Clear Sky Science · en
Tumor cell villages define the co-dependency of tumor and microenvironment in liver cancer
Why tiny “villages” inside tumors matter
Cancer is often described as a mass of rogue cells, but this study of liver cancer shows a far more organized picture. Inside tumors, the cells arrange themselves into tiny “villages” that cooperate with nearby support cells, blood vessels, and immune cells. Understanding how these miniature communities form and function could reveal why some tumors are more aggressive than others and point to new ways to disrupt them.
Looking at tumors cell by cell in their native habitat
Most modern cancer studies pull cells out of the tumor and read their genetic activity in a test tube, which loses crucial information about where each cell sat and who its neighbors were. In this work, researchers combined two powerful approaches in samples from seven patients with primary liver cancer. They used a high‑resolution imaging platform to measure RNA molecules directly in slices of tumor tissue, mapping over 2.3 million cells in place, and paired this with single‑cell RNA sequencing from the same tumors. This let them see not only what each cell was doing, but also where it lived within the tumor and what types of cells surrounded it. 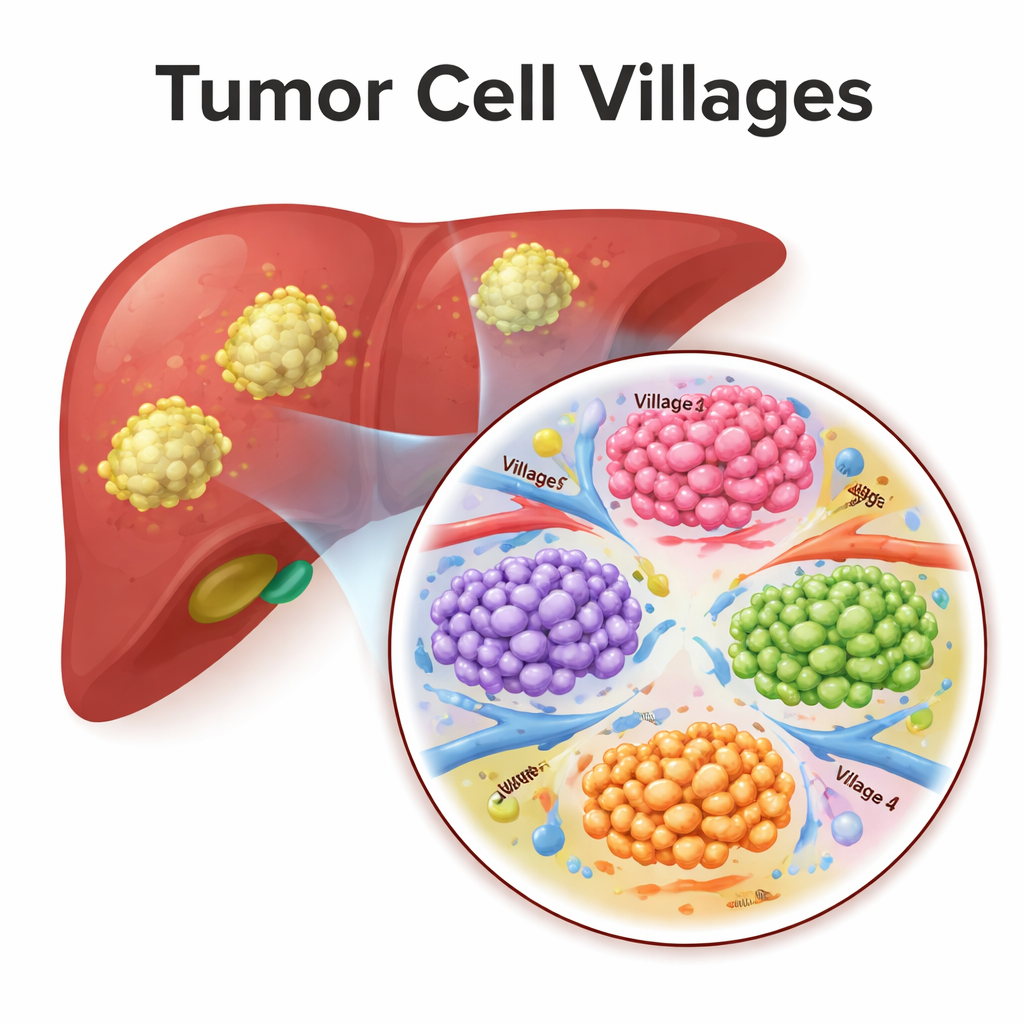
Discovering distinct cancer cell “personalities”
By analyzing gene activity patterns, the team identified a dozen different “states” of malignant liver cancer cells. Some were focused on rapid division, others on repairing stress, responding to immune attack, moving and invading nearby tissue, or mimicking normal liver or bile duct cells. These states closely matched patterns seen in other cancer types, suggesting common themes across tumors, but with a liver‑specific twist. Normal, non‑cancerous epithelial cells in the same samples showed a much narrower range of states, highlighting how unusually diverse the tumor cells had become.
The neighborhood shapes who the cancer cells become
The researchers then asked how the local neighborhood influences these cancer cell states. For every malignant cell, they cataloged all neighboring cells within about the width of a human hair and grouped similar neighborhoods into what they called Spatial Dynamics Networks. Some neighborhoods were packed with blood vessels, others were dominated by tumor cells, while still others were rich in immune cells or fibroblasts (cells that build connective tissue). Cancer cells in a given state did not appear at random. For example, cells undergoing a shape‑shifting process linked to invasion tended to sit near dense networks of blood vessels, while highly dividing cells clustered in tumor‑heavy regions with few non‑tumor cells. This non‑random pattern shows that the microenvironment and cancer cell behavior are tightly intertwined.
Tumor “villages” as cooperative ecosystems
To move beyond single cells, the team used a type of deep learning model that treats each tumor as a graph of connected cells. The model learned from both the internal state of each malignant cell and the type of neighborhood it occupied. Clustering these learned representations revealed eight recurring tumor “villages.” Each village mixed specific cancer cell states with characteristic surroundings—for instance, some villages combined blood‑vessel‑rich areas with invasive cells, while others resembled “germinal centers” of fast‑dividing tumor cells. When the scientists looked at hundreds of additional liver cancer patients using other datasets, they found that patients whose tumors were dominated by certain villages (labelled 3–8) tended to have worse survival than those enriched for villages 1 and 2, suggesting that village composition captures how dangerous a tumor’s ecosystem is. 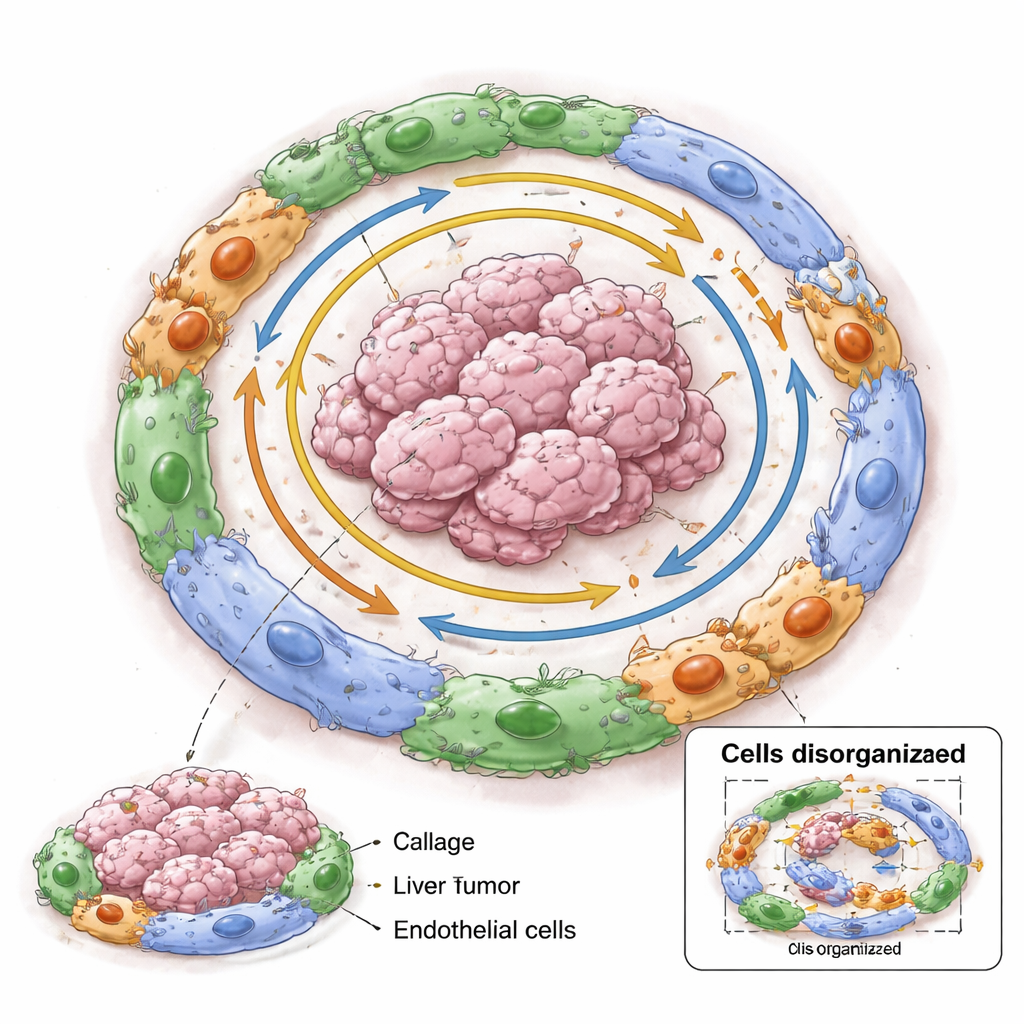
Molecular deals between cancer cells and their neighbors
Within each village, the researchers searched for pairs of genes—one active in tumor cells, one in nearby non‑tumor cells—that rose and fell together in space. These “co‑dependent” pairs varied from village to village and weakened as the distance between cells increased, pointing to highly local molecular relationships. One striking example involved a protein called SPINK1 made by tumor cells and collagen‑producing fibroblasts known as matrix cancer‑associated fibroblasts. SPINK1‑positive tumor cells were frequently found next to these fibroblasts, and signaling between them, likely through a known receptor called EGFR, was linked to features of metastasis. When the researchers simulated “shuffling” the neighbors around tumor cells in their models, the ability to correctly recognize villages dropped, especially in those rich in SPINK1‑linked interactions, implying that these molecular deals help hold village structure together.
What this means for patients
For a lay reader, the key message is that liver tumors are not random piles of cells. They self‑organize into small communities—villages—where cancer cells and surrounding support cells depend on each other to grow, spread, and resist treatment. Some village types are tied to better survival, others to worse, and each harbors its own set of molecular dependencies. In the future, therapies might be designed not just to kill individual cancer cells, but to break up or starve specific villages by targeting the relationships between tumor cells and their neighbors, potentially making the whole tumor less resilient.
Citation: Liu, M., Hernandez, M.O., Castven, D. et al. Tumor cell villages define the co-dependency of tumor and microenvironment in liver cancer. Nat Commun 17, 1986 (2026). https://doi.org/10.1038/s41467-026-69797-z
Keywords: liver cancer, tumor microenvironment, spatial transcriptomics, intratumor heterogeneity, cancer cell states