Clear Sky Science · en
Trivalent titanium in high-titanium lunar ilmenite
Why Moon Rocks Still Hold New Surprises
The Moon may look quiet and familiar in the night sky, but the rocks brought back by the Apollo astronauts are still revealing hidden stories about how our nearest neighbor formed and evolved. This study focuses on one such rock from the Apollo 17 mission and shows that a common lunar mineral, ilmenite, holds chemical clues to just how oxygen‑poor, or "reducing," the Moon’s interior once was. Those clues not only refine our picture of ancient lunar volcanoes but also point to a simple new way to read the redox history of other airless worlds.
A Special Basalt from the Lunar Seas
The researchers examined a high‑titanium lava rock, Apollo sample 75035, formed about 3.8 billion years ago during a peak in lunar volcanic activity. This basalt comes from a lava "sea" and is unusually rich in the dark mineral ilmenite, which stores much of the rock’s titanium. Because ilmenite crystals formed early and in large amounts in this rock, they likely captured the chemical conditions of the molten magma as it cooled. Before looking at the tiniest details, the team confirmed that their chip of 75035 was representative of the whole rock and that its surface had not been altered by space weathering—tiny impacts and solar wind damage that can mask a mineral’s original history.
Looking Inside Minerals Atom by Atom
To probe the ilmenite, the team used advanced electron microscopes and a technique called electron energy loss spectroscopy, which measures how electrons lose energy as they pass through a thin slice of material. This allows scientists to map both which elements are present and what electrical charge, or "valence," those atoms have. Multiple methods—including X‑ray fluorescence, X‑ray tomography, and elemental mapping—showed that ilmenite in 75035 contains more titanium than expected from its usual chemical recipe, where iron and titanium are normally present in a simple one‑to‑one ratio. Crucially, the crystal structure looked well ordered, so the extra titanium could not easily be explained by defects or impurities.
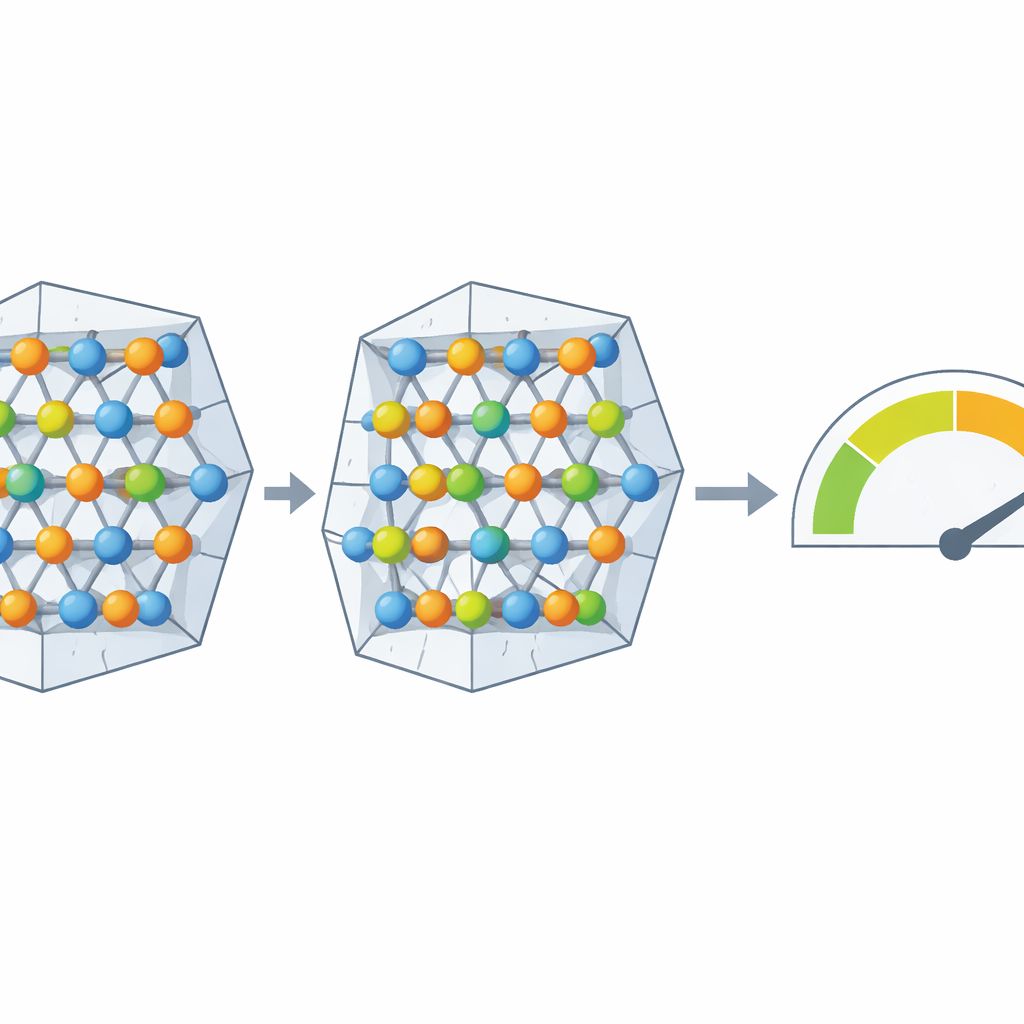
A New Form of Titanium in Lunar Ilmenite
The key question was what form this excess titanium takes. By closely examining the fine structure of titanium’s spectral "edges," the team detected a fingerprint of trivalent titanium, a form with one fewer positive charge than the more common tetravalent state. Their measurements indicate that about 13% of the titanium atoms in 75035 ilmenite are trivalent. This amount neatly matches the observed excess of titanium over iron, implying a revised ideal formula in which some iron is replaced and both cation sites in the crystal are partly filled by trivalent titanium. Because the sample shows no space‑weathering features, the team attributes this unusual titanium entirely to the conditions in the original magma, not to later surface processes.
Clues to an Oxygen‑Poor Moon
Trivalent titanium forms more readily in environments that are very poor in oxygen. The authors compared their measurements with existing laboratory experiments in which ilmenite and related minerals were grown at known temperatures and oxygen levels. Extrapolating those relationships, they estimate that the magma that crystallized 75035 ilmenite had an oxygen fugacity at least 1.6 orders of magnitude below the standard iron–wüstite reference buffer, placing it among the more strongly reducing conditions known for lunar magmas. A survey of hundreds of previously analyzed lunar ilmenite grains suggests that similar excess titanium is widespread in samples from many missions, hinting that trivalent titanium may be common across the Moon.
From Moon Mineral to Planetary Gauge
Because ilmenite is abundant and widely distributed in lunar rocks, tying its titanium valence to oxygen conditions opens the door to a powerful new tool. If experiments can more precisely calibrate how the proportion of trivalent titanium in ilmenite varies with temperature and oxygen level, this single mineral could serve as an "oxybarometer"—a built‑in gauge of how oxidizing or reducing a magma once was. That would let scientists extract detailed redox histories from very small rock chips, not only for the Moon but also for other worlds where oxygen is scarce, such as Mercury or certain asteroids, deepening our understanding of how rocky bodies differentiate and evolve over time.
Citation: Vira, A.D., Burgess, K.D., First, E.C. et al. Trivalent titanium in high-titanium lunar ilmenite. Nat Commun 17, 2712 (2026). https://doi.org/10.1038/s41467-026-69770-w
Keywords: lunar basalts, ilmenite, trivalent titanium, oxygen fugacity, planetary volcanism