Clear Sky Science · en
Composite SMG5-SMG6 PIN domain formation is essential for NMD
How Cells Protect Themselves from Faulty Messages
Our cells constantly read genetic messages (mRNAs) to make proteins. But these messages sometimes contain early stop signals that would produce truncated, potentially harmful proteins. The process that destroys such faulty messages is called nonsense‑mediated mRNA decay (NMD). This paper uncovers how two key proteins, SMG5 and SMG6, physically cooperate to cut defective messages, explaining a long‑standing mystery in how cellular quality control works.
Finding the Hidden Partnership
For years, scientists knew that SMG6 can cut RNA directly, while SMG5 was thought to be “catalytically dead” and mainly a helper or scaffold. Yet experiments in cells showed something puzzling: SMG6 could not work properly without SMG5, and NMD collapsed if either one was missing. The authors used state‑of‑the‑art structure prediction (AlphaFold), biochemical assays with purified proteins, and genetic tests in human cell lines to resolve this contradiction. Their models predicted that the tail regions of SMG5 and SMG6, called PIN domains, dock next to each other to form a joint structure. This composite unit—dubbed a “cPIN”—was proposed to be the true cutting machine in NMD. 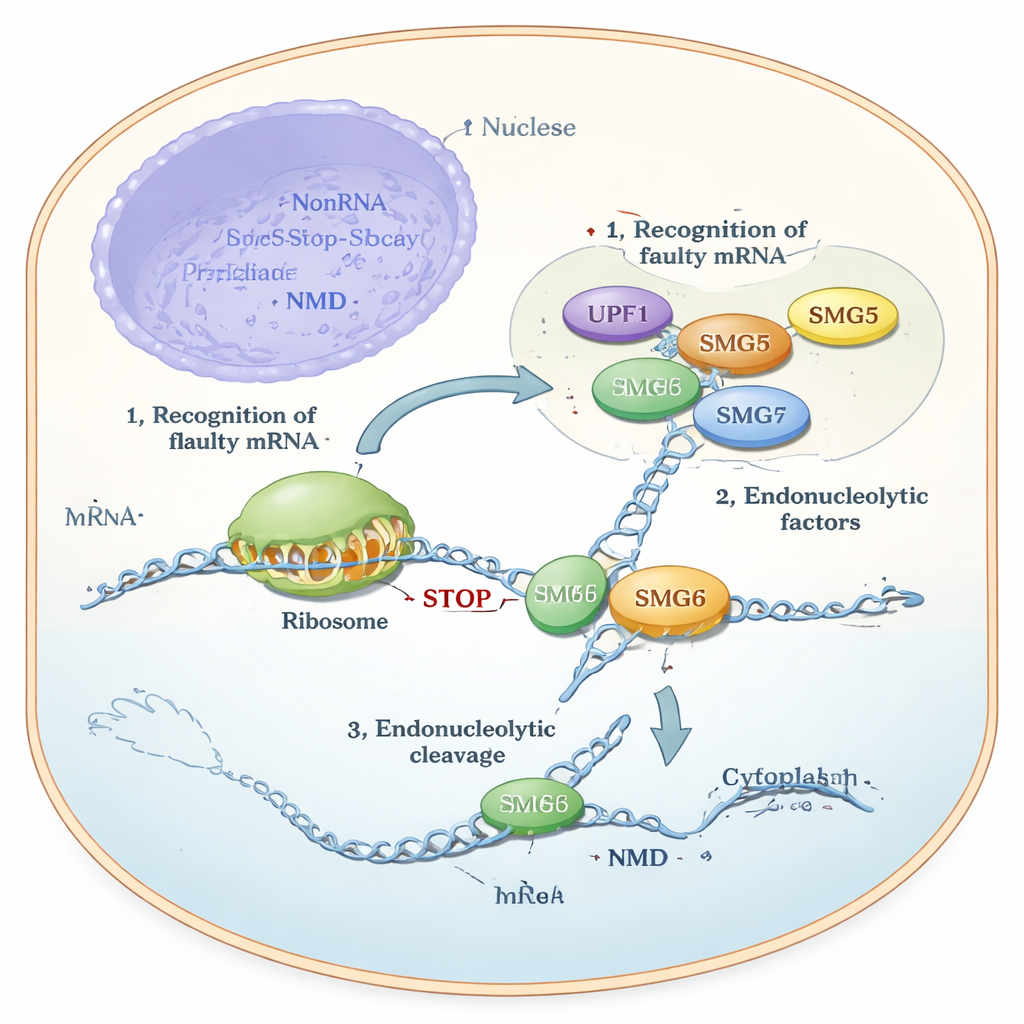
Rebuilding the Molecular Scissors in a Test Tube
To test the prediction, the team produced fragments of human SMG5 and SMG6 in bacteria and purified them. On their own, SMG6 showed only weak ability to cut a designed RNA substrate, and SMG5 alone showed virtually none. But when the two fragments were mixed, cutting activity rose sharply, even under conditions where contaminating enzymes were unlikely to play a role. The same effect appeared on both linear and circular RNA‑based test molecules, indicating that the enhanced cutting truly came from the SMG5–SMG6 pair. Chemical crosslinking and mass spectrometry further showed that the two proteins come into close contact, supporting the idea of a direct, if transient, partnership.
Completing the Cutting Edge
The structural models suggested exactly how SMG5 boosts SMG6. SMG6 contributes four acidic amino acids that hold metal ions at the heart of the cutting site, as is typical for this family of enzymes. Surprisingly, the models placed an additional acidic residue from SMG5 right next to them, effectively extending the catalytic pocket. Other positively charged residues on SMG5 were predicted to grip the RNA backbone and help position it for cleavage. When the researchers mutated these critical SMG5 or SMG6 residues, the composite complex lost much of its cutting power in vitro. The same mutations also failed to rescue NMD in engineered human cells where the normal protein had been depleted, tightly linking the structural model to real cellular function. 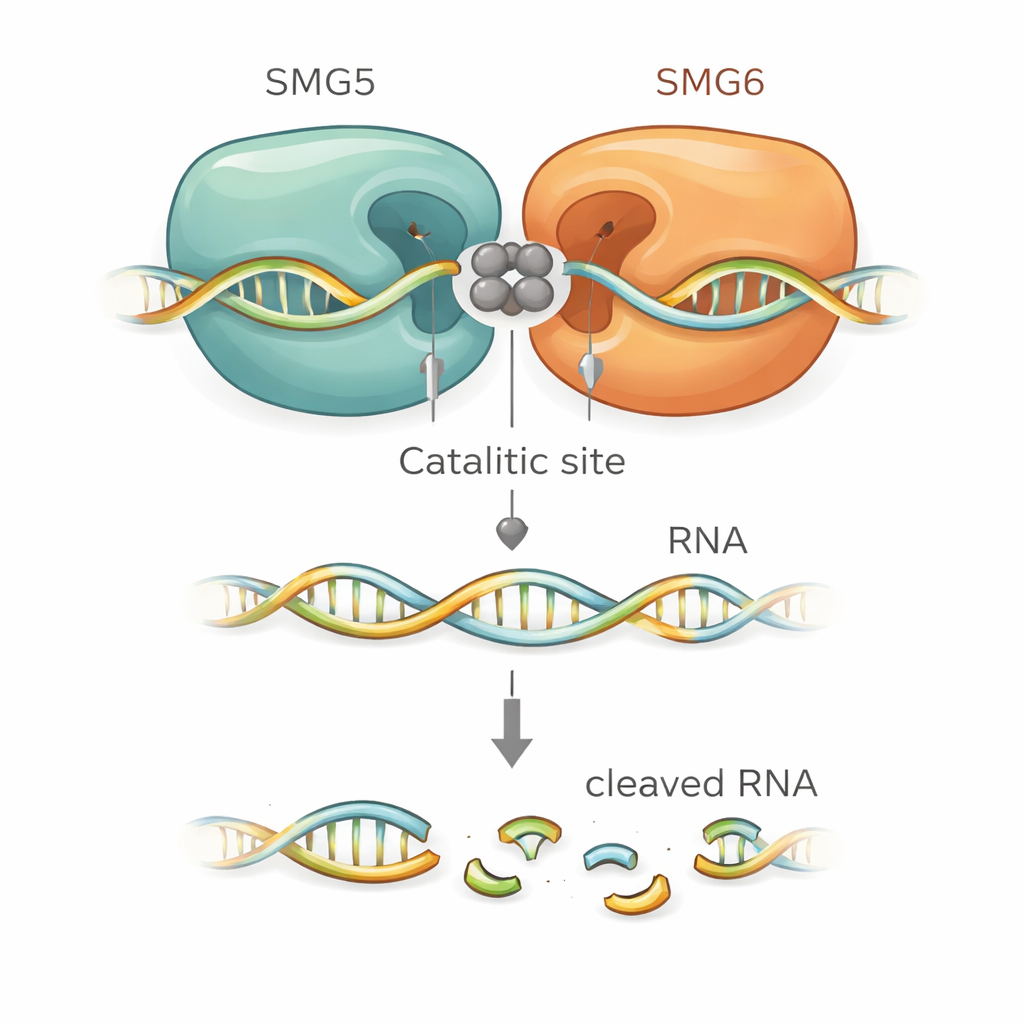
Proving that Both Partners Are Essential in Cells
Because completely removing SMG5 or SMG6 kills cells, the team used a “degron” system that lets them tag each protein so it can be rapidly destroyed with a small molecule. Combining this fast degradation with RNA interference allowed near‑total removal of SMG5, SMG6, or a central regulator called UPF1. Genome‑wide RNA sequencing then revealed what happens to NMD when each factor is lost. Depleting SMG5 or UPF1 produced almost identical changes in cellular RNAs, with faulty, NMD‑sensitive transcripts accumulating strongly. Removing SMG6 gave a very similar, though slightly milder, effect. These data show that SMG5 and SMG6 are not optional parallel branches of the pathway; rather, they act together with UPF1 as core components of one main decay route.
Why This Matters for Cellular Health
In simple terms, the study shows that SMG5 and SMG6 snap together to form a single, powerful molecular pair of scissors that cuts defective genetic messages. SMG6 provides most of the blade, but SMG5 supplies a missing edge and helps hold the RNA in place, turning a weak cutter into an efficient one. This composite “cPIN” explains why cells absolutely require both proteins to keep their RNA messages clean. By clarifying how NMD’s key cutting step is switched on only when faulty messages are recognized, the work offers a clearer picture of how cells prevent the buildup of toxic, truncated proteins and finely tune gene expression.
Citation: Kurscheidt, K., Theunissen, S., Pasquali, N. et al. Composite SMG5-SMG6 PIN domain formation is essential for NMD. Nat Commun 17, 1934 (2026). https://doi.org/10.1038/s41467-026-69819-w
Keywords: nonsense-mediated mRNA decay, RNA quality control, SMG5 SMG6, mRNA surveillance, gene expression regulation