Clear Sky Science · en
Dynamic transcriptomic remodeling in grafted human neural progenitor cells uncovers mechanisms for vision preservation in a rat model of retinitis pigmentosa
Why protecting fading vision matters
Retinitis pigmentosa is a group of inherited diseases that slowly rob people of their sight, often starting with night blindness and advancing toward legal blindness. Because hundreds of different gene mutations can cause this condition, treating each one individually with gene therapy is difficult. This study explores a more universal approach: transplanting human neural progenitor cells—immature nerve cells grown in the lab—into the eye to protect the remaining light-sensing cells. Understanding exactly how these grafted cells behave and help preserve vision could shape future treatments not just for retinitis pigmentosa but for other blinding disorders as well.
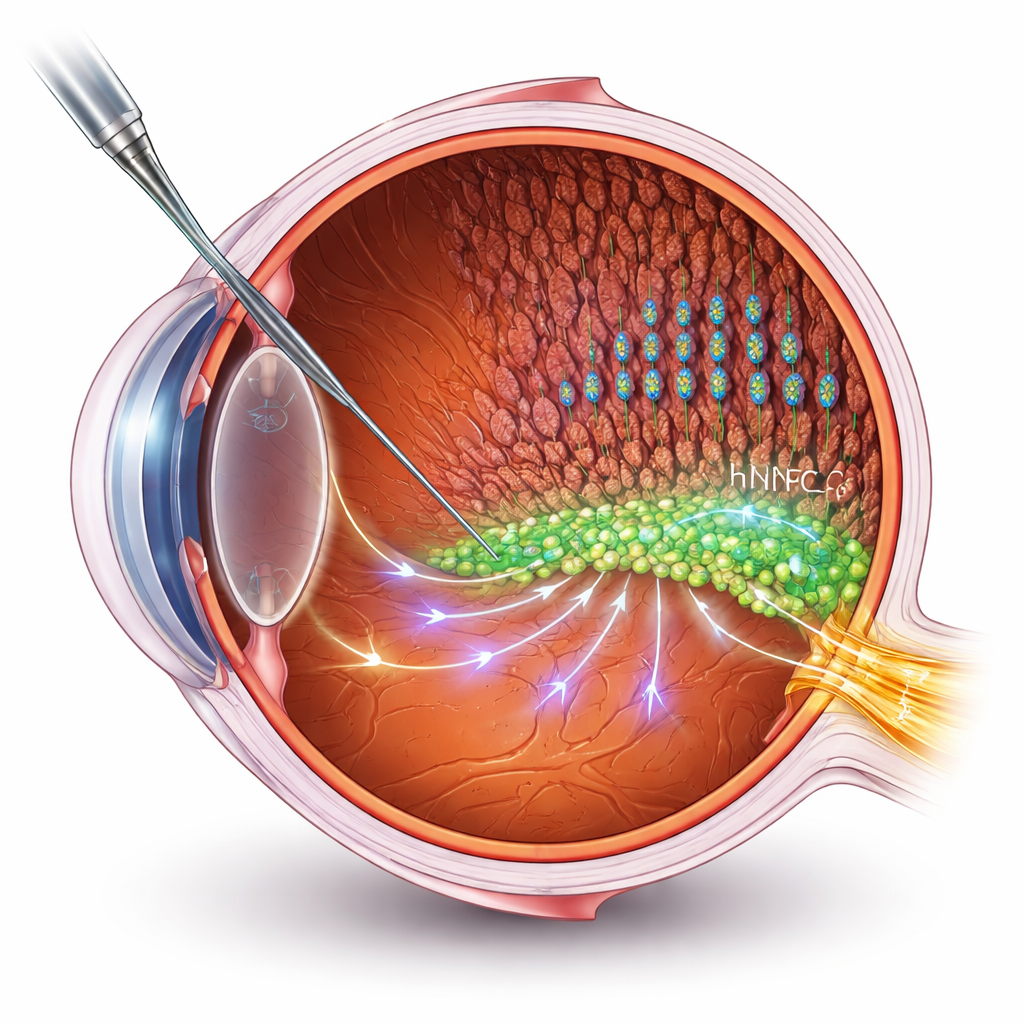
Helping cells instead of replacing them
Many stem cell therapies aim to replace dead or damaged cells. But in advanced retinal diseases, building new connections to the brain is a major technical hurdle. Here, the researchers take a different route: using human neural progenitor cells (hNPCs) as “helpers” that support the patient’s own retinal cells. They transplanted hNPCs into the subretinal space of a well-known rat model of retinitis pigmentosa, then measured vision using electrical recordings and behavioral tests. The treated rats kept better visual function and more intact layers of photoreceptors—the rods and cones that capture light—than untreated animals, even though degeneration still slowly progressed.
How grafted cells change inside the diseased eye
To see what happens at a molecular level, the team used single-cell RNA sequencing, a technique that profiles which genes are turned on in thousands of individual cells. They followed the grafted hNPCs at two time points, roughly early and later stages after transplantation. Instead of turning into new photoreceptors, most of the transplanted cells matured into astrocyte-like support cells, with a smaller fraction showing markers of oligodendrocytes or immature neurons. Over time these cells stabilized, adopting features that suggest they are tuned to survive in a stressful, degenerating environment and to interact closely with neighboring retinal cells.
Protective signals, metabolism, and cleanup
The hNPCs produced a rich mix of protective proteins known as trophic factors. Among them were MANF and MYDGF, which are linked to cell survival, reduced oxidative stress, and dampened inflammation, as well as other factors like midkine and pleiotrophin that support growth and movement of neural cells. These molecules appeared to act both on the graft itself and on the surrounding retina. In treated rats, rods and cones showed gene activity patterns closer to healthy controls, with improved support for visual signaling and cell maintenance. The grafted cells also boosted pathways involved in clearing cellular debris and adjusting metabolism, suggesting they help photoreceptors cope with energy demands and toxic byproducts that build up during disease.
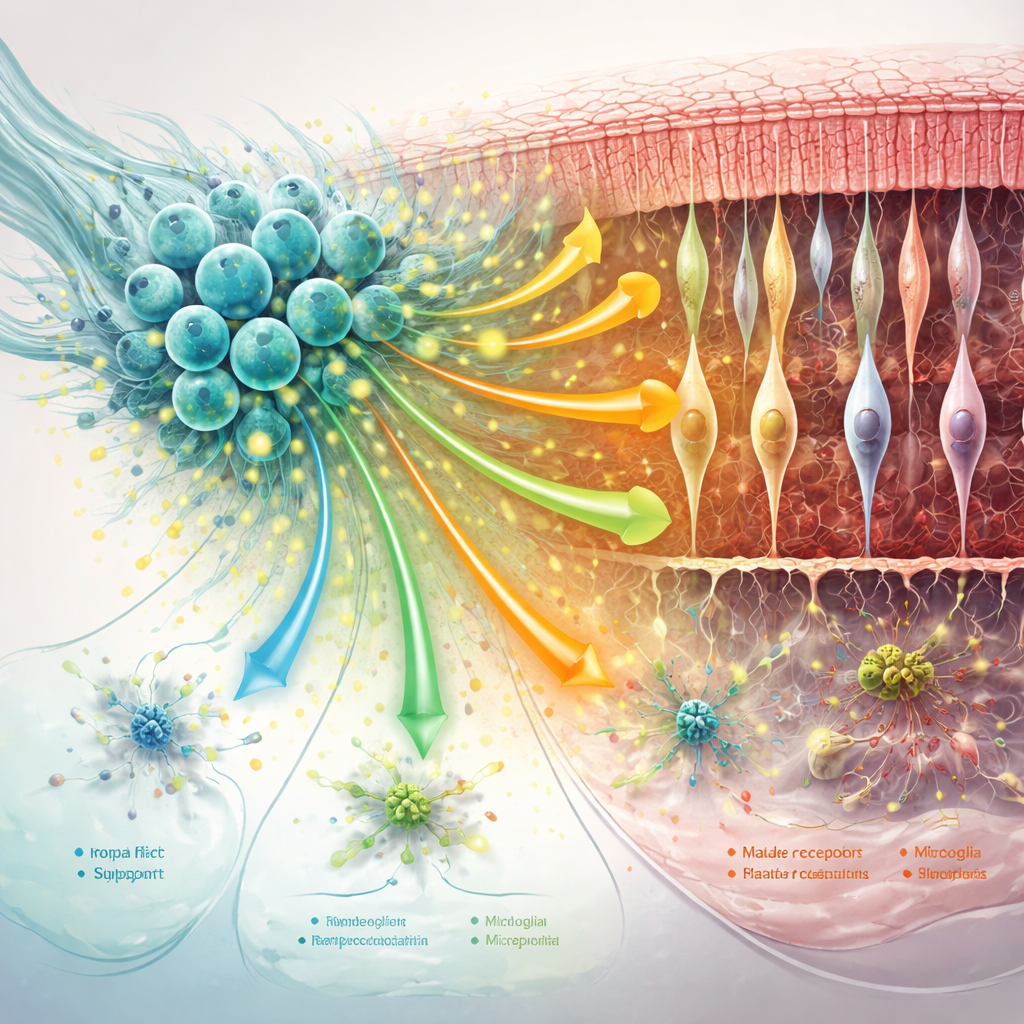
Calming support cells and reshaping the retinal neighborhood
The retina’s support network—Müller glia, microglia, and the surrounding matrix of proteins—plays a big role in either worsening or slowing degeneration. The study found that hNPC treatment toned down key markers of harmful “reactive” changes in Müller glia and shifted microglia away from overly inflammatory states, especially at earlier time points. At the same time, communication signals that help stabilize synapses, stick cells together, and maintain the scaffold around photoreceptors were strengthened. As the disease advanced, some of these benefits faded: overall cell-to-cell communication weakened, certain protective pathways—including MANF-related signaling—declined, and support cells adopted more stress- and immune-focused profiles.
What this means for future eye therapies
For a non-specialist, the take-home message is that transplanted human neural progenitor cells can act like on-site “first responders” in a degenerating retina. They do not rebuild the retina from scratch, but they release protective molecules, adjust metabolism, calm damaging inflammation, and help keep the local structure intact, all of which slow vision loss. However, this protective effect wanes over time as the environment becomes more hostile and the dialogue between graft and host cells weakens. The authors suggest that boosting key factors such as MANF and improving the health of the host retina—possibly through gene engineering of the graft or combination treatments—may be essential to turn a temporary reprieve into long-lasting preservation of sight.
Citation: Shahin, S., Bell, S., Lu, B. et al. Dynamic transcriptomic remodeling in grafted human neural progenitor cells uncovers mechanisms for vision preservation in a rat model of retinitis pigmentosa. Nat Commun 17, 2164 (2026). https://doi.org/10.1038/s41467-026-69776-4
Keywords: retinitis pigmentosa, stem cell therapy, retinal degeneration, neuroprotection, single-cell RNA sequencing