Clear Sky Science · en
Beta cell-derived cholecystokinin drives obesity-associated pancreatic adenocarcinoma development
When Extra Weight Talks to the Pancreas
Obesity is known to raise the risk of pancreatic cancer, one of the deadliest cancers, but the biological “conversation” that links extra body fat to tumors has been murky. This study uncovers an unexpected messenger: a digestive hormone called cholecystokinin, or CCK, that is produced inside insulin-making cells of the pancreas. By tracing how these cells change under metabolic stress, the authors show how a hormone meant to help the body cope can instead fuel cancer, and suggest new ways to intercept the disease before it takes hold.
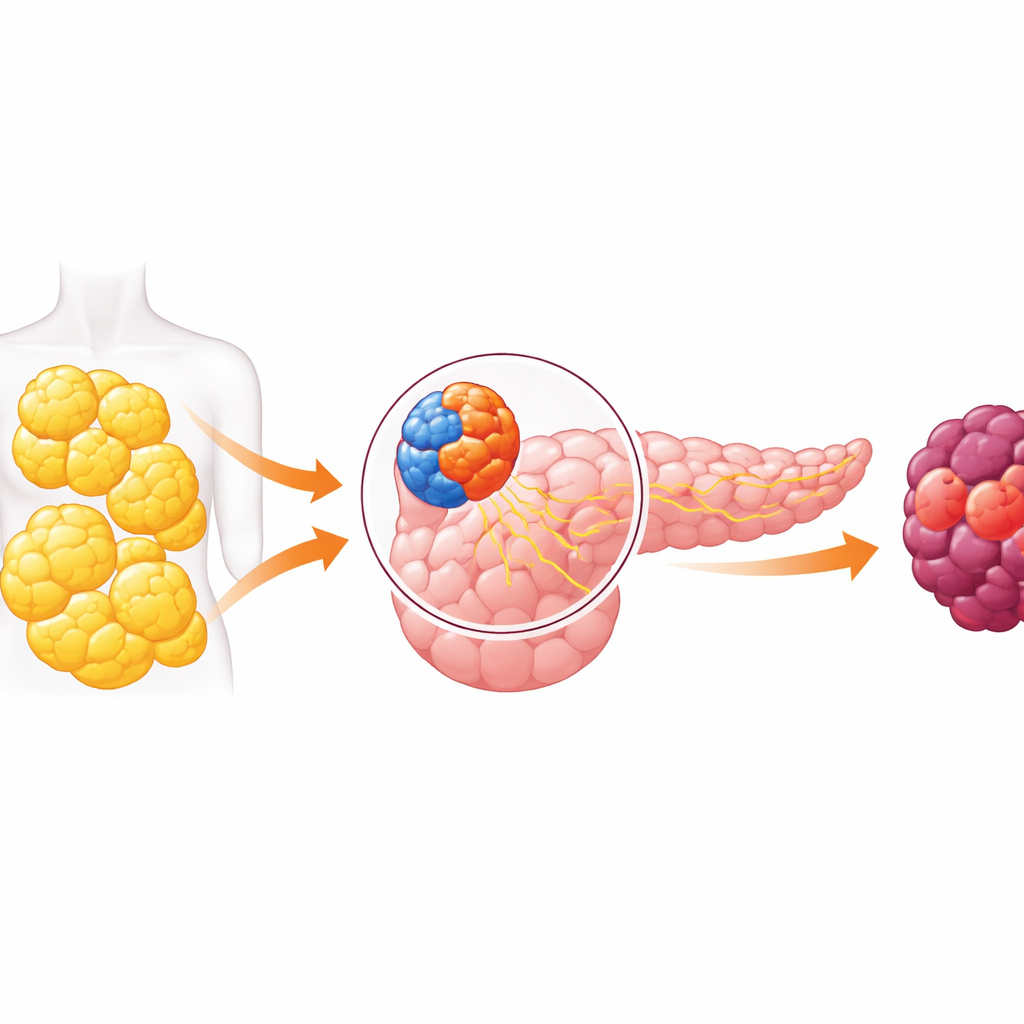
A Two-Sided Organ and a Dangerous Dialogue
The pancreas has two main jobs. One side, the endocrine part, contains tiny islands of cells that release hormones like insulin to control blood sugar. The other, the exocrine part, makes digestive juices and includes the cells that most often give rise to pancreatic cancer. For years, researchers assumed these compartments worked largely in isolation. Recent work has overturned that idea, showing that signals from hormone-producing cells can influence nearby digestive cells. This paper dives into that cross-talk and finds that, in obesity, it can push the digestive side of the pancreas toward cancer.
Stress in Hormone Cells Sparks a New Signal
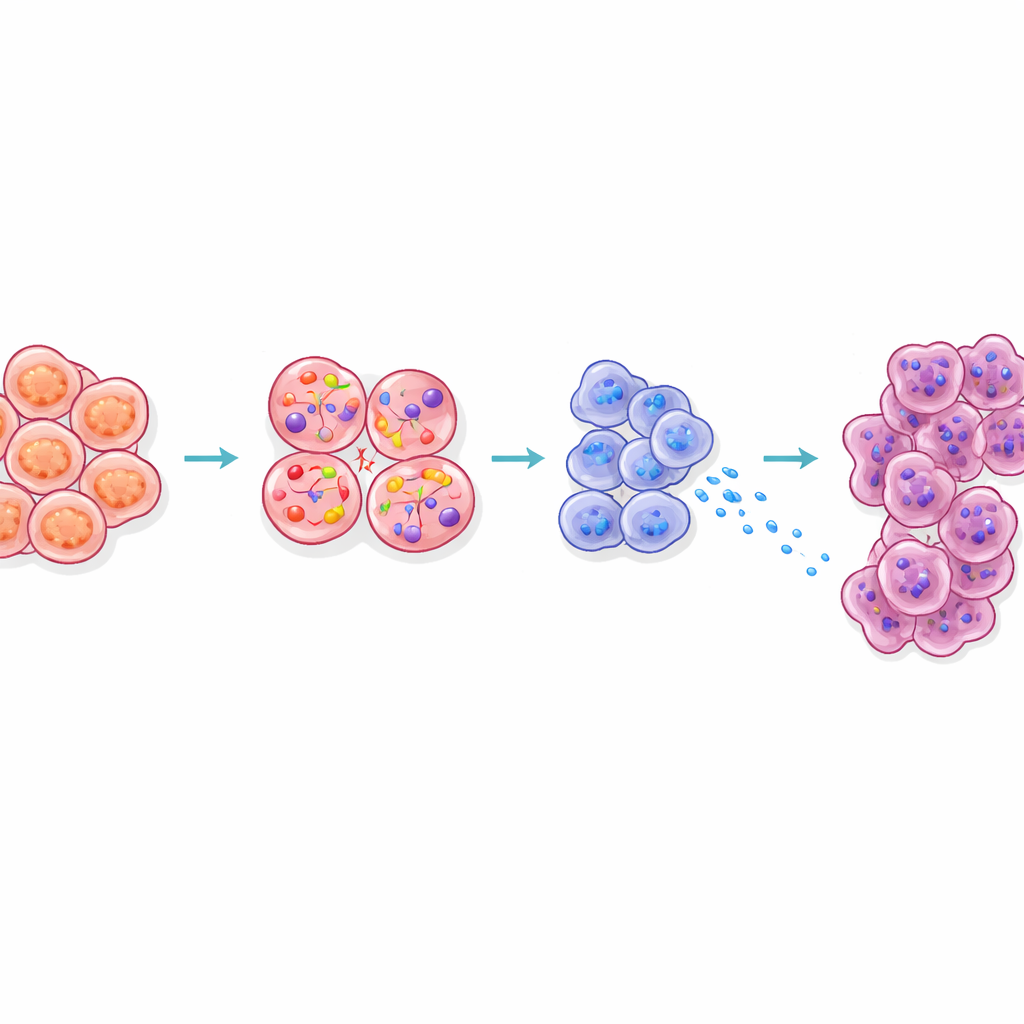
In obesity, insulin-producing beta cells are under constant pressure to meet the body’s rising demand for insulin. Using single-cell RNA sequencing and advanced computational tools, the researchers followed individual beta cells as mice progressed from lean to overweight to severely obese. They found that a subset of immature beta cells expanded and gradually lost their specialized insulin identity. As these cells became stressed, markers of protein-folding strain and cellular damage rose, while insulin production fell. At the same time, these cells began to make high levels of CCK, a hormone usually associated with the gut that can stimulate digestive cells to grow and change.
From Helpful Hormone to Cancer Fuel
To test whether this newly made CCK was merely a byproduct of stress or an active driver of disease, the team engineered mice whose beta cells produced extra CCK, even without obesity. These animals developed pancreatic pre-cancer and invasive tumors to a similar extent as obese mice, despite having normal weight and blood sugar. In contrast, when CCK was removed specifically from the pancreas of obese mice, tumor burden dropped sharply, even though the animals were still heavy and had high insulin levels. Across multiple mouse models, CCK levels in the pancreas closely tracked with the amount of tumor, while insulin levels tended to move in the opposite direction. This pointed to CCK—not insulin—as the dominant hormone linking obesity to pancreatic cancer in these systems.
How the Signal Reaches and Primes Neighboring Cells
The study also examined how CCK from beta cells reshapes the surrounding tissue. Acinar cells, which normally secrete digestive enzymes, sit right next to islets in the pancreas. In obese mice, acinar cells near islets displayed a stress signature, produced more digestive enzymes and proteins called Reg, and shifted into states known to be more easily transformed into cancer. Blocking CCK signaling in obese mice reduced these Reg signals. Three-dimensional imaging further revealed that early precancerous lesions tended to arise unusually close to islets in obese animals. Together, these results suggest that CCK diffusing from stressed beta cells creates a “hot zone” around islets where digestive cells are primed to become tumors.
Shared Patterns with Human Diabetes and a Key Switch
By comparing mouse data with human pancreatic tissue from people with and without type 2 diabetes, the authors found striking parallels: beta cells from diabetic donors mapped onto the same stressed, less mature states seen in obese mice. They then used a regulatory analysis to ask what turns on CCK in beta cells and identified a stress-responsive pathway centered on the protein cJun. When they blocked the upstream JNK–cJun pathway in obese mice or in beta-like cells in a dish, CCK levels fell, and cJun was seen binding near a regulatory region of the CCK gene. This suggests that chronic stress activates JNK–cJun, which in turn flips beta cells into a CCK-producing mode.
What This Means for People at Risk
To a lay reader, the central message is that, under the strain of obesity and diabetes, insulin cells in the pancreas do not just falter—they adapt in a way that inadvertently encourages nearby cells to form cancer. They do this by switching on CCK, a hormone that acts like a growth and stress signal for neighboring digestive cells, helping to create a fertile ground for tumors. Because CCK from the pancreas is not essential for maintaining blood sugar control, targeting this specific hormone source or its stress-activated switch could, in principle, reduce pancreatic cancer risk in people with obesity or type 2 diabetes without undermining overall hormone balance. The work reframes pancreatic cancer as a disease of misdirected communication between the organ’s two sides and highlights new avenues for early prevention.
Citation: Garcia, C.C., Venkat, A., McQuaid, D.C. et al. Beta cell-derived cholecystokinin drives obesity-associated pancreatic adenocarcinoma development. Nat Commun 17, 3292 (2026). https://doi.org/10.1038/s41467-026-69821-2
Keywords: pancreatic cancer, obesity, beta cells, cholecystokinin, type 2 diabetes