Clear Sky Science · en
S-nitrosoglutathione reductase GSNOR drives age-related obesity by promoting adipose tissue whitening through de-nitrosation of Beclin-1
Why midlife weight gain matters
Many people notice that staying lean becomes harder in their 40s and 50s, even without eating more or exercising less. This midlife weight gain raises the risk of diabetes, heart disease, and other illnesses, yet its biological causes are still being uncovered. This study reveals a previously hidden culprit in fat tissue itself: an enzyme called GSNOR that changes how fat cells handle energy as we age, pushing them to store more and burn less.
Heat-making fat versus storage fat
Our bodies contain more than one kind of fat. Classic white fat is the familiar energy store, made of large, pale cells packed with a single big droplet. Beige and brown fat, in contrast, are packed with tiny droplets and mitochondria, the cell’s “furnaces” that burn fuel to produce heat. In youth, having more beige fat helps keep weight and blood sugar in check. As animals and people age, however, this beige fat tends to “whiten”: the mitochondria are cleared out, cells swell with stored fat, and overall metabolism slows.
An aging switch in fat tissue
The researchers discovered that GSNOR levels rise specifically in subcutaneous white fat from middle-aged mice and men, but not in all fat depots. GSNOR controls a chemical signal based on nitric oxide that modifies many proteins. When the team deleted the GSNOR gene in mice, these animals largely resisted age-related weight gain. They carried less fat, more lean mass, had better blood sugar control, and burned more energy, yet they did not eat less or move more than normal mice. Their inguinal fat pads contained smaller cells and more beige-like features, with higher mitochondrial markers and stronger heat-producing activity.
Turning up GSNOR drives whitening and obesity
To test the opposite direction, the scientists boosted GSNOR only in fat cells, either by viral delivery to one fat depot or by creating a knock-in mouse that overexpressed the enzyme in adipose tissue. In both cases, fat cells became larger and paler, mitochondrial markers fell, and genes linked to beige fat and heat production were suppressed. The knock-in mice were visibly obese, less able to maintain body temperature in the cold, and showed lower oxygen consumption and energy expenditure. Importantly, this shift did not stem from extra food intake or reduced fat breakdown, pointing instead to a reprogramming of fat cell identity toward energy storage.
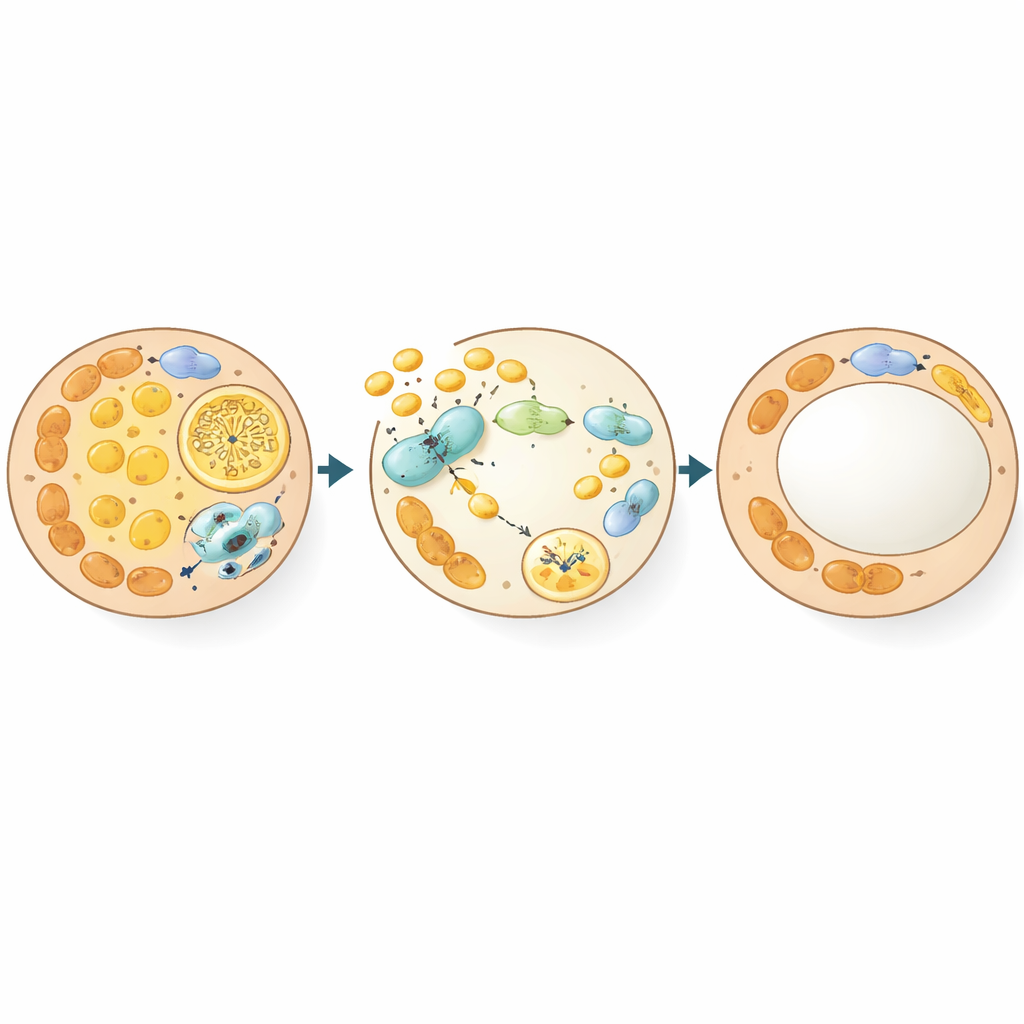
A hidden brake on cellular recycling
Diving deeper, the team asked how GSNOR reshapes fat cells at the molecular level. Using sensitive proteomic methods, they mapped proteins whose nitric-oxide-based modification changed when GSNOR was high. A central target emerged: Beclin-1, a key organizer of autophagy, the process by which cells recycle components, including mitochondria. Normally, nitric oxide can attach to Beclin-1 at a specific cysteine site, acting as a brake on autophagy. When GSNOR levels rise, this modification is removed, Beclin-1 binds more strongly to its partner ATG14, and autophagy ramps up. In fat cells, this heightened recycling preferentially clears mitochondria, tipping beige cells back toward the whiter, storage-oriented state. Electron microscopy and molecular markers confirmed that GSNOR-rich or Beclin-1–mutant fat had more autophagosomes and fewer mitochondria during the beige-to-white transition.
Rewiring Beclin-1 confirms the mechanism
To prove that this single site on Beclin-1 matters, the authors engineered mice in which that cysteine was replaced so it could no longer carry the nitric-oxide tag. These Beclin-1C351A animals showed more autophagy in fat, faster loss of beige features, and greater whitening after cold or drug-induced browning was withdrawn, even though their overall body weight did not immediately change. In cell cultures, blocking GSNOR could keep beige traits only when Beclin-1 still had its modifiable site; once mutated, GSNOR loss no longer helped. Finally, in middle-aged mice, locally silencing GSNOR in one fat depot restored beige markers, shrank fat cells, and reduced local fat mass, underscoring that this pathway can be manipulated in adult animals.
What this means for midlife weight gain
In plain terms, this work identifies GSNOR as an age-activated switch in fat tissue that accelerates the loss of “good,” heat-making beige fat by over-activating the cell’s recycling machinery through Beclin-1. As GSNOR rises in midlife, it strips a protective nitric-oxide tag from Beclin-1, allowing excess cleanup of mitochondria, whitening of fat, slower metabolism, and increased susceptibility to obesity. The findings suggest that drugs which gently inhibit GSNOR in subcutaneous fat—or otherwise restore the nitric-oxide mark on Beclin-1—could help preserve beige fat function and combat age-related weight gain and metabolic decline.
Citation: Qiao, X., Xie, T., Zhang, Y. et al. S-nitrosoglutathione reductase GSNOR drives age-related obesity by promoting adipose tissue whitening through de-nitrosation of Beclin-1. Nat Commun 17, 3059 (2026). https://doi.org/10.1038/s41467-026-69793-3
Keywords: age-related obesity, beige fat, GSNOR, autophagy, nitric oxide signaling