from the guts of the high-fat-fed mice displayed a wide spread in performance; about a quarter had clearly stronger bile-acid–breaking power. The standout variant, named W5S9, metabolized bile acids 77 percent better than its parent strain. This diversity, and the presence of both winners and losers, highlighted how the host’s gut environment applies rich, multifaceted pressures that simple lab media cannot mimic. It also confirmed that the intestine can act as a powerful “bioreactor,” continuously generating and testing genetic variants in conditions that closely resemble real life.
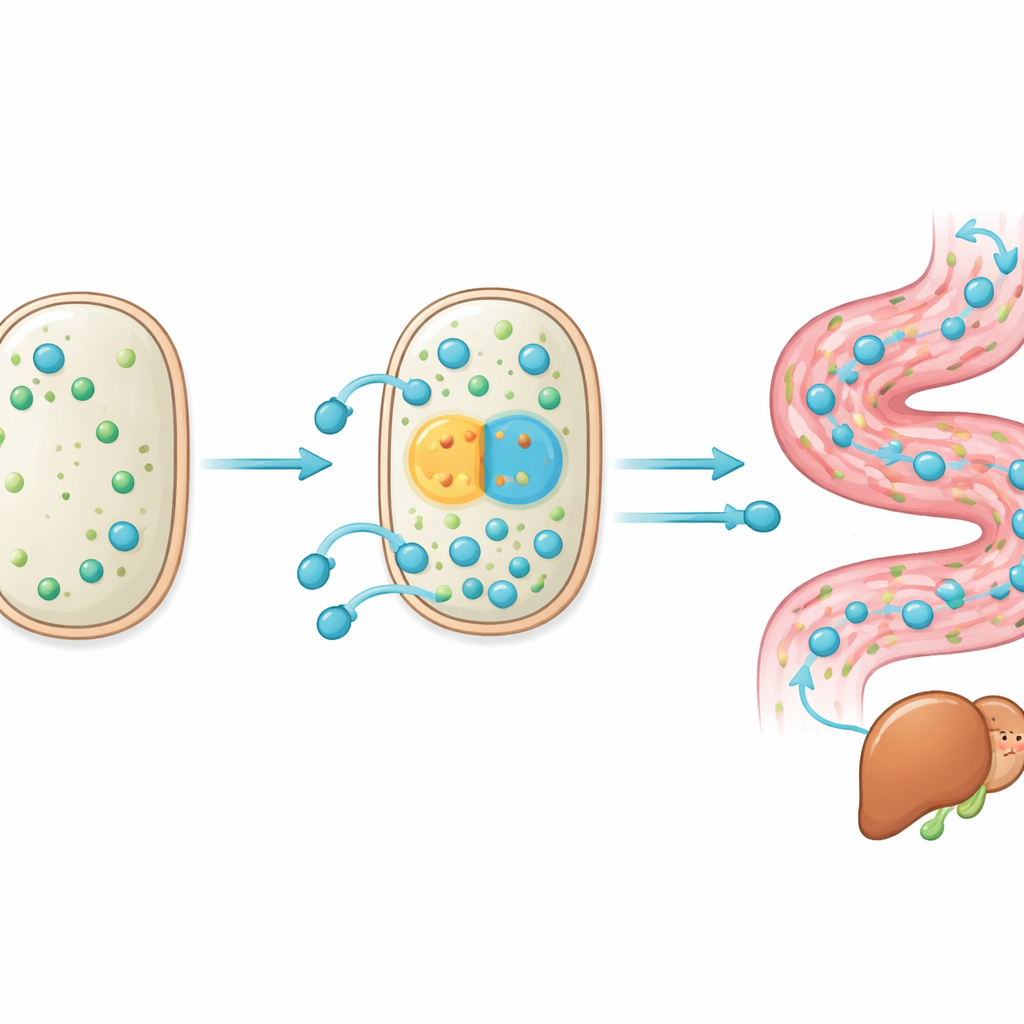
Zooming in on the mutations that matter
To understand what changed inside W5S9, the team sequenced its genome and measured which genes were more or less active compared with the original strain. Among hundreds of small DNA differences, two stood out. One lay just upstream of a gene called cbh, which produces a bile salt–cutting enzyme; this change acted like a stronger on-switch, boosting production of the enzyme under bile stress. The second mutation altered the structure and activity of a transporter protein (MDR) that pumps processed bile acids out of the cell. Lab tests showed that these tweaks made the probiotic better at both chopping conjugated bile acids and exporting the resulting products, improving its survival in harsh bile-rich conditions. In other words, evolution inside the gut fine-tuned both the “scissors” and the “exit door” of the bile-acid pathway.
Protecting fatty livers in a diet-stressed body
The crucial test was whether this gut-trained strain could actually protect an animal from liver damage. The researchers used a mouse model of NAFLD, in which a long-term high-fat diet causes weight gain, fatty accumulation in the liver, and inflammation. Mice were split into four groups: normal diet, high-fat diet alone, high-fat plus the original probiotic, and high-fat plus the adapted W5S9 strain. Both probiotic groups showed improvements over the high-fat group, but W5S9 consistently did better. These mice gained less weight, had healthier cholesterol profiles, showed lower markers of liver injury and inflammation, and had visibly fewer fat droplets in their liver tissue. Detailed chemical analysis of feces revealed that W5S9 more strongly reduced disease-associated bile acids and nudged the bile acid pool back toward a healthier balance, without dramatically reshaping the overall gut community.
What this means for future probiotic medicines
For non-specialists, the key message is that the researchers did not merely find a “good” probiotic—they used the body itself to sculpt a better one. By letting natural selection operate inside germ-free mice under a carefully designed high-fat, high-bile environment, they produced a strain that more efficiently handles bile acids and, as a result, better shields the liver from diet-induced damage. Because no foreign DNA was added, these evolved microbes remain non-GMO, which may ease regulatory and public acceptance. The broader implication is that similar host-guided evolution strategies could be tailored to other conditions, from inflammatory bowel disease to metabolic and even neurological disorders, opening the door to personalized, functionally tuned live microbial therapies.
Citation: Han, Z., Sun, Z., Liu, X. et al. Harnessing a germ‑free mouse gut bioreactor for directed evolution of probiotics to combat non-alcoholic fatty liver disease. Nat Commun 17, 3133 (2026). https://doi.org/10.1038/s41467-026-69823-0
Keywords: probiotics, gut microbiome, bile acids, fatty liver disease, directed evolution