Clear Sky Science · en
Computation and resource efficient genome-wide association analysis for large-scale imaging studies
Peering Into the Genetic Blueprint of the Brain
Why do some people’s brains age more gracefully, resist mental illness, or support stronger memory and learning? Modern brain scans and genetic tests promise answers, but the sheer volume of data has been overwhelming. This study introduces a new way to connect tiny DNA differences to detailed brain images, making it finally practical to search the entire genome against millions of brain measurement points. The approach not only slashes computing costs and storage needs, it also uncovers hidden genetic patterns that link specific brain regions to traits like education, depression, and schizophrenia.
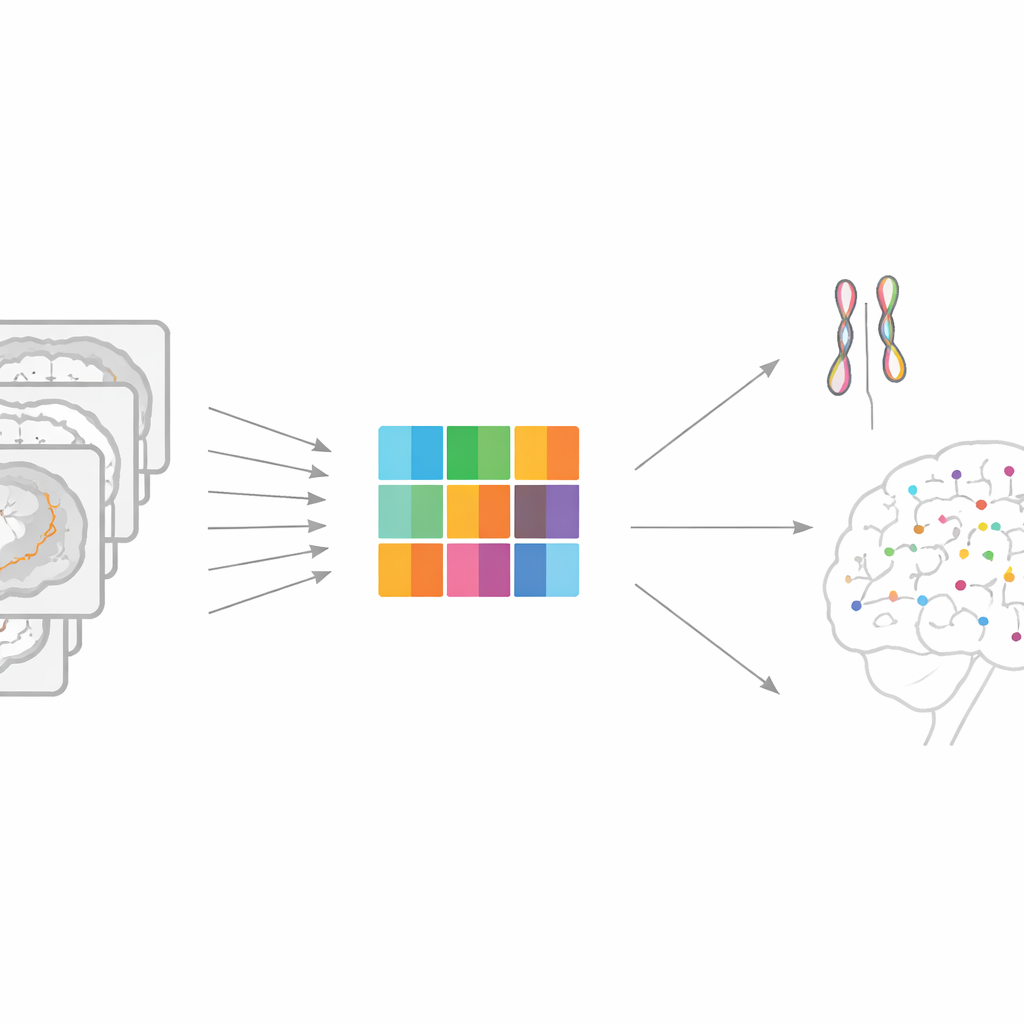
From Blurry Averages to Detailed Brain Maps
Most large genetic studies of the brain simplify images into a few hundred summary measurements, like the overall volume of a region. That shortcut makes analysis feasible, but it blurs away fine-grained detail. Each brain scan actually contains tens of thousands of tiny locations, or voxels, where structure and wiring can vary. A direct “voxel-by-voxel” scan across the whole genome would be ideal scientifically, but in practice it explodes into trillions of tests, requiring enormous computing power and producing summary files that are too large to share or reuse.
A Smarter Way to Compress Brain Images
The authors propose a framework called Representation learning-based Voxel-level Genetic Analysis (RVGA) to tackle this bottleneck. RVGA first cleans up each brain image by separating smooth, meaningful structure from random scanner noise. It then learns a small set of underlying patterns—like basic building blocks of shape and texture—that can be combined to reconstruct the original image. Each individual’s brain is summarized not by every voxel, but by scores on these patterns, reducing the data size by one to three orders of magnitude while retaining most of the signal. These pattern scores are then treated as traits in a standard genome-wide association study, which is much faster to run.
Rebuilding the Full Picture From Small Pieces
Crucially, RVGA does not stop at these compressed traits. Using the learned patterns, it mathematically “projects back” the genetic findings from the pattern level to every voxel in the image. This trick allows researchers to recover detailed, voxel-level association maps without ever having to fit billions of separate models. All that needs to be stored and shared are three compact ingredients: the genetic results for the patterns, the image patterns themselves, and the way the pattern scores vary across people. From this minimal “triplet,” RVGA can reconstruct full-resolution maps of genetic effects, estimate how much genetics contributes to variation at each voxel, and compute how genetics is shared between voxels and with outside traits.
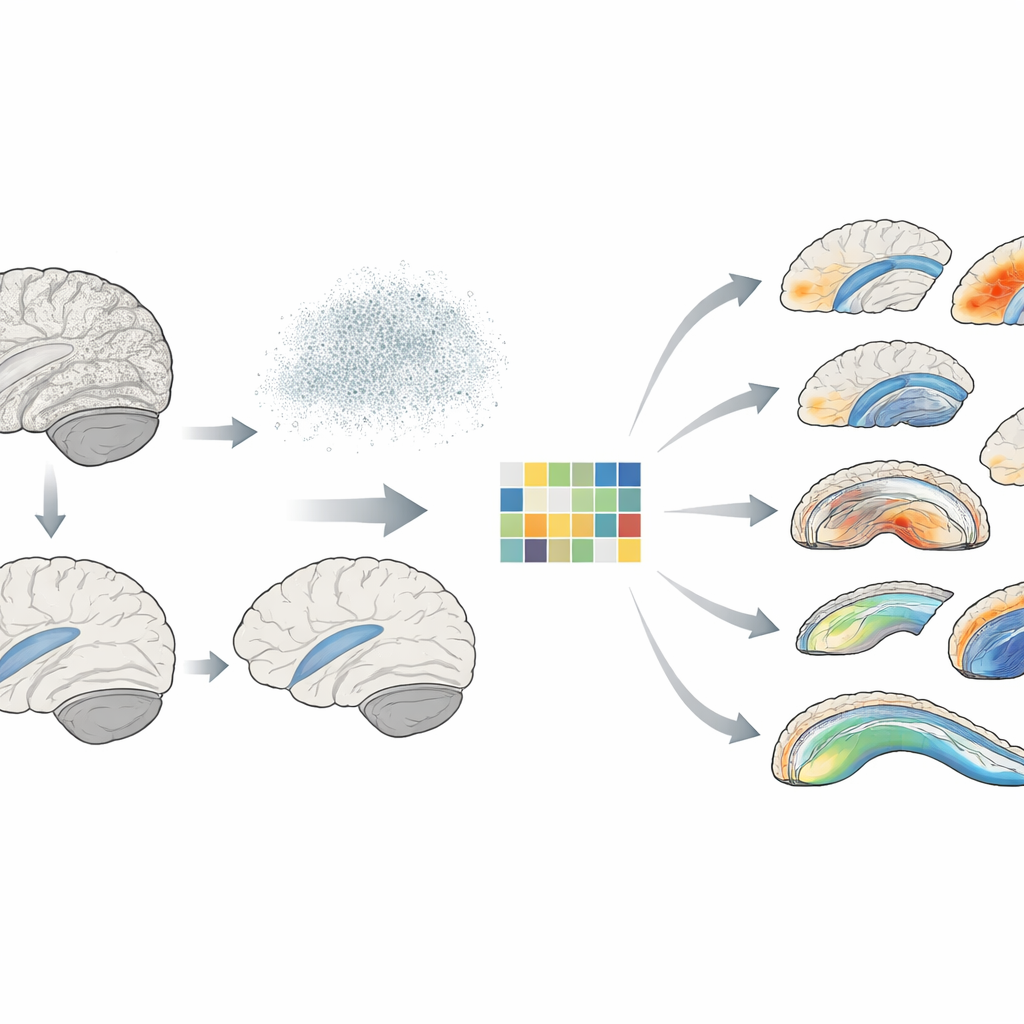
What the New Method Reveals in Real Brains
The team applied RVGA to brain scans and genetic data from more than 53,000 participants in the UK Biobank. They focused on the detailed shape of the hippocampus—crucial for memory—and the fine structure of major white matter pathways that connect different brain regions. Using RVGA, they identified 39 previously unreported genetic regions affecting hippocampal shape and 275 new regions influencing white matter microstructure, while also replicating many known findings. The method cut the size of the resulting genetic summary files by about 229-fold, making them far easier to share. It also revealed that genetic influence is far from uniform: some hippocampal subregions showed much higher heritability than others, and certain white matter segments carried especially strong genetic signatures.
Links to Education, Mood, and Mental Illness
Because RVGA can be combined with genetic results from other studies, the authors built “atlases” of how brain voxels share genetic roots with brain disorders and related traits. They found, for example, that parts of the hippocampal tail and nearby structures share positive genetic links with educational attainment, while another subregion, the presubiculum, shows a negative link. In white matter, specific segments of the anterior corona radiata shared genetic influences with schizophrenia, and parts of the corpus callosum showed negative genetic ties with bipolar disorder. Many of these patterns confirm earlier region-level findings, but RVGA refines them to precise subregions, hinting at more targeted biological pathways.
Why This Matters for Brain Health
By making ultra-detailed genetic scans of the brain both feasible and shareable, RVGA opens the door to a new generation of imaging genetics studies. Researchers can now see exactly which tiny patches of brain tissue are influenced by particular genetic variants, how strongly, and in what way these patches share genetic roots with cognition and mental illness. Over time, such maps may help pinpoint biological circuits that could be monitored, protected, or even targeted in personalized treatments. The method also generalizes beyond the brain to other imaging-rich organs, promising a broader shift from blurry averages to high-resolution genetic insight.
Citation: Jiang, Z., Stein, J., Li, T. et al. Computation and resource efficient genome-wide association analysis for large-scale imaging studies. Nat Commun 17, 3313 (2026). https://doi.org/10.1038/s41467-026-69816-z
Keywords: imaging genetics, brain MRI, genome-wide association, hippocampus, white matter